|
Apigenin
Apigenin (4′,5,7-trihydroxyflavone), found in many plants, is a natural product belonging to the flavone class that is the aglycone of several naturally occurring glycosides. It is a yellow crystalline solid that has been used to dye wool. Sources in nature Apigenin is found in many fruits and vegetables, but parsley, celery, celeriac, and chamomile tea are the most common sources. Apigenin is particularly abundant in the flowers of chamomile plants, constituting 68% of total flavonoids. Dried parsley can contain about 45 mg apigenin/gram of the herb, and dried chamomile flower about 3-5 mg/gram. The apigenin content of fresh parsley is reportedly 215.5 mg/100 grams, which is much higher than the next highest food source, green celery hearts providing 19.1 mg/100 grams. Biosynthesis Apigenin is biosynthetically derived from the general phenylpropanoid pathway and the flavone synthesis pathway. The phenylpropanoid pathway starts from the aromatic a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Apigenin Biosynthesis
Apigenin (4′,5,7-trihydroxyflavone), found in many plants, is a natural product belonging to the flavone class that is the aglycone of several naturally occurring glycosides. It is a yellow crystalline solid that has been used to dye wool. Sources in nature Apigenin is found in many fruits and vegetables, but parsley, celery, celeriac, and chamomile tea are the most common sources. Apigenin is particularly abundant in the flowers of chamomile plants, constituting 68% of total flavonoids. Dried parsley can contain about 45 mg apigenin/gram of the herb, and dried chamomile flower about 3-5 mg/gram. The apigenin content of fresh parsley is reportedly 215.5 mg/100 grams, which is much higher than the next highest food source, green celery hearts providing 19.1 mg/100 grams. Biosynthesis Apigenin is biosynthetically derived from the general phenylpropanoid pathway and the flavone synthesis pathway. The phenylpropanoid pathway starts from the aromatic amin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Apiin
Apiin is a natural flavonoid, a diglycoside of the flavone apigenin found in the winter-hardy plants parsley and celery, and in banana leaf. The glycoside moiety at carbon-7 of apigenin, ''O''-β-D-apiofuranosyl(→)2-β-D-glucosyl, is carried by several other flavones in parsley plant and seed. The sugar apiose possibly play a role in winter hardiness of celery, duckweed and parsley.page 136 "Advances in Carbohydrate Chemistry and Biochemistry", Volume 31, See also *Apiose Apiose is a branched-chain sugar found as residues in galacturonans-type pectins; that occurs in parsley and many other plants. Apiose is a component of cell wall polysaccharides. Apiose 1-reductase uses D-apiitol and NAD+ to produce api ... References Flavone glycosides {{Aromatic-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Apigetrin
Apigetrin is a chemical compound that can be found in dandelion coffee and in ''Teucrium gnaphalodes ''Teucrium gnaphalodes'' is a plant species in the genus ''Teucrium''. It is endemic to the Iberian Peninsula and grows at altitudes between 200 and 1500 m. It flowers from March to July. The flavones diosmin, cirsimaritin, salvigenin, cirsili ...''. References External links Apigetrin on chemlink.com Flavone glucosides {{organic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chamomile
Chamomile (American English) or camomile (British English; see spelling differences) ( or ) is the common name for several plants of the family Asteraceae. Two of the species, ''Matricaria recutita'' and ''Anthemis nobilis'', are commonly used to make herbal infusions for beverages. There is insufficient scientific evidence that consuming chamomile in foods or beverages has any beneficial effects on health. Etymology The word ''chamomile'' is derived via the French and Latin, from the Greek grc, χαμαίμηλον, khamaimēlon, earth apple, label=none, from grc, χαμαί, khamai, on the ground, label=none, and grc, μῆλον, mēlon, apple, label=none. First used in the 13th century, the spelling ''chamomile'' corresponds to the Latin and the Greek . The spelling ''camomile'' is a British derivation from the French. Species Some commonly used species include: * ''Matricaria chamomilla'' – often called "German chamomile" or "Water of Youth" * ''Chamaemelum ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rhoifolin
Rhoifolin is a chemical compound. It is first isolated from plant ''Rhus succedanea''. The term "Rhoi" derived from generic name of plant Rhus. It is a flavone, a type of flavonoid isolated from '' Boehmeria nivea'', China grass or ramie (leaf), from ''Citrus limon'', Canton lemon (leaf), from ''Citrus x aurantium'', the bigarade or bitter orange (plant), from ''Citrus x paradisi'', the grapefruit (leaf), from '' Ononis campestris'', the cammock (shoot) and from ''Sabal serratula'', the serenoa ''Serenoa repens'', commonly known as saw palmetto, is the sole species currently classified in the genus ''Serenoa''. It is a small palm, growing to a maximum height around . It is endemic to the subtropical and tropical Southeastern United S ... or sabal fruit (plant). References Flavone glycosides {{Aromatic-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vitexin
Vitexin is an apigenin flavone glucoside, a chemical compound found in the passion flower, ''Vitex agnus-castus'' (chaste tree or chasteberry), in the ''Phyllostachys nigra'' bamboo leaves, in the pearl millet (Pennisetum millet), and in Hawthorn. Metabolism Goitrogenicity of millet flavones : Vitexin inhibits thyroid peroxidase thus contributing to goiter. * Vitexin beta-glucosyltransferase * Vitexin 2"-O-rhamnoside 7-O-methyltransferase See also * Isovitexin (or homovitexin, saponaretin) is the apigenin-6-''C''-glucoside. * Orientin Orientin is a flavone, a chemical flavonoid-like compound. It is the 8-C glucoside of luteolin. Natural occurrences Orientin is found in ''Adonis vernalis'', in '' Anadenanthera colubrina'' and ''Anadenanthera peregrina'', and in the '' Phyllost ..., the 3'-OH derivative References External links Vitexin on RDchemicals.com {{flavone Flavone glucosides C-glycoside natural phenols ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dandelion Coffee
Dandelion 'coffee' (also dandelion tea) is a tisane made from the root of the dandelion plant. The roasted dandelion root pieces and the beverage have some resemblance to coffee in appearance and taste, and it is thus commonly considered a coffee substitute. Dandelion root is used for both medicinal and culinary purposes and is thought to be a detoxifying herb. History The usage of the dandelion plant dates back to the ancient Egyptians, Greeks and Romans. Additionally, for over a thousand years, Chinese traditional medicine has been known to incorporate the plant. Susanna Moodie explained how to prepare dandelion 'coffee' in her memoir of living in Canada, ''Roughing it in the Bush'' (1852), where she mentions that she had heard of it from an article published in the 1830s in ''New York Albion'' by a certain Dr. Harrison. Dandelion 'coffee' was later mentioned in a '' Harpers New Monthly Magazine'' story in 1886. In 1919, dandelion root was noted as a source of cheap 'coffee' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flavonoid
Flavonoids (or bioflavonoids; from the Latin word ''flavus'', meaning yellow, their color in nature) are a class of polyphenolic secondary metabolites found in plants, and thus commonly consumed in the diets of humans. Chemically, flavonoids have the general structure of a 15-carbon skeleton, which consists of two phenyl rings (A and B) and a heterocyclic ring (C, the ring containing the embedded oxygen). This carbon structure can be abbreviated C6-C3-C6. According to the IUPAC nomenclature, they can be classified into: *flavonoids or bioflavonoids *isoflavonoids, derived from 3-phenyl chromen-4-one (3-phenyl-1,4-benzopyrone) structure *neoflavonoids, derived from 4-phenylcoumarine (4-phenyl-1,2-benzopyrone) structure The three flavonoid classes above are all ketone-containing compounds and as such, anthoxanthins ( flavones and flavonols). This class was the first to be termed bioflavonoids. The terms flavonoid and bioflavonoid have also been more loosely used to describe non ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Malonyl-CoA
Malonyl-CoA is a coenzyme A derivative of malonic acid. Functions It plays a key role in chain elongation in fatty acid biosynthesis and polyketide biosynthesis. Fatty acid biosynthesis Malonyl-CoA provides 2-carbon units to fatty acids and commits them to fatty acid chain synthesis. Malonyl-CoA is formed by carboxylating acetyl-CoA using the enzyme acetyl-CoA carboxylase. One molecule of acetyl-CoA joins with a molecule of bicarbonate,Nelson D, Cox M (2008) ''Lehninger principles of biochemistry''. 5th Ed: p. 806 requiring energy rendered from ATP. Malonyl-CoA is utilised in fatty acid biosynthesis by the enzyme malonyl coenzyme A:acyl carrier protein transacylase (MCAT). MCAT serves to transfer malonate from malonyl-CoA to the terminal thiol of ''holo''-acyl carrier protein (ACP). Polyketide biosynthesis MCAT is also involved in bacterial polyketide biosynthesis. The enzyme MCAT together with an acyl carrier protein (ACP), and a polyketide synthase (PKS) and chain-length f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyketide Synthase
Polyketides are a class of natural products derived from a precursor molecule consisting of a chain of alternating ketone (or reduced forms of a ketone) and methylene groups: (-CO-CH2-). First studied in the early 20th century, discovery, biosynthesis, and application of polyketides has evolved. It is a large and diverse group of secondary metabolites caused by its complex biosynthesis which resembles that of fatty acid synthesis. Because of this diversity, polyketides can have various medicinal, agricultural, and industrial applications. Many polyketides are medicinal or exhibit acute toxicity. Biotechnology has enabled discovery of more naturally-occurring polyketides and evolution of new polyketides with novel or improved bioactivity. History Naturally produced polyketides by various plants and organisms have been used by humans since before studies on them began in the 19th and 20th century. In 1893, J. Norman Collie synthesized detectable amounts of orcinol by heating dehy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
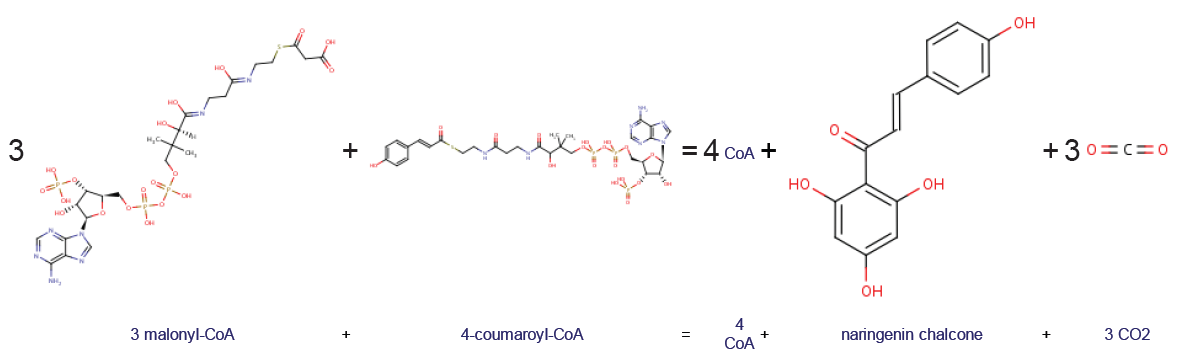
Chalcone Synthase
Chalcone synthase or naringenin-chalcone synthase (CHS) is an enzyme ubiquitous to higher plants and belongs to a family of polyketide synthase enzymes (PKS) known as type III PKS. Type III PKSs are associated with the production of chalcones, a class of organic compounds found mainly in plants as natural defense mechanisms and as synthetic intermediates. CHS was the first type III PKS to be discovered. It is the first committed enzyme in flavonoid biosynthesis. The enzyme catalyzes the conversion of 4-coumaroyl-CoA and malonyl-CoA to naringenin chalcone. Function CHS catalysis serves as the initial step for flavonoid biosynthesis. Flavonoids are important plant secondary metabolites that serve various functions in higher plants. These include pigmentation, UV protection, fertility, antifungal defense and the recruitment of nitrogen-fixing bacteria. CHS is believed to act as a central hub for the enzymes involved in the flavonoid pathway. Studies have shown that these enzyme ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Merck Index
''The Merck Index'' is an encyclopedia of chemical substance, chemicals, pharmaceutical drug, drugs and biomolecule, biologicals with over 10,000 monographs, monograph on single substances or groups of related chemical compound, compounds published online by the Royal Society of Chemistry. History The first edition of the Merck's Index was published in 1889 by the German chemical company Merck Group, Emanuel Merck and was primarily used as a sales catalog for Merck's growing list of chemicals it sold. The American subsidiary was established two years later and continued to publish it. During World War I the US government seized Merck's US operations and made it a separate American "Merck" company that continued to publish the Merck Index. In 2012 the Merck Index was licensed to the Royal Society of Chemistry. An online version of The Merck Index, including historic records and new updates not in the print edition, is commonly available through research libraries. It also include ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |