|
Asinger Reaction
The Asinger-reaction (sometimes referred to as the Asinger-4 component reaction or A-4CR for short) is a multicomponent reaction for the synthesis of 3-thiazolines and other related heterocycles. It is named after Friedrich Asinger who first reported it in 1956. Process An α-halogenated carbonyl-component reacts with sodium hydrosulfide (NaSH) and forms a Thiol ''in situ''. The thiol reacts directly with another carbonyl component and ammonia to form a thiazoline. The reaction works also by using elemental sulphur, an α–substituted ketone, another carbonyl component and ammonia; in this case, a mixture of products is formed. The formation of 3-thiazolines also occurs by using α- thioaldehyde or α-thioketone and ammonia. A simplified route of the Asinger-reaction was developed at Degussa. An α-halogenated carbonyl compound reacts with sodium hydrosulfide (NaSH) and forms a Thiol In organic chemistry, a thiol (; ), or thiol derivative, is any organosulfur compound of t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Multicomponent Reaction
In chemistry, a multi-component reaction (or MCR), sometimes referred to as a "Multi-component Assembly Process" (or MCAP), is a chemical reaction where three or more compounds react to form a single product. By definition, multicomponent reactions are those reactions whereby more than two reactants combine in a sequential manner to give highly selective products that retain majority of the atoms of the starting material. History and Types Multicomponent Reactions Multicomponent reactions have been known for over 150 years. The first documented multicomponent reaction was the Strecker synthesis of α-amino cyanides in 1850 from which α-amino acids could be derived. A multitude of MCRs exist today, of which the isocyanide based MCRs are the most documented. Other MCRs include free-radical mediated MCRs, MCRs based on organoboron compounds and metal-catalyzed MCRs. Isocyanide based MCRs are most frequently exploited because the isocyanide is an extraordinary functional group. It ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
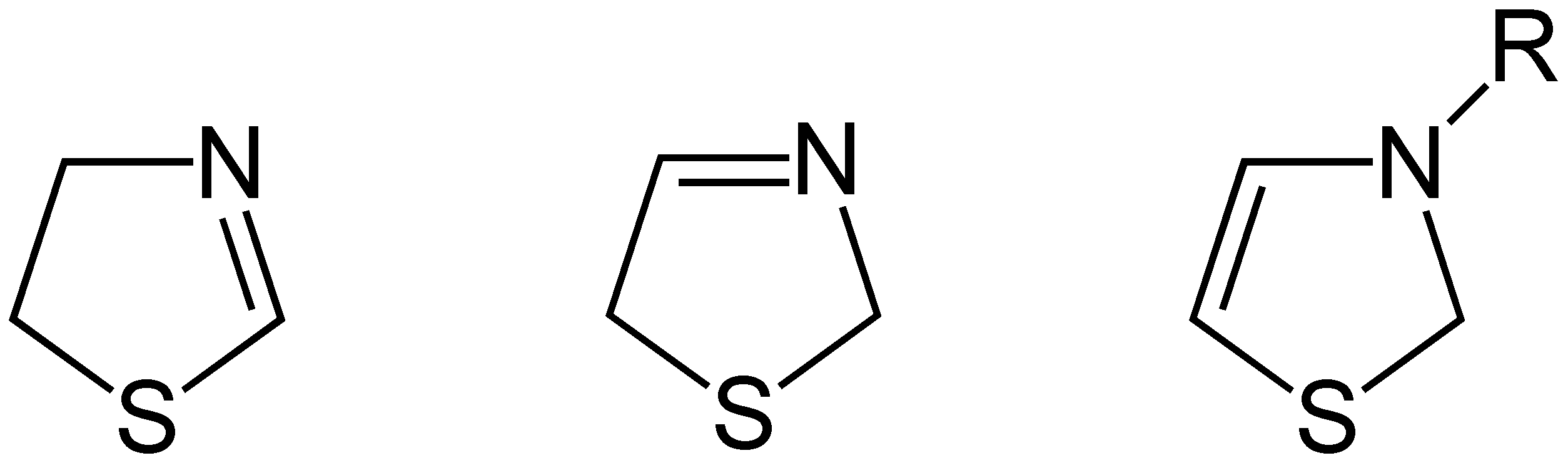
Thiazoline
Thiazolines (or dihydrothiazoles) are a group of isomeric 5-membered heterocyclic compounds containing both sulfur and nitrogen in the ring. Although unsubstituted thiazolines are rarely encountered themselves, their derivatives are more common and some are bioactive. For example, in a common post-translational modification, cysteine residues are converted into thiazolines. The name thiazoline originates from the Hantzsch–Widman nomenclature. Isomers Three structural isomers of thiazoline exist depending on the position of the double bond. These forms do not readily interconvert and hence are not tautomers. Of these 2-thiazoline is the most common. A fourth structure exists in which the N and S atoms are adjacent; this known as isothiazoline. Synthesis Thiazolines were first prepared by dialkylation of thioamides by Richard Willstatter in 1909. 2-Thiazolines are commonly prepared from 2-aminoethanethiols (e.g. cysteamine). They may also be synthesized via the A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Friedrich Asinger
Friedrich Asinger (26 June 1907 in Freiland/Niederdonau (Austria); – 7 March 1999 in Aachen) was an Austrian chemist and professor for Technical Chemistry. He is well known for his development of a multi-component reaction, the Asinger reaction for the synthesis of 3-thiazolines. Life and work Asinger grew up with an older brother and two sisters in Lower Austria, as son of the head of a paper and cardboard factory. His mother came from a family of innkeepers. He graduated in 1924 from the upper secondary school in Krems an der Donau at the age of 17. He studied chemistry at the Vienna University of Technology, where he became in 1932 an academic student of Friedrich Böck (1876–1958). He successfully defended his PhD thesis on "Über den Einfluß von Substituenten auf die Verseifungsgeschwindigkeit von Benzalchlorid (The Influence of substituents on the saponification rate of benzal chloride)" and graduated with distinction. Asinger spent several years as department head ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiol
In organic chemistry, a thiol (; ), or thiol derivative, is any organosulfur compound of the form , where R represents an alkyl or other organic substituent. The functional group itself is referred to as either a thiol group or a sulfhydryl group, or a sulfanyl group. Thiols are the sulfur analogue of alcohols (that is, sulfur takes the place of oxygen in the hydroxyl () group of an alcohol), and the word is a blend of "''thio-''" with "alcohol". Many thiols have strong odors resembling that of garlic or rotten eggs. Thiols are used as odorants to assist in the detection of natural gas (which in pure form is odorless), and the "smell of natural gas" is due to the smell of the thiol used as the odorant. Thiols are sometimes referred to as mercaptans () or mercapto compounds, a term introduced in 1832 by William Christopher Zeise and is derived from the Latin ('capturing mercury')''Oxford American Dictionaries'' ( Mac OS X Leopard). because the thiolate group () bonds very stro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous waste, particularly among aquatic organisms, and it contributes significantly to the nutritional needs of terrestrial organisms by serving as a precursor to 45% of the world's food and fertilizers. Around 70% of ammonia is used to make fertilisers in various forms and composition, such as urea and Diammonium phosphate. Ammonia in pure form is also applied directly into the soil. Ammonia, either directly or indirectly, is also a building block for the synthesis of many pharmaceutical products and is used in many commercial cleaning products. It is mainly collected by downward displacement of both air and water. Although common in nature—both terrestrially and in the outer planets of the Solar System—and in wide use, ammonia is bot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bond C=O). The simplest ketone is acetone (where R and R' is methyl), with the formula . Many ketones are of great importance in biology and in industry. Examples include many sugars (ketoses), many steroids (e.g., testosterone), and the solvent acetone. Nomenclature and etymology The word ''ketone'' is derived from ''Aketon'', an old German word for ''acetone''. According to the rules of IUPAC nomenclature, ketone names are derived by changing the suffix ''-ane'' of the parent alkane to ''-anone''. Typically, the position of the carbonyl group is denoted by a number, but traditional nonsystematic names are still generally used for the most important ketones, for example acetone and benzophenone. These nonsystematic names are considered re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thioaldehyde
In organic chemistry, a thial or thioaldehyde is a functional group which is similar to an aldehyde, , in which a sulfur (S) atom replaces the oxygen (O) atom of the aldehyde (R represents an alkyl or aryl group). Thioaldehydes are even more reactive than thioketones. Unhindered thioaldehydes are generally too reactive to be isolated — for example, thioformaldehyde, , condenses to the cyclic trimer 1,3,5-trithiane. Thioacrolein, , formed by decomposition of allicin from garlic, undergoes a self Diels-Alder reaction giving isomeric vinyldithiins. While thioformaldehyde is highly reactive, it is found in interstellar space along with its mono- and di-deuterated isotopologues. With sufficient steric bulk, however, stable thioaldehydes can be isolated. In early work, the existence of thioaldehydes was inferred by trapping processes. For instance the reaction of with benzaldehyde was proposed to form thiobenzaldehyde, which forms a cycloadduct with the dithiophosphine ylides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thioketone
In organic chemistry, thioketones (; also known as thiones or thiocarbonyls) are organosulfur compounds related to conventional ketones in which the oxygen has been replaced by a sulfur. Instead of a structure of , thioketones have the structure , which is reflected by the prefix "thio-" in the name of the functional group. Unhindered alkylthioketones typically tend to form polymers or rings. Structure and bonding The C=S bond length of thiobenzophenone is 1.63 Å, which is comparable to 1.64 Å, the C=S bond length of thioformaldehyde, measured in the gas phase. Due to steric interactions, the phenyl groups are not coplanar and the dihedral angle SC-CC is 36°. Unhindered dialkylthiones polymerize or oligomerize but thiocamphor is well characterized red solid. Consistent with the double bond rule, most alkyl thioketones are unstable with respect to dimerization.Organosulfur Chemistry I: Topics in Current Chemistry, 1999, Volume 204/1999, 127-181, The energy difference between ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aminoacid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha amino acids appear in the genetic code. Amino acids can be classified according to the locations of the core structural functional groups, as Alpha and beta carbon, alpha- , beta- , gamma- or delta- amino acids; other categories relate to Chemical polarity, polarity, ionization, and side chain group type (aliphatic, Open-chain compound, acyclic, aromatic, containing hydroxyl or sulfur, etc.). In the form of proteins, amino acid '' residues'' form the second-largest component (water being the largest) of human muscles and other tissues. Beyond their role as residues in proteins, amino acids participate in a number of processes such as neurotransmitter transport and biosynthesis. It is thought that they played a key role in enabling li ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cysteine
Cysteine (symbol Cys or C; ) is a semiessential proteinogenic amino acid with the formula . The thiol side chain in cysteine often participates in enzymatic reactions as a nucleophile. When present as a deprotonated catalytic residue, sometimes the symbol Cyz is used. The deprotonated form can generally be described by the symbol Cym as well. The thiol is susceptible to oxidation to give the disulfide derivative cystine, which serves an important structural role in many proteins. In this case, the symbol Cyx is sometimes used. When used as a food additive, it has the E number E920. Cysteine is encoded by the codons UGU and UGC. The sulfur-containing amino acids cysteine and methionine are more easily oxidized than the other amino acids. Structure Like other amino acids (not as a residue of a protein), cysteine exists as a zwitterion. Cysteine has chirality in the older / notation based on homology to - and -glyceraldehyde. In the newer ''R''/''S'' system of design ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

-3D-balls.png)


