|
Bromoform 1
Bromoform (CHBr3) is a brominated organic solvent, colorless liquid at room temperature, with a high refractive index, very high density, and sweet odor is similar to that of chloroform. It is one of the four haloforms, the others being fluoroform, chloroform, and iodoform. Bromoform can be prepared by the haloform reaction using acetone and sodium hypobromite, by the electrolysis of potassium bromide in ethanol, or by treating chloroform with aluminium bromide. Currently its main use is as a laboratory reagent. Structure The molecule adopts tetrahedral molecular geometry with C3v symmetry. Uses Only small quantities of bromoform are currently produced industrially in the United States. In the past, it was used as a solvent, sedative and flame retardant, but now it is mainly used as a laboratory reagent, for example as an extraction solvent. Bromoform also has medical uses; injections of bromoform are sometimes used instead of epinephrine to treat severe asthma cases. Bromoform' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
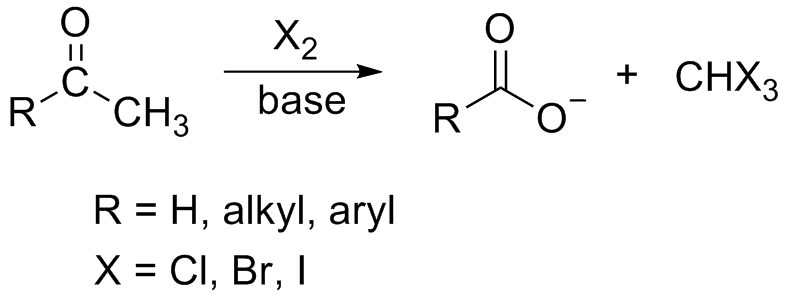
Haloform Reaction
In chemistry, the haloform reaction is a chemical reaction in which a haloform (, where X is a halogen) is produced by the exhaustive halogenation of an acetyl group (, where R can be either a hydrogen atom, an alkyl or an aryl group), in the presence of a base. The reaction can be used to transform acetyl groups into carboxyl groups () or to produce chloroform (), bromoform (), or iodoform (). Note that fluoroform () can't be prepared in this way. Mechanism In the first step, the halogen dis-proportionates in the presence of hydroxide to give the halide and hypohalite. :Br2 + 2 OH- -> Br- + BrO- + H2O If a secondary alcohol is present, it is oxidized to a ketone by the hypohalite: If a methyl ketone is present, it reacts with the hypohalite in a three-step process: 1. Under basic conditions, the ketone undergoes keto-enol tautomerisation. The enolate undergoes electrophilic attack by the hypohalite (containing a halogen with a formal +1 charge). : 2. When the α(al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Royal Society Of Chemistry
The Royal Society of Chemistry (RSC) is a learned society (professional association) in the United Kingdom with the goal of "advancing the chemistry, chemical sciences". It was formed in 1980 from the amalgamation of the Chemical Society, the Royal Institute of Chemistry, the Faraday Society, and the Society for Analytical Chemistry with a new Royal Charter and the dual role of learned society and professional body. At its inception, the Society had a combined membership of 34,000 in the UK and a further 8,000 abroad. The headquarters of the Society are at Burlington House, Piccadilly, London. It also has offices in Thomas Graham House in Cambridge (named after Thomas Graham (chemist), Thomas Graham, the first president of the Chemical Society) where ''RSC Publishing'' is based. The Society has offices in the United States, on the campuses of The University of Pennsylvania and Drexel University, at the University City Science Center in Philadelphia, Pennsylvania, in both Beijing a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrolysis
In chemistry and manufacturing, electrolysis is a technique that uses direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of elements from naturally occurring sources such as ores using an electrolytic cell. The voltage that is needed for electrolysis to occur is called the decomposition potential. The word "lysis" means to separate or break, so in terms, electrolysis would mean "breakdown via electricity". Etymology The word "electrolysis" was introduced by Michael Faraday in 1834, using the Greek words "amber", which since the 17th century was associated with electrical phenomena, and ' meaning "dissolution". Nevertheless, electrolysis, as a tool to study chemical reactions and obtain pure elements, precedes the coinage of the term and formal description by Faraday. History In the early nineteenth century, William Nicholson and Anthony Carlisle sought to further Volt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Asparagopsis
''Asparagopsis'' is a genus of edible red macroalgae (Rhodophyta). The species ''Asparagopsis taxiformis'' is found throughout the tropical and subtropical regions, while '' Asparagopsis armata'' is found in warm temperate regions. Both species are highly invasive, and have colonised the Mediterranean Sea. A third accepted species is ''A. svedelii'', while others are of uncertain status. Taxonomy and nomenclature The genus ''Asparagopsis'' belongs to the order Bonnemaisoniales, and family Bonnemaisoniaceae. , there are three confirmed species: *'' Asparagopsis armata'' Harvey, 1855 *''Asparagopsis taxiformis'' (Delile) Trevisan de Saint-Léon, 1845 *''Asparagopsis svedelli'' (W. R. Taylor) Other possible species are still unconfirmed: *''Asparagopsis delilei'' (Montagne) *''Asparagopsis hamifera'' (Hariot) *''Asparagopsis sanfordiana'' (Harvey) This genus, particularly ''Asparagopsis taxiformis'', is also a complex species line which is composed of six cryptic lineages wi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
FutureFeed
FutureFeed is a seaweed-based feed ingredient for livestock that is currently being developed by a team from Australia's Commonwealth Scientific and Industrial Research Organisation (CSIRO). The primary component of FutureFeed is dried ''Asparagopsis'', a genus of red algae, which has been shown to reduce the methane (CH4) emissions of ruminant livestock by up to 99%. It is added to fodder at feedlots in dosages of 1-2% dietary intake to achieve this result. FutureFeed is currently being developed in collaboration with James Cook University (JCU) and Meat and Livestock Australia (MLA), with the primary goal of scaling for mainstream commercial use. History Historical evidence suggests that farmers in Ancient Greece deliberately grazed cattle near beaches as a result of the productivity benefits it provided. This was also the case for Icelandic farmers in the 18th century. In the early 2010s, Canadian dairy farmer, Joe Dorgan, noticed that cattle in paddocks adjacent to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CSIRO
The Commonwealth Scientific and Industrial Research Organisation (CSIRO) is an Australian Government The Australian Government, also known as the Commonwealth Government, is the national government of Australia, a federal parliamentary constitutional monarchy. Like other Westminster-style systems of government, the Australian Government ... agency responsible for scientific research. CSIRO works with leading organisations around the world. From its headquarters in Canberra, CSIRO maintains more than 50 sites across Australia and in France, Chile and the United States, employing about 5,500 people. Federally funded scientific research began in Australia years ago. The Advisory Council of Science and Industry was established in 1916 but was hampered by insufficient available finance. In 1926 the research effort was reinvigorated by establishment of the Council for Scientific and Industrial Research (CSIR), which strengthened national science leadership and increased ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Seaweed
Seaweed, or macroalgae, refers to thousands of species of macroscopic, multicellular, marine algae. The term includes some types of '' Rhodophyta'' (red), ''Phaeophyta'' (brown) and ''Chlorophyta'' (green) macroalgae. Seaweed species such as kelps provide essential nursery habitat for fisheries and other marine species and thus protect food sources; other species, such as planktonic algae, play a vital role in capturing carbon, producing at least 50% of Earth's oxygen. Natural seaweed ecosystems are sometimes under threat from human activity. For example, mechanical dredging of kelp destroys the resource and dependent fisheries. Other forces also threaten some seaweed ecosystems; a wasting disease in predators of purple urchins has led to a urchin population surge which destroyed large kelp forest regions off the coast of California. Humans have a long history of cultivating seaweeds for their uses. In recent years, seaweed farming has become a global agricultural practic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methanogenesis
Methanogenesis or biomethanation is the formation of methane coupled to energy conservation by microbes known as methanogens. Organisms capable of producing methane for energy conservation have been identified only from the domain Archaea, a group phylogenetically distinct from both eukaryotes and bacteria, although many live in close association with anaerobic bacteria. Other forms of methane production that are not coupled to ATP synthesis exist within all three domains of life. The production of methane is an important and widespread form of microbial metabolism. In anoxic environments, it is the final step in the decomposition of biomass. Methanogenesis is responsible for significant amounts of natural gas accumulations, the remainder being thermogenic. Biochemistry Methanogenesis in microbes is a form of anaerobic respiration. Methanogens do not use oxygen to respire; in fact, oxygen inhibits the growth of methanogens. The terminal electron acceptor in methanogenesis is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flame Retardant
The term flame retardants subsumes a diverse group of chemicals that are added to manufactured materials, such as plastics and textiles, and surface finishes and coatings. Flame retardants are activated by the presence of an ignition source and are intended to prevent or slow the further development of ignition by a variety of different physical and chemical methods. They may be added as a copolymer during the polymerisation process, or later added to the polymer at a moulding or extrusion process or (particularly for textiles) applied as a topical finish. Mineral flame retardants are typically additive while organohalogen and organophosphorus compounds can be either reactive or additive. Classes Both Reactive and Additive Flame retardants types, can be further separated into four distinct classes: * Minerals such as aluminium hydroxide (ATH), magnesium hydroxide (MDH), huntite and hydromagnesite, various hydrates, red phosphorus, and boron compounds, mostly borates. * Organ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sedative
A sedative or tranquilliser is a substance that induces sedation by reducing irritability or excitement. They are CNS depressants and interact with brain activity causing its deceleration. Various kinds of sedatives can be distinguished, but the majority of them affect the neurotransmitter gamma-aminobutyric acid (GABA). In spite of the fact that each sedative acts in its own way, most produce relaxing effects by increasing GABA activity. This group is related to hypnotics. The term ''sedative'' describes drugs that serve to calm or relieve anxiety, whereas the term ''hypnotic'' describes drugs whose main purpose is to initiate, sustain, or lengthen sleep. Because these two functions frequently overlap, and because drugs in this class generally produce dose-dependent effects (ranging from anxiolysis to loss of consciousness) they are often referred to collectively as ''sedative-hypnotic'' drugs. Sedatives can be used to produce an overly-calming effect ( alcohol being the m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Symmetry Group
In group theory, the symmetry group of a geometric object is the group of all transformations under which the object is invariant, endowed with the group operation of composition. Such a transformation is an invertible mapping of the ambient space which takes the object to itself, and which preserves all the relevant structure of the object. A frequent notation for the symmetry group of an object ''X'' is ''G'' = Sym(''X''). For an object in a metric space, its symmetries form a subgroup of the isometry group of the ambient space. This article mainly considers symmetry groups in Euclidean geometry, but the concept may also be studied for more general types of geometric structure. Introduction We consider the "objects" possessing symmetry to be geometric figures, images, and patterns, such as a wallpaper pattern. For symmetry of physical objects, one may also take their physical composition as part of the pattern. (A pattern may be specified formally as a scalar field, a funct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrahedral Molecular Geometry
In a tetrahedral molecular geometry, a central atom is located at the center with four substituents that are located at the corners of a tetrahedron. The bond angles are cos−1(−) = 109.4712206...° ≈ 109.5° when all four substituents are the same, as in methane () as well as its heavier analogues. Methane and other perfectly symmetrical tetrahedral molecules belong to point group Td, but most tetrahedral molecules have lower symmetry. Tetrahedral molecules can be chiral. Tetrahedral bond angle The bond angle for a symmetric tetrahedral molecule such as CH4 may be calculated using the dot product of two vectors. As shown in the diagram, the molecule can be inscribed in a cube with the tetravalent atom (e.g. carbon) at the cube centre which is the origin of coordinates, O. The four monovalent atoms (e.g. hydrogens) are at four corners of the cube (A, B, C, D) chosen so that no two atoms are at adjacent corners linked by only one cube edge. If the edge length of the cube ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


_Figure_8.jpeg/1200px-Asparagopsis_armata_(10.3897-BDJ.8.e57510)_Figure_8.jpeg)



.jpg)