|
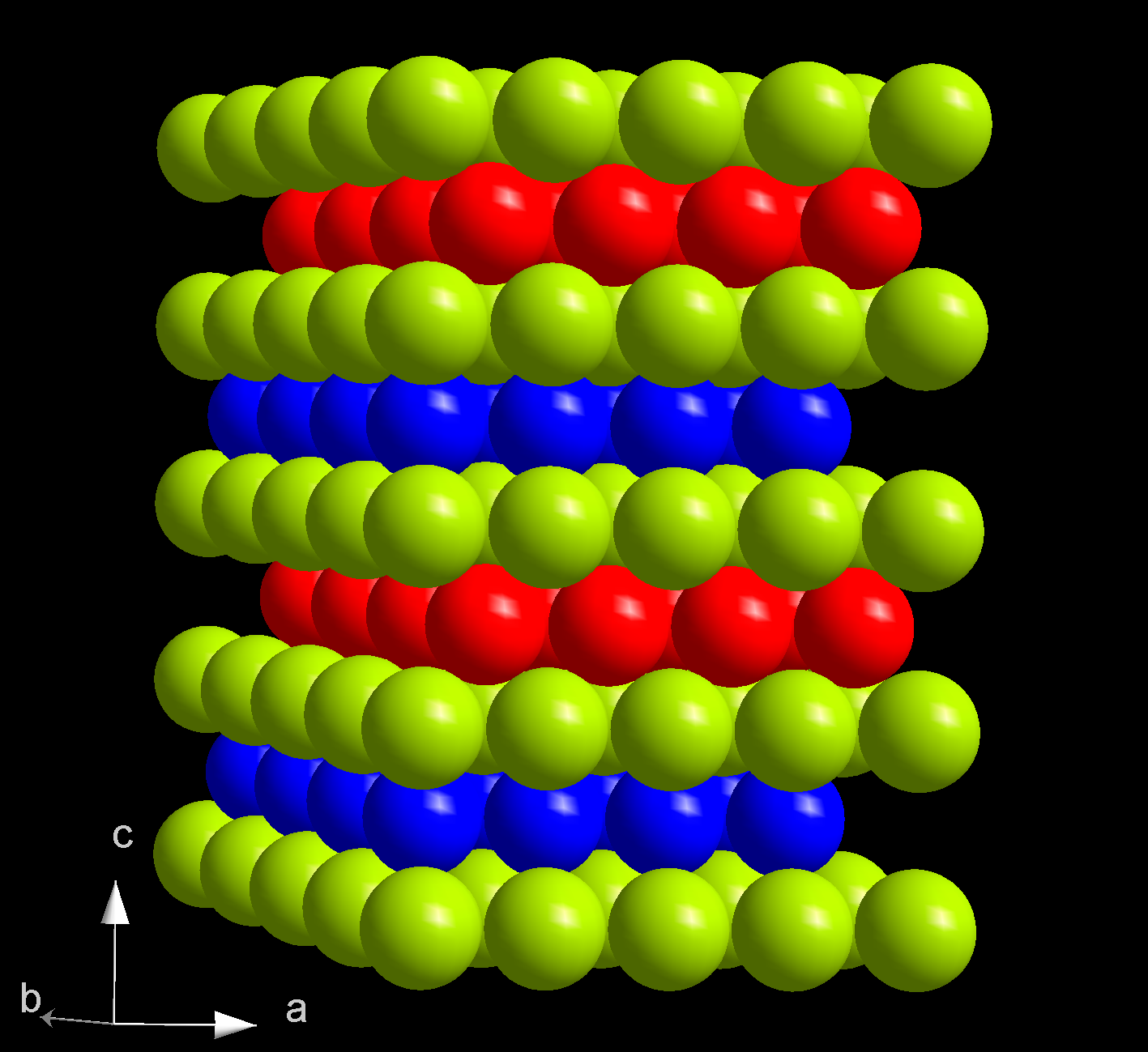
Americium(III) Oxide
Americium(III) oxide or americium sesquioxide is an oxide of the element americium. It has the empirical formula Am2O3. Since all isotopes of americium are only artificially produced, americium (III) oxide has no natural occurrence. The colour depends on the crystal structure, of which there are more than one. It is soluble in acids. Formation Americium(III) oxide can be made by heating americium dioxide in hydrogen at 600°C. :2 AmO2 + H2 -> Am2O3 + H2O Forms The hexagonal form is coloured tan, and the cubic form is coloured red-brown the same as persimmon The persimmon is the edible fruit of a number of species of trees in the genus ''Diospyros''. The most widely cultivated of these is the Oriental persimmon, ''Diospyros kaki'' ''Diospyros'' is in the family Ebenaceae, and a number of non-pers .... The cubic form converts to the hexagonal form on heating to 800°C. The cubic form is non-stoichimetric with variable oxygen composition. It darkens with increasing oxygen. Re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pearson Symbol
The Pearson symbol, or Pearson notation, is used in crystallography as a means of describing a crystal structure, and was originated by W. B. Pearson. The symbol is made up of two letters followed by a number. For example: * Diamond structure, ''cF''8 * Rutile structure, ''tP''6 The two (italicised) letters specify the Bravais lattice. The lower-case letter specifies the crystal family, and the upper-case letter the centering type. The number at the end of the Pearson symbol gives the number of the atoms in the conventional unit cell.Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005 IR-3.4.4, pp. 49–51; IR-11.5, pp. 241–242. |
Americium Dioxide
Americium is a synthetic radioactive chemical element with the symbol Am and atomic number 95. It is a transuranic member of the actinide series, in the periodic table located under the lanthanide element europium, and thus by analogy was named after the Americas. Americium was first produced in 1944 by the group of Glenn T. Seaborg from Berkeley, California, at the Metallurgical Laboratory of the University of Chicago, as part of the Manhattan Project. Although it is the third element in the transuranic series, it was discovered fourth, after the heavier curium. The discovery was kept secret and only released to the public in November 1945. Most americium is produced by uranium or plutonium being bombarded with neutrons in nuclear reactors – one tonne of spent nuclear fuel contains about 100 grams of americium. It is widely used in commercial ionization chamber smoke detectors, as well as in neutron sources and industrial gauges. Several unusual applications, such as nu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Americium(II) Oxide
Americium is a synthetic radioactive chemical element with the symbol Am and atomic number 95. It is a transuranic member of the actinide series, in the periodic table located under the lanthanide element europium, and thus by analogy was named after the Americas. Americium was first produced in 1944 by the group of Glenn T. Seaborg from Berkeley, California, at the Metallurgical Laboratory of the University of Chicago, as part of the Manhattan Project. Although it is the third element in the transuranic series, it was discovered fourth, after the heavier curium. The discovery was kept secret and only released to the public in November 1945. Most americium is produced by uranium or plutonium being bombarded with neutrons in nuclear reactors – one tonne of spent nuclear fuel contains about 100 grams of americium. It is widely used in commercial ionization chamber smoke detectors, as well as in neutron sources and industrial gauges. Several unusual applications, such as nuclear ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Curium(III) Oxide
Curium(III) oxide is a Chemical compound, compound composed of curium and oxygen with the chemical formula . It is a crystalline solid with a unit cell that contains two curium atoms and three oxygen atoms. The simplest synthesis equation involves the reaction of curium(III) metal with O2−: 2 Cm3+ + 3 O2− ---> Cm2O3. Curium trioxide can exist as five Polymorphism (materials science), polymorphic forms.Milman, V., Winkler, B., and C.J. Pickard (2003). “Crystal Structures of Curium Compounds: An Ab Initio Study''.” Journal of Nuclear Materials'' (322): 165-179. Two of the forms exist at extremely high temperatures, making it difficult for experimental studies to be done on the formation of their structures. The three other possible forms which curium sesquioxide can take are the body-centered cubic form, the Monoclinic crystal system, monoclinic form, and the Hexagonal lattice, hexagonal form.Petit, L., Svane, A., Szotek, Z., Temmerman, W.M., and G. M. Stocks (2009). “Electr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxide
An oxide () is a chemical compound that contains at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion of oxygen, an O2– (molecular) ion. with oxygen in the oxidation state of −2. Most of the Earth's crust consists of oxides. Even materials considered pure elements often develop an oxide coating. For example, aluminium foil develops a thin skin of Al2O3 (called a passivation layer) that protects the foil from further corrosion.Greenwood, N. N.; & Earnshaw, A. (1997). Chemistry of the Elements (2nd Edn.), Oxford:Butterworth-Heinemann. . Stoichiometry (the measurable relationship between reactants and chemical equations of a equation or reaction) Oxides are extraordinarily diverse in terms of stoichiometries and in terms of the structures of each stoichiometry. Most elements form oxides of more than one stoichiometry. A well known example is carbon monoxide and carbon dioxide.Greenwood, N. N.; & Earnshaw, A. (1997). Chemistry ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Americium
Americium is a synthetic radioactive chemical element with the symbol Am and atomic number 95. It is a transuranic member of the actinide series, in the periodic table located under the lanthanide element europium, and thus by analogy was named after the Americas. Americium was first produced in 1944 by the group of Glenn T. Seaborg from Berkeley, California, at the Metallurgical Laboratory of the University of Chicago, as part of the Manhattan Project. Although it is the third element in the transuranic series, it was discovered fourth, after the heavier curium. The discovery was kept secret and only released to the public in November 1945. Most americium is produced by uranium or plutonium being bombarded with neutrons in nuclear reactors – one tonne of spent nuclear fuel contains about 100 grams of americium. It is widely used in commercial ionization chamber smoke detectors, as well as in neutron sources and industrial gauges. Several unusual applications, such as nu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Americium
Americium (95Am) is an artificial element, and thus a standard atomic weight cannot be given. Like all artificial elements, it has no known stable isotopes. The first isotope to be synthesized was 241Am in 1944. The artificial element decays by ejecting alpha particles. Americium has an atomic number of 95 (the number of protons in the nucleus of the americium atom). Despite being an order of magnitude longer lived than , the former is harder to obtain than the latter as more of it is present in spent nuclear fuel. Nineteen radioisotopes of americium—223Am, 229Am, 230Am, and those ranging from 232Am to 247Am—have been characterized, with the most stable being 243Am with a half-life of 7,370 years, and 241Am with a half-life of 432.2 years. All of the remaining radioactive isotopes have half-lives that are less than 51 hours, and the majority of these have half-lives that are less than 100 minutes. This element also has 8 meta states, with the most stable being 242m1Am (t1 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Americium Dioxide
Americium is a synthetic radioactive chemical element with the symbol Am and atomic number 95. It is a transuranic member of the actinide series, in the periodic table located under the lanthanide element europium, and thus by analogy was named after the Americas. Americium was first produced in 1944 by the group of Glenn T. Seaborg from Berkeley, California, at the Metallurgical Laboratory of the University of Chicago, as part of the Manhattan Project. Although it is the third element in the transuranic series, it was discovered fourth, after the heavier curium. The discovery was kept secret and only released to the public in November 1945. Most americium is produced by uranium or plutonium being bombarded with neutrons in nuclear reactors – one tonne of spent nuclear fuel contains about 100 grams of americium. It is widely used in commercial ionization chamber smoke detectors, as well as in neutron sources and industrial gauges. Several unusual applications, such as nu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Persimmon
The persimmon is the edible fruit of a number of species of trees in the genus ''Diospyros''. The most widely cultivated of these is the Oriental persimmon, ''Diospyros kaki'' ''Diospyros'' is in the family Ebenaceae, and a number of non-persimmon species of the genus are grown for ebony timber. In 2019, China produced 75% of the world total of persimmons. Description Like the tomato, persimmons are not commonly considered to be berries, but Morphology (biology), morphologically the fruit is in fact a berry (botany), berry. The tree ''Diospyros kaki'' is the most widely cultivated species of persimmon. Typically the tree reaches in height and is round-topped. It usually stands erect, but sometimes can be crooked or have a willowy appearance. The leaves are long, and are Glossary of leaf morphology#oblong, oblong in shape with brown-hairy Petiole (botany), petioles in length. They are leathery and glossy on the upper surface, brown and silky underneath. The leaves are dec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Americium Compounds
Americium compounds are compounds containing the element americium (Am). These compounds can form in the +2, +3, and +4, although the +3 oxidation state is the most common. The +5, +6 and +7 oxidation states have also been reported. Oxides Three americium oxides are known, with the oxidation states +2 (AmO), +3 (), and +4 (). Americium(II) oxide was prepared in minute amounts and has not been characterized in detail. Americium(III) oxide is a red-brown solid with a melting point of 2205 °C.Wiberg, p. 1972 Americium(IV) oxide is the main form of solid americium which is used in nearly all its applications. Like most other actinide dioxides, it is a black solid with a cubic (fluorite) crystal structure.Greenwood, p. 1267 The oxalate of americium(III), vacuum dried at room temperature, has the chemical formula . Upon heating in vacuum, it loses water at 240 °C and starts decomposing into at 300 °C, the decomposition completes at about 470 °C.Penneman, p. 4 T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |