|
Carbene
In organic chemistry, a carbene is a molecule containing a neutral carbon atom with a valence of two and two unshared valence electrons. The general formula is or where the R represents substituents or hydrogen atoms. The term "carbene" may also refer to the specific compound , also called methylene, the parent hydride from which all other carbene compounds are formally derived. Carbenes are classified as either singlets or triplets, depending upon their electronic structure. Most carbenes are very short lived, although persistent carbenes are known. One well-studied carbene is dichlorocarbene , which can be generated ''in situ'' from chloroform and a strong base. Structures and bonding The two classes of carbenes are singlet and triplet carbenes. Singlet carbenes are spin-paired. In the language of valence bond theory, the molecule adopts an sp2 hybrid structure. Triplet carbenes have two unpaired electrons. Most carbenes have a nonlinear triplet ground state, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
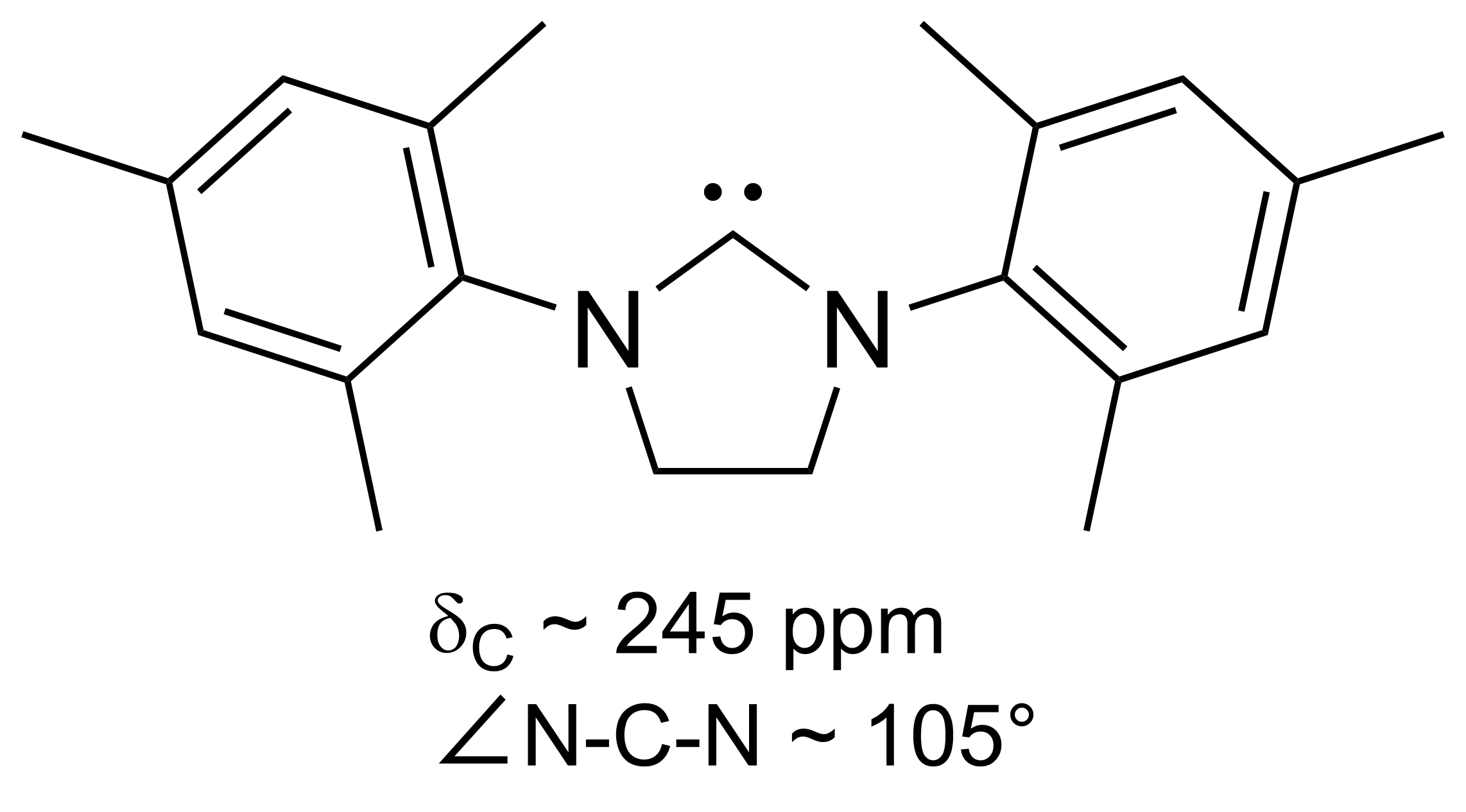
Persistent Carbene
A persistent carbene (also known as stable carbene) is a type of carbene demonstrating particular stability. The best-known examples and by far largest subgroup are the ''N''-heterocyclic carbenes (NHC) (sometimes called Arduengo carbenes), for example diaminocarbenes with the general formula (R2N)2C:, where the four R moieties are typically alkyl and aryl groups. The groups can be linked to give heterocyclic carbenes, such as those derived from imidazole, imidazoline, thiazole or triazole. Traditionally carbenes are viewed as so reactive that were only studied indirectly, such as by trapping reactions. This situation has changed dramatically with the emergence of persistent carbenes. Although they are fairly reactive substances, undergoing dimerization, many can be isolated as pure substances. Persistent carbenes tend to exist in the singlet. Their stability is only partly due to steric hindrance by bulky groups. Some singlet carbenes are thermodynamically stable and can be iso ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbene
In organic chemistry, a carbene is a molecule containing a neutral carbon atom with a valence of two and two unshared valence electrons. The general formula is or where the R represents substituents or hydrogen atoms. The term "carbene" may also refer to the specific compound , also called methylene, the parent hydride from which all other carbene compounds are formally derived. Carbenes are classified as either singlets or triplets, depending upon their electronic structure. Most carbenes are very short lived, although persistent carbenes are known. One well-studied carbene is dichlorocarbene , which can be generated ''in situ'' from chloroform and a strong base. Structures and bonding The two classes of carbenes are singlet and triplet carbenes. Singlet carbenes are spin-paired. In the language of valence bond theory, the molecule adopts an sp2 hybrid structure. Triplet carbenes have two unpaired electrons. Most carbenes have a nonlinear triplet ground state, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylene Radical
Methylene (systematically named methylidene and dihydridocarbon; also called carbene) is an organic compound with the chemical formula (also written ). It is a colourless gas that fluoresces in the mid-infrared range, and only persists in dilution, or as an adduct. Methylene is the simplest carbene.Roald Hoffman (2005), ''Molecular Orbitals of Transition Metal Complexes''. Oxford. It is usually detected only at very low temperatures, or as a short-lived intermediate in chemical reactions. W. B. DeMore and S. W. Benson (1964), Preparation, properties, and reactivity of methylene'. In ''Advances in Photochemistry'', John Wiley & Sons, 453 pages. Nomenclature The trivial name ''carbene'' is the preferred IUPAC name. The systematic names ''methylidene'' and ''dihydridocarbon'', valid IUPAC names, are constructed according to the substitutive and additive nomenclatures, respectively. ''Methylidene'' is viewed as methane with two hydrogen atoms removed. By default, this name ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
9-fluorenylidene
9-Fluorenylidene is an aryl carbene derived from the bridging methylene group of fluorene. Fluorenylidene has the unusual property that the triplet ground state is only 1.1 kcal/mol (4.6 kJ/mol) lower in energy than the singlet state. For this reason, fluorenylidene has been studied extensively in organic chemistry. Fluorenylidene is a reactive intermediate. Reactions involving fluorenylidene proceed through either the triplet or singlet state carbene, and the products formed depend on the relative concentration of spin states in solution, as influenced by experimental conditions. The rate of intersystem crossing is determined by the temperature and concentration of specific spin-trapping agents. Structure The ground state is believed to be a bent triplet, with two orthogonal sp hybrid orbitals singly occupied by unpaired spins. One electron occupies an orbital of sigma symmetry in the plane of the rings, while the other occupies an orbital of pi symmetry, which interacts ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dichlorocarbene
Dichlorocarbene is the reactive intermediate with chemical formula CCl2. Although this chemical species has not been isolated, it is a common intermediate in organic chemistry, being generated from chloroform. This bent diamagnetic molecule rapidly inserts into other bonds. Preparation Dichlorocarbene is most commonly generated by reaction of chloroform and a base such as potassium ''tert''-butoxide or aqueous sodium hydroxide. A phase transfer catalyst, for instance benzyltriethylammonium bromide, facilitates the migration of the hydroxide in the organic phase. :HCCl3 + NaOH → CCl2 + NaCl + H2O Other reagents and routes Another precursor to dichlorocarbene is ethyl trichloroacetate. Upon treatment with sodium methoxide it releases CCl2. Phenyl(trichloromethyl)mercury decomposes thermally to release CCl2. :PhHgCCl3 → CCl2 + PhHgCl Dichlorodiazirine, which is stable in the dark, decomposes into dichlorocarbene and nitrogen via photolysis. Dichlorocarbene can also be ob ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Orbital Hybridisation
In chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals to form new ''hybrid orbitals'' (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds in valence bond theory. For example, in a carbon atom which forms four single bonds the valence-shell s orbital combines with three valence-shell p orbitals to form four equivalent sp3 mixtures in a tetrahedral arrangement around the carbon to bond to four different atoms. Hybrid orbitals are useful in the explanation of molecular geometry and atomic bonding properties and are symmetrically disposed in space. Usually hybrid orbitals are formed by mixing atomic orbitals of comparable energies. History and uses Chemist Linus Pauling first developed the hybridisation theory in 1931 to explain the structure of simple molecules such as methane (CH4) using atomic orbitals. Pauling pointed out that a carbon atom forms fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diradical
In chemistry, a diradical is a molecular species with two electrons occupying molecular orbitals (MOs) which are degenerate. The term "diradical" is mainly used to describe organic compounds, where most diradicals are extremely reactive and in fact rarely isolated. Diradicals are even-electron molecules but have one fewer bond than the number permitted by the octet rule. Examples of diradical species can also be found in coordination chemistry, for example among bis(1,2-dithiolene) metal complexes. Spin states Diradicals are usually triplets. The phrases ''singlet'' and ''triplet'' are derived from the multiplicity of states of diradicals in electron spin resonance: a singlet diradical has one state (S = 0, Ms = 2*0+1 = 1, ms = 0) and exhibits no signal in EPR and a triplet diradical has 3 states (S = 1, Ms = 2*1+1 = 3, ms = -1; 0; 1) and shows in EPR 2 peaks (if no hyperfine splitting). The triplet state has total spin quantum number S = 1 and is paramagnetic. Therefore ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Valence Bond Theory
In chemistry, valence bond (VB) theory is one of the two basic theories, along with molecular orbital (MO) theory, that were developed to use the methods of quantum mechanics to explain chemical bonding. It focuses on how the atomic orbitals of the dissociated atoms combine to give individual chemical bonds when a molecule is formed. In contrast, molecular orbital theory has orbitals that cover the whole molecule. History Lothar Meyer in his 1864 book, ''Die modernen Theorien der Chemie'', contained an early version of the periodic table containing 28 elements, classified elements into six families by their valence—for the first time, elements had been grouped according to their valence. Works on organizing the elements by atomic weight, until then had been stymied by the widespread use of equivalent weights for the elements, rather than atomic weights. In 1916, G. N. Lewis proposed that a chemical bond forms by the interaction of two shared bonding electrons, with the repr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Paramagnetic Resonance
Electron paramagnetic resonance (EPR) or electron spin resonance (ESR) spectroscopy is a method for studying materials that have unpaired electrons. The basic concepts of EPR are analogous to those of nuclear magnetic resonance (NMR), but the spins excited are those of the electrons instead of the atomic nuclei. EPR spectroscopy is particularly useful for studying metal complexes and organic radicals. EPR was first observed in Kazan State University by Soviet physicist Yevgeny Zavoisky in 1944, and was developed independently at the same time by Brebis Bleaney at the University of Oxford. Theory Origin of an EPR signal Every electron has a magnetic moment and spin quantum number s = \tfrac , with magnetic components m_\mathrm = + \tfrac or m_\mathrm = - \tfrac . In the presence of an external magnetic field with strength B_\mathrm , the electron's magnetic moment aligns itself either antiparallel ( m_\mathrm = - \tfrac ) or parallel ( m_\mathrm = + \tfrac ) to the fi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Substituent
A substituent is one or a group of atoms that replaces (one or more) atoms, thereby becoming a moiety in the resultant (new) molecule. (In organic chemistry and biochemistry, the terms ''substituent'' and '' functional group'', as well as '' side chain'' and ''pendant group'', are used almost interchangeably to describe those branches from the parent structure, though certain distinctions are made in polymer chemistry. In polymers, side chains extend from the backbone structure. In proteins, side chains are attached to the alpha carbon atoms of the amino acid backbone.) The suffix ''-yl'' is used when naming organic compounds that contain a single bond replacing one hydrogen; ''-ylidene'' and ''-ylidyne'' are used with double bonds and triple bonds, respectively. In addition, when naming hydrocarbons that contain a substituent, positional numbers are used to indicate which carbon atom the substituent attaches to when such information is needed to distinguish between ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Pair
In chemistry, an electron pair or Lewis pair consists of two electrons that occupy the same molecular orbital but have opposite spins. Gilbert N. Lewis introduced the concepts of both the electron pair and the covalent bond in a landmark paper he published in 1916. Because electrons are fermions, the Pauli exclusion principle forbids these particles from having the same quantum numbers. Therefore, for two electrons to occupy the same orbital, and thereby have the same orbital quantum number, they must have different spin quantum number. This also limits the number of electrons in the same orbital to two. The pairing of spins is often energetically favorable, and electron pairs therefore play a large role in chemistry. They can form a chemical bond between two atoms, or they can occur as a lone pair of valence electrons. They also fill the core levels of an atom. Because the spins are paired, the magnetic moment of the electrons cancel one another, and the pair's contri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |