|
Bite Angle
In coordination chemistry the bite angle is the ligand–metal–ligand bond angle of coordination complex containing a bidentate ligand. This geometric parameter is used to classify chelation, chelating ligands, including those in organometallic complexes. It is most often discussed in terms of catalysis, as changes in bite angle can affect not just the activity and selectivity of a catalytic reaction but even allow alternative reaction pathways to become accessible. Although the parameter can be applied generally to any chelating ligand, it is commonly applied to describe diphosphine ligands, as they can adopt a wide range of bite angles. Diamines Diamines form a wide range of coordination complexes. They typically form 5- and 6-membered chelate rings. Examples of the former include ethylenediamine and 2,2'-bipyridine, 2,2′-bipyridine. Six-membered chelate rings are formed by 1,3-diaminopropane. The bite angle in such complexes is usually near 90°. Longer chain diamines, whi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PdCl2(dppe)-3D-balls
Palladium(II) chloride, also known as palladium dichloride and palladous chloride, are the chemical compounds with the formula PdCl2. PdCl2 is a common starting material in palladium chemistry – palladium-based catalysts are of particular value in organic synthesis. It is prepared by the reaction of chlorine with palladium metal at high temperatures. Structure Two forms of PdCl2 are known, denoted α and β. In both forms, the palladium centres adopt a square-planar coordination geometry that is characteristic of Pd(II). Furthermore, in both forms, the Pd(II) centers are linked by μ2-chloride bridges. The α-form of PdCl2 is a polymer, consisting of "infinite" slabs or chains. The β-form of PdCl2 is molecular, consisting of an octahedral cluster of six Pd atoms. Each of the twelve edges of this octahedron is spanned by Cl−. PtCl2 adopts similar structures, whereas NiCl2 adopts the CdCl2 motif, featuring hexacoordinated Ni(II). Two further polymorphs, γ-PdCl2 and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cone Angle
In coordination chemistry, the ligand cone angle (a common example being the Tolman cone angle or ''θ'') is a measure of the steric bulk of a ligand in a transition metal coordination complex. It is defined as the solid angle formed with the metal at the vertex and the outermost edge of the van der Waals spheres of the ligand atoms at the perimeter of the cone (see figure). Tertiary phosphine ligands are commonly classified using this parameter, but the method can be applied to any ligand. The term ''cone angle'' was first introduced by Chadwick A. Tolman, a research chemist at DuPont. Tolman originally developed the method for phosphine ligands in nickel complexes, determining them from measurements of accurate physical models. Asymmetric cases The concept of cone angle is most easily visualized with symmetrical ligands, e.g. PR3. But the approach has been refined to include less symmetrical ligands of the type PRR′R″ as well as diphosphines. In such asymmetric cases, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
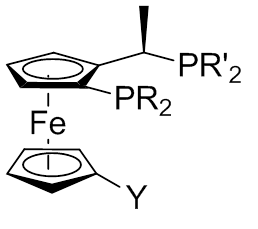
Josiphos Ligands
A Josiphos ligand is a type of chiral diphosphine which has been modified to be substrate (chemistry), substrate-specific; they are widely used for enantioselective synthesis.[3]H-U. Blaser, W. Brieden, B. Pugin, F. Spindler, M. Studer and A. Togni, Top. Catal ., 2002, 19, 3. They are named after the technician who made the first one, Josi Puleo. Applications Homogeneous catalysis is often used for Enantioselective synthesis, enantioselective transformations. The ligands carry chiral information and thus they are modified for individual substrates. Ligands can also influence the chemoselectivity of the catalyst. The Josiphos ligands, often called privileged ligands, are important because of their ability to give high yields in enantioselective synthesis. Josiphos ligands were developed in the 1990s by Antonio Togni in studies on Ferrocene, ferrocenyl ligands previously discovered by T. Hayashi (1986). These studies focused of an Au(I)-catalyzed aldol reaction at The Central Rese ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Constrained Geometry Complex
In organometallic chemistry, a "constrained geometry complex" (CGC) is a kind of catalyst used for the production of polyolefins such as polyethylene and polypropylene. The catalyst was one of the first major departures from metallocene-based catalysts and ushered in much innovation in the development of new plastics. Structure CGC complexes feature a pi-bonded moiety (e.g. cyclopentadienyl) linked to one of the other ligands on the same metal centre in such a way that the angle at this metal between the centroid of the pi-system and the additional ligand is smaller than in comparable unbridged complexes. More specifically, the term CGC was used for ansa-bridged cyclopentadienyl amido complexes, although the definition goes far beyond this class of compounds. The term CGC is frequently used in connection with other more or less related ligand systems that may or may not be isolobal and/or isoelectronic with the ansa-bridged cyclopentadienyl amido ligand system. Furthermore, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a Catalysis, catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or Saturated and unsaturated compounds, saturate organic compounds. Hydrogenation typically constitutes the addition of pairs of hydrogen atoms to a molecule, often an alkene. Catalysts are required for the reaction to be usable; non-catalytic hydrogenation takes place only at very high temperatures. Hydrogenation reduces Double bond, double and Triple bond, triple bonds in hydrocarbons. Process Hydrogenation has three components, the Saturated and unsaturated compounds, unsaturated substrate, the hydrogen (or hydrogen source) and, invariably, a catalyst. The redox, reduction reaction is carried out at different temperatures and pressures depending upon the substrate and the activity of the catalyst. Related or competing reactions The same ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrocyanation
In organic chemistry, hydrocyanation is a process for conversion of alkenes to nitriles. The reaction involves the addition of hydrogen cyanide and requires a catalyst. This conversion is conducted on an industrial scale for the production of precursors to nylon. Hydrocyanation of unactivated alkenes Industrially, hydrocyanation is commonly performed on alkenes catalyzed by nickel complexes of phosphite () ligands. A general reaction is shown:Piet W.N.M. van Leeuwen "Homogeneous Catalysis: Understanding the Art", 2004, Wiley-VCH, Weinheim. :RCH=CH2 + HCN -> RCH2-CH2-CN Stoichiometry and mechanism The reaction involves the addition of and cyanide () to the substrate. Usually the substrate is an alkene and the product is a nitrile. The reaction proceeds via the oxidative addition of HCN to a low-valent metal complex to give a hydrido cyanide complex. Subsequent binding of the alkene gives the intermediate , which then undergoes migratory insertion to give an alkylmetal cya ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Xantphos
Xantphos is an organophosphorus compound derived from the heterocycle xanthene. It is used as a bidentate diphosphine ligand and is noteworthy for having a particularly wide bite angle (108°). Such ligands are useful in the hydroformylation of alkenes. Illustrative of its wide bite angle, it forms both cis and trans adducts of platinum(II) chloride. In the latter context, xantphos is classified as a trans-spanning ligand. A related bidentate ligand with a greater bite angle is spanphos. The ligand is prepared by double directed lithiation of 9,9-dimethylxanthene with sec-butyllithium followed by treatment with chlorodiphenylphosphine.{{cite journal , author = Mirko Kranenburg, Yuri E. M. van der Burgt, Paul C. J. Kamer, Piet W. N. M. van Leeuwen, Kees Goubitz, and Jan Fraanje , title = New Diphosphine Ligands Based on Heterocyclic Aromatics Inducing Very High Regioselectivity in Rhodium-Catalyzed Hydroformylation: Effect of the Bite Angle , year = 1995 , journal = Organ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
VSEPR Theory
Valence shell electron pair repulsion (VSEPR) theory ( , ), is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. It is also named the Gillespie-Nyholm theory after its two main developers, Ronald Gillespie and Ronald Nyholm. The premise of VSEPR is that the valence electron pairs surrounding an atom tend to repel each other and will, therefore, adopt an arrangement that minimizes this repulsion. This in turn decreases the molecule's energy and increases its stability, which determines the molecular geometry. Gillespie has emphasized that the electron-electron repulsion due to the Pauli exclusion principle is more important in determining molecular geometry than the electrostatic repulsion. The insights of VSEPR theory are derived from topological analysis of the electron density of molecules. Such quantum chemical topology (QCT) methods include the electron localization function (ELF) and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
-3D-balls.png)
-chloride-xtal-3D-balls.png)