|
Benzidine Rearrangement
Benzidine (trivial name), also called 1,1'-biphenyl-4,4'-diamine (systematic name), is an organic compound with the formula (C6H4NH2)2. It is an aromatic amine. It is a component of a test for cyanide. Related derivatives are used in the production of dyes. Benzidine has been linked to bladder and pancreatic cancer. Synthesis and properties Benzidine is prepared in a two step process from nitrobenzene. First, the nitrobenzene is converted to 1,2-diphenylhydrazine, usually using iron powder as the reducing agent. Treatment of this hydrazine with mineral acids induces a rearrangement reaction to 4,4'-benzidine. Smaller amounts of other isomers are also formed. The benzidine rearrangement, which proceeds intramolecularly, is a classic mechanistic puzzle in organic chemistry. : The conversion is described as a ,5 igmatropic reaction. : In terms of its physical properties, 4,4'-benzidine is poorly soluble in cold water but can be recrystallized from hot water, where it cryst ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biphenyl
Biphenyl (also known as diphenyl, phenylbenzene, 1,1′-biphenyl, lemonene or BP) is an organic compound that forms colorless crystals. Particularly in older literature, compounds containing the functional group consisting of biphenyl less one hydrogen (the site at which it is attached) may use the prefixes xenyl or diphenylyl. It has a distinctively pleasant smell. Biphenyl is an aromatic hydrocarbon with a molecular formula (C6H5)2. It is notable as a starting material for the production of polychlorinated biphenyls (PCBs), which were once widely used as dielectric fluids and heat transfer agents. Biphenyl is also an intermediate for the production of a host of other organic compounds such as emulsifiers, optical brighteners, crop protection products, and plastics. Biphenyl is insoluble in water, but soluble in typical organic solvents. The biphenyl molecule consists of two connected phenyl rings. Properties and occurrence Biphenyl occurs naturally in coal tar, crude oi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzidine Rearrangement
Benzidine (trivial name), also called 1,1'-biphenyl-4,4'-diamine (systematic name), is an organic compound with the formula (C6H4NH2)2. It is an aromatic amine. It is a component of a test for cyanide. Related derivatives are used in the production of dyes. Benzidine has been linked to bladder and pancreatic cancer. Synthesis and properties Benzidine is prepared in a two step process from nitrobenzene. First, the nitrobenzene is converted to 1,2-diphenylhydrazine, usually using iron powder as the reducing agent. Treatment of this hydrazine with mineral acids induces a rearrangement reaction to 4,4'-benzidine. Smaller amounts of other isomers are also formed. The benzidine rearrangement, which proceeds intramolecularly, is a classic mechanistic puzzle in organic chemistry. : The conversion is described as a ,5 igmatropic reaction. : In terms of its physical properties, 4,4'-benzidine is poorly soluble in cold water but can be recrystallized from hot water, where it cryst ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tolidine
2-Tolidine (orthotolidine, o-tolidine; not to be confused with O-Toluidine, ''o''-toluidine) is an organic compound with the chemical formula (C6H4(CH3)NH2)2. Several isomers are known; the 3-tolidine derivative is also important commercially. It is a colorless compound although commercial samples are often colored. It is slightly soluble in water. It forms salt (chemistry), salts with acids, such as the hydrochloride, which is commercially available. 2-Tolidine can be produced by a benzidine rearrangement from a hydrazine derivative derived from 2-Nitrotoluene, 2-nitrotoluene. :(CH3C6H4)2N2H2 → (C6H3(CH3)NH2)2 Uses 2-Tolidine is a commercial aromatic amine used mainly for dye production.K. Hunger. W. Herbst "Pigments, Organic" in ''Ullmann's Encyclopedia of Industrial Chemistry'', Wiley-VCH, Weinheim, 2012. 2-Tolidine is an intermediate for the production of soluble azo dyes and insoluble pigments used particularly in the textile, leather and paper industries. : It has a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
3,3'-Dichlorobenzidine
3,3'-Dichlorobenzidine is an organic compound with the formula (C6H3Cl(NH2))2. The pure compound is pale yellow, but commercial samples are often colored. It is barely soluble in water and is often supplied as a wet paste. It is widely used in the production of diarylide yellow pigments used in the production of printing inks. Its use in the production of dyes has been largely discontinued because of concerns about carcinogenicity. Preparation and reactions 3,3'-Dichlorobenzidine is prepared in two steps from 2-nitrochlorobenzene. The first step involves reduction with zinc in base to afford 2,2'-dichlorodi phenylhydrazine. This intermediate undergoes the benzidine rearrangement to afford 3,3'-dichlorobenzidine. Aqueous solutions of 3,3'-dichlorobenzidine degrade in light to monochloro derivative. It undergoes chlorination (for example in water treatment plants) to give the tetrachloro derivative. The most widely practiced reaction of 3,3'-dichlorobenzidine is its double diazot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Luminol
Luminol (C8H7N3O2) is a chemical that exhibits chemiluminescence, with a blue glow, when mixed with an appropriate oxidizing agent. Luminol is a white-to-pale-yellow crystalline solid that is soluble in most polar organic solvents, but insoluble in water. Forensic investigators use luminol to detect trace amounts of blood at crime scenes, as it reacts with the iron in hemoglobin. Biologists use it in cellular assays to detect copper, iron, cyanides, as well as specific proteins via western blotting. When luminol is sprayed evenly across an area, trace amounts of an activating oxidant make the luminol emit a blue glow that can be seen in a darkened room. The glow only lasts about 30 seconds, but can be documented photographically. The glow is stronger in areas receiving more spray; the intensity of the glow does not indicate the amount of blood or other activator present. Synthesis Luminol is synthesized in a two-step process, beginning with 3-nitrophthalic acid. First, hyd ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%–6% by weight) in water for consumer use, and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or " high-test peroxide", decomposes explosively when heated and has been used as a propellant in rocketry. Hydrogen peroxide is a reactive oxygen species and the simplest peroxide, a compound having an oxygen–oxygen single bond. It decomposes slowly when exposed to light, and rapidly in the presence of organic or reactive compounds. It is typically stored with a stabilizer in a weakly acidic solution in a dark bottle to block light. Hydrogen peroxide is found in biological systems including the human body. Enzymes that use or decompose hydrogen peroxide are classified as peroxidases. Properties The boiling p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenolphthalein
Phenolphthalein ( ) is a chemical compound with the formula C20 H14 O4 and is often written as "HIn", "HPh", "phph" or simply "Ph" in shorthand notation. Phenolphthalein is often used as an indicator in acid–base titrations. For this application, it turns colorless in acidic solutions and pink in basic solutions. It belongs to the class of dyes known as phthalein dyes. Phenolphthalein is slightly soluble in water and usually is dissolved in alcohols for use in experiments. It is a weak acid, which can lose H+ ions in solution. The nonionized phenolphthalein molecule is colorless and the double deprotonated phenolphthalein ion is fuchsia. Further loss of proton in higher pH occurs slowly and leads to a colorless form. Phenolphthalein ion in concentrated sulfuric acid is orange red due to sulfonation. Uses pH indicator Phenolphthalein's common use is as an indicator in acid-base titrations. It also serves as a component of universal indicator, together with methyl red ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Blue
Blue is one of the three primary colours in the RYB colour model (traditional colour theory), as well as in the RGB (additive) colour model. It lies between violet and cyan on the spectrum of visible light. The eye perceives blue when observing light with a dominant wavelength between approximately 450 and 495 nanometres. Most blues contain a slight mixture of other colours; azure contains some green, while ultramarine contains some violet. The clear daytime sky and the deep sea appear blue because of an optical effect known as Rayleigh scattering. An optical effect called Tyndall effect explains blue eyes. Distant objects appear more blue because of another optical effect called aerial perspective. Blue has been an important colour in art and decoration since ancient times. The semi-precious stone lapis lazuli was used in ancient Egypt for jewellery and ornament and later, in the Renaissance, to make the pigment ultramarine, the most expensive of all pigments. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
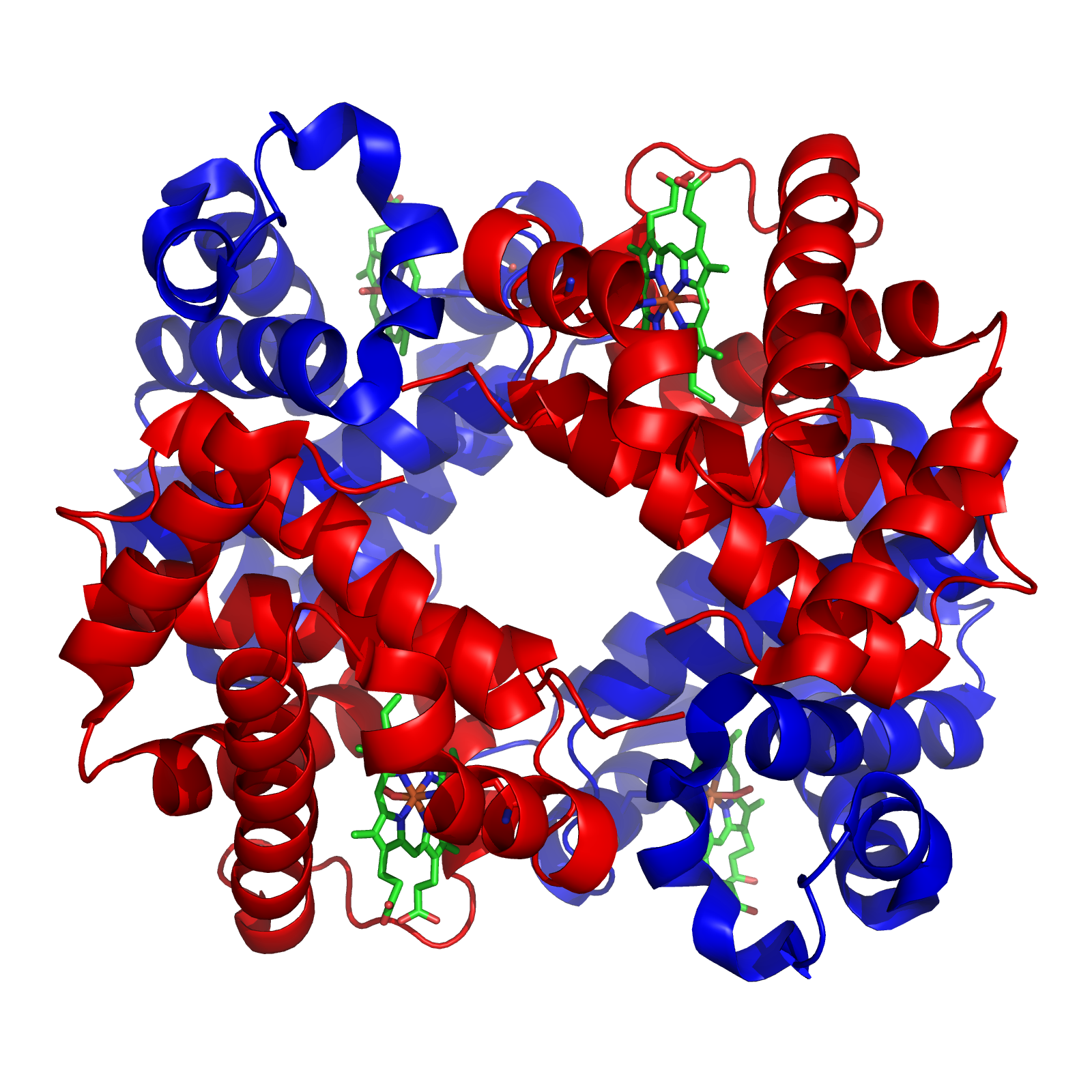
Enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the react ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Blood
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells. Blood in the circulatory system is also known as ''peripheral blood'', and the blood cells it carries, ''peripheral blood cells''. Blood is composed of blood cells suspended in blood plasma. Plasma, which constitutes 55% of blood fluid, is mostly water (92% by volume), and contains proteins, glucose, mineral ions, hormones, carbon dioxide (plasma being the main medium for excretory product transportation), and blood cells themselves. Albumin is the main protein in plasma, and it functions to regulate the colloidal osmotic pressure of blood. The blood cells are mainly red blood cells (also called RBCs or erythrocytes), white blood cells (also called WBCs or leukocytes) and platelets (also called thrombocytes). The most abundant cells in vertebrate blood a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Congo Red
Congo red is an organic compound, the sodium salt of 3,3′-( ,1′-biphenyl4,4′-diyl)bis(4-aminonaphthalene-1-sulfonic acid). It is an azo dye. Congo red is water-soluble, yielding a red colloidal solution; its solubility is greater in organic solvents. However, the use of Congo red has long been abandoned, primarily because of its carcinogenic properties.Klaus Hunger, Peter Mischke, Wolfgang Rieper, Roderich Raue, Klaus Kunde, Aloys Engel: "Azo Dyes" in ''Ullmann’s Encyclopedia of Industrial Chemistry'', 2005, Wiley-VCH, Weinheim.. History Congo red was first synthesized in 1883 by Paul Böttiger, who had been employed at Friedrich Bayer Company in Elberfeld, Germany. He was looking for textile dyes that did not require a mordant step. The company which had a right of first refusal to his inventions was not interested in this bright red color, so he filed the patent under his own name and sold it to the AGFA company of Berlin. AGFA marketed the dye under the name "Congo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |