|
Azobisisobutylonitrile
Azobisisobutyronitrile (abbreviated AIBN) is an organic compound with the formula CH3)2C(CN)sub>2N2. This white powder is soluble in alcohols and common organic solvents but is insoluble in water. It is often used as a foamer in plastics and rubber and as a radical initiator. As an azo initiator, radicals resulting from AIBN have multiple benefits over common organic peroxides. For example, they do not have oxygenated byproducts or much yellow discoloration. Additionally, they do not cause too much grafting and therefore are often used when making adhesives, acrylic fibers, detergents, etc. Mechanism of decomposition In its most characteristic reaction, AIBN decomposes, eliminating a molecule of nitrogen gas to form two 2-cyanoprop-2-yl radicals: : Because azobisisobutyronitrile readily gives off free radicals, it is often used as a radical initiator. This happens at temperatures above 40 °C, but in experiments is more commonly done at temperatures between 66&nbs ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ABCN
1,1′-Azobis(cyclohexanecarbonitrile) or ACHN is a radical initiator. The molecular formula is NCC6H10N=NC6H10CN. It is a white solid that is soluble in aromatic solvents. ACHN has a 10-hour half-life in toluene at 88 °C. See also * Azobisisobutylonitrile (AIBN) is another commonly used free radical initiator References Azo compounds Nitriles Radical initiators {{Organic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tributyltin Hydride
Tributyltin hydride is an organotin compound with the formula (C4H9)3SnH. It is a colorless liquid that is soluble in organic solvents. The compound is used as a source of hydrogen atoms in organic synthesis. Synthesis and characterization The compound is produced by reduction of tributyltin oxide with polymethylhydrosiloxane: : 2 " eSi(H)Osub>n" + (Bu3Sn)2O → " eSi(OH)Osub>n" + 2 Bu3SnH The hydride is a distillable liquid that is mildly sensitive to air, decomposing to (Bu3Sn)2O. Its IR spectrum exhibits a strong band at 1814 cm−1 for ''ν''Sn−H. Applications It is a specialized reagent in organic synthesis. Combined with azobisisobutyronitrile (AIBN) or by irradiation with light, tributyltin hydride converts organic halides (and related groups) to the corresponding hydrocarbon. This process occurs via a radical chain mechanism involving the radical Bu3Sn•. [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoyl Peroxide
Benzoyl peroxide is a chemical compound (specifically, an organic peroxide) with structural formula , often abbreviated as (BzO)2. In terms of its structure, the molecule can be described as two benzoyl (, Bz) groups connected by a peroxide (). It is a white granular solid with a faint odour of benzaldehyde, poorly soluble in water but soluble in acetone, ethanol, and many other organic solvents. Benzoyl peroxide is an oxidizer, but it is principally used as in the production of polymers. Benzoyl peroxide is mainly used in production of plastics and for bleaching flour, hair, plastics and textiles. Synthesis, structure, and chemical reactivity The original 1858 synthesis by Liebig reacted benzoyl chloride with barium peroxide, a reaction described by the following idealized equation: : Benzoyl peroxide is usually prepared by treating hydrogen peroxide with benzoyl chloride under alkaline conditions. : As in many other peroxides, the C-O-O-C linkage has a dihedral angle ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Production Of AIBN
Production may refer to: Economics and business * Production (economics) * Production, the act of manufacturing goods * Production, in the outline of industrial organization, the act of making products (goods and services) * Production as a statistic, gross domestic product * Production line Arts, entertainment, and media Motion pictures * Production, film distributor of a company * Production, Filmmaking#Production, phase of filmmaking * Production, video production Other uses in arts, entertainment, and media * Production (album), ''Production'' (album), by Mirwais, 2000 * Production, magic (illusion)#Categories of effects, category of illusory magic trick * Production, Video game development#Production, phase of video games development * Production, Record producer's role * Production, Theatre, theatrical performance Science and technology * Production, Deployment environment#Production, deployment environment where changes go "live" and users interact with it * Production (c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
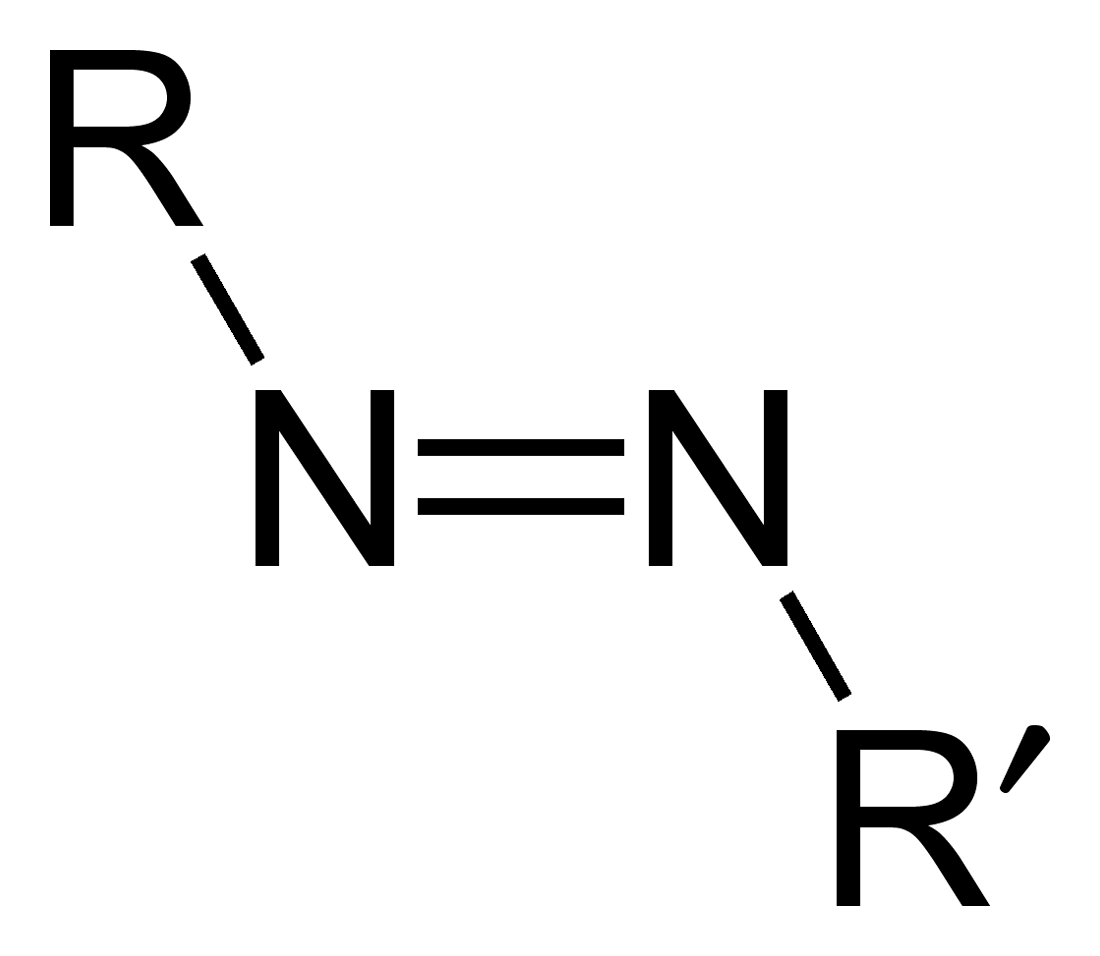
Azo Compound
Azo compounds are organic compounds bearing the functional group diazenyl (, in which R and R′ can be either aryl or alkyl groups). IUPAC defines azo compounds as: "Derivatives of diazene (diimide), , wherein both hydrogens are substituted by hydrocarbyl groups, e.g. azobenzene or diphenyldiazene." The more stable derivatives contain two aryl groups. The group is called an ''azo group'' (, ). Many textile and leather articles are dyed with azo dyes and pigments. Aryl azo compounds Aryl azo compounds are usually stable, crystalline species. Azobenzene is the prototypical aromatic azo compound. It exists mainly as the ''trans'' isomer, but upon illumination, converts to the ''cis'' isomer. Aromatic azo compounds can be synthesized by azo coupling, which entails an electrophilic substitution reaction where an aryl diazonium cation is attacked by another aryl ring, especially those substituted with electron-donating groups: :ArN2+ + Ar'H -> ArN=NAr' + H+ Since diazoniu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrazine
Hydrazine is an inorganic compound with the chemical formula . It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour. Hydrazine is highly toxic unless handled in solution as, for example, hydrazine hydrate (). Hydrazine is mainly used as a foaming agent in preparing polymer foams, but applications also include its uses as a precursor to polymerization catalysts, pharmaceuticals, and agrochemicals, as well as a long-term storable propellant for in-space spacecraft propulsion. Additionally, hydrazine is used in various rocket fuels and to prepare the gas precursors used in air bags. Hydrazine is used within both nuclear and conventional electrical power plant steam cycles as an oxygen scavenger to control concentrations of dissolved oxygen in an effort to reduce corrosion. the world hydrazine hydrate market amounted to $350 million. About two million tons of hydrazine hydrate were used in foam blowing agents in 2015. Hydrazines r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetone Cyanohydrin
Acetone cyanohydrin (ACH) is an organic compound used in the production of methyl methacrylate, the monomer of the transparent plastic polymethyl methacrylate (PMMA), also known as acrylic. It liberates hydrogen cyanide easily, so it is used as a source of such. For this reason, this cyanohydrin is also highly toxic. Preparation In the laboratory, this compound may be prepared by treating sodium cyanide with acetone, followed by acidification: : Considering the high toxicity of acetone cyanohydrin, a lab scale production has been developed using a microreactor-scale flow chemistry to avoid needing to manufacture and store large quantities of the reagent. Alternatively, a simplified procedure involves the action of sodium or potassium cyanide on the sodium bisulfite adduct of acetone prepared ''in situ''. This gives a less pure product, one that is nonetheless suitable for most syntheses. Reactions Acetone cyanohydrin is an intermediate en route to methyl methacrylate. Treat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyacrylic Acid
Poly(acrylic acid) (PAA; trade name Carbomer) is a polymer with the formula (CH2-CHCO2H)n. It is a derivative of acrylic acid (CH2=CHCO2H). In addition to the homopolymers, a variety of copolymers and crosslinked polymers, and partially deprotonated derivatives thereof are known and of commercial value. In a water solution at neutral pH, PAA is an anionic polymer, i.e., many of the side chains of PAA lose their protons and acquire a negative charge. Partially or wholly deprotonated PAAs are polyelectrolytes, with the ability to absorb and retain water and swell to many times their original volume. These properties – acid-base and water-attracting – are the bases of many applications. = Synthesis = PAA is produced by free radical polymerization. Initiators include potassium persulfate and AIBN. Polyacrylic acid is a polyolefin. It can be viewed as polyethylene with carboxylic acid (CO2H) substituents on alternating carbons. Owing to these groups, alternating carbon atoms ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrohalogenation Of Alkene Mechanism
A hydrohalogenation reaction is the electrophilic addition of hydrohalic acids like hydrogen chloride or hydrogen bromide to alkenes to yield the corresponding haloalkanes. : If the two carbon atoms at the double bond are linked to a different number of hydrogen atoms, the halogen is found preferentially at the carbon with fewer hydrogen substituents, an observation known as Markovnikov's rule. This is due to the abstraction of a hydrogen atom by the alkene from the acid (HX) to form the most stable carbocation (relative stability: 3°>2°>1°>methyl), as well as generating a halogen anion. A simple example of a hydrochlorination is that of indene with hydrogen chloride gas (no solvent): : Alkynes also undergo hydrohalogenation reactions. Depending on the exact substrate, alkyne hydrohalogenation can proceed though a concerted protonation/nucleophilic attack (AdE3) or stepwise by first protonating the alkyne to form a vinyl cation, followed by attack of HX/X− to give the produc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrohalogenation Of Alkene Reaction
A hydrohalogenation reaction is the electrophilic addition of hydrohalic acids like hydrogen chloride or hydrogen bromide to alkenes to yield the corresponding haloalkanes. : If the two carbon atoms at the double bond are linked to a different number of hydrogen atoms, the halogen is found preferentially at the carbon with fewer hydrogen substituents, an observation known as Markovnikov's rule. This is due to the abstraction of a hydrogen atom by the alkene from the acid (HX) to form the most stable carbocation (relative stability: 3°>2°>1°>methyl), as well as generating a halogen anion. A simple example of a hydrochlorination is that of indene with hydrogen chloride gas (no solvent): : Alkynes also undergo hydrohalogenation reactions. Depending on the exact substrate, alkyne hydrohalogenation can proceed though a concerted protonation/nucleophilic attack (AdE3) or stepwise by first protonating the alkyne to form a vinyl cation, followed by attack of HX/X− to give the produc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrohalogenation
A hydrohalogenation reaction is the electrophilic addition of hydrohalic acids like hydrogen chloride or hydrogen bromide to alkenes to yield the corresponding haloalkanes. : If the two carbon atoms at the double bond are linked to a different number of hydrogen atoms, the halogen is found preferentially at the carbon with fewer hydrogen substituents, an observation known as Markovnikov's rule. This is due to the abstraction of a hydrogen atom by the alkene from the acid (HX) to form the most stable carbocation (relative stability: 3°>2°>1°>methyl), as well as generating a halogen anion. A simple example of a hydrochlorination is that of indene with hydrogen chloride gas (no solvent): : Alkynes also undergo hydrohalogenation reactions. Depending on the exact substrate, alkyne hydrohalogenation can proceed though a concerted protonation/nucleophilic attack (AdE3) or stepwise by first protonating the alkyne to form a vinyl cation, followed by attack of HX/X− to give the produc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
_Reaction.png)
.png)