|
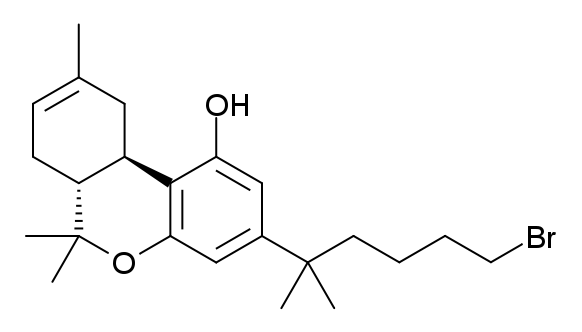
AM-919
AM-919 (part of the AM cannabinoid series) is an analgesic drug which is a cannabinoid receptor agonist. It is a derivative of HU-210 which has been substituted with a 6β-(3-hydroxypropyl) group. This adds a "southern" aliphatic hydroxyl group to the molecule as seen in the CP-series of nonclassical cannabinoid drugs, and so AM-919 represents a hybrid structure between the classical dibenzopyran and nonclassical cannabinoid families. AM-919 is somewhat less potent than HU-210 itself, but is still a potent agonist at both CB1 and CB2 with moderate selectivity for CB1, with a Ki of 2.2 nM at CB1 and 3.4 nM at CB2. See also * AM-4030 AM-4030 is an analgesic drug which is a cannabinoid receptor agonist. It is a derivative of HU-210 which has been substituted with a 6β-((E)-3-hydroxyprop-1-enyl) group. This adds a "southern" aliphatic hydroxyl group to the molecule as seen in ... References Benzochromenes Primary alcohols Phenols AM cannabinoids ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
List Of AM Cannabinoids
Alexandros Makriyannis is a professor in the Department of Medicinal Chemistry at Northeastern University, where his research group has synthesized many new compounds with cannabinoid activity. Some of those are: See also * List of CP cannabinoids * List of JWH cannabinoids * List of HU cannabinoids * List of miscellaneous designer cannabinoids Since the first synthetic cannabinoids were discovered in recreational drug products in 2008, new synthetic cannabinoids with no precedent in the scientific literature continue to be identified. These synthetic cannabinoids appear to be rationally ... References {{Cannabinoids ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
AM-4030
AM-4030 is an analgesic drug which is a cannabinoid receptor agonist. It is a derivative of HU-210 which has been substituted with a 6β-((E)-3-hydroxyprop-1-enyl) group. This adds a "southern" aliphatic hydroxyl group to the molecule as seen in the CP-series of nonclassical cannabinoid drugs, and so AM-4030 represents a hybrid structure between the classical and nonclassical cannabinoid families, with the 6-hydroxyalkyl chain rigidified with a double bond with defined stereochemistry. This gives AM-4030 a greater degree of selectivity, so while it is still a potent agonist at both CB1 and CB2, it is reasonably selective for CB1, with a Ki of 0.7nM at CB1 and 8.6nM at CB2, a selectivity of around 12x. Resolution of the enantiomers of AM-4030 yields an even more potent compound, although with less selectivity, with the (-) enantiomer AM-4030a having a Ki of 0.6nM at CB1 and 1.1nM at CB2. See also * AM-919 * AM-938 AM-938 (part of the AM cannabinoid series) is an analgesi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Analgesic
An analgesic drug, also called simply an analgesic (American English), analgaesic (British English), pain reliever, or painkiller, is any member of the group of drugs used to achieve relief from pain (that is, analgesia or pain management). It is typically used to induce cooperation with a medical procedure. Analgesics are conceptually distinct from anesthetics, which temporarily reduce, and in some instances eliminate, sensation, although analgesia and anesthesia are neurophysiologically overlapping and thus various drugs have both analgesic and anesthetic effects. Analgesic choice is also determined by the type of pain: For neuropathic pain, traditional analgesics are less effective, and there is often benefit from classes of drugs that are not normally considered analgesics, such as tricyclic antidepressants and anticonvulsants. Various analgesics, such as many NSAIDs, are available over the counter in most countries, whereas various others are prescription drugs owing ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cannabinoid Receptor Agonist
Cannabinoids () are several structural classes of compounds found in the cannabis plant primarily and most animal organisms (although insects lack such receptors) or as synthetic compounds. The most notable cannabinoid is the phytocannabinoid tetrahydrocannabinol (THC) (delta-9-THC), the primary intoxicating compound in cannabis. Cannabidiol (CBD) is a major constituent of temperate Cannabis plants and a minor constituent in tropical varieties. At least 113 distinct phytocannabinoids have been isolated from cannabis, although only four (i.e., THCA, CBDA, CBCA and their common precursor CBGA) have been demonstrated to have a biogenetic origin. It was reported in 2020 that phytocannabinoids can be found in other plants such as rhododendron, licorice and liverwort, and earlier in Echinacea. Phytocannabinoids are multi-ring phenolic compounds structurally related to THC, but endocannabinoids are fatty acid derivatives. Nonclassical synthetic cannabinoids (cannabimimetics) include amin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
HU-210
HU-210 is a synthetic cannabinoid that was first chemical synthesis, synthesized in 1988 from (1R,5S)-myrtenol by a group led by Raphael Mechoulam at the Hebrew University. HU-210 is 100 to 800 times more potent than natural Tetrahydrocannabinol, THC from cannabis (drug), cannabis and has an extended duration of action. HU-210 has a binding affinity of 0.061nM at CB1 and 0.52nM at CB2 in cloned human cannabinoid receptors. Compared to Delta-9-THC of 40.7nM at CB1. HU-210 is the (–)-1,1-dimethylheptyl analog of 11-hydroxy- Δ8- tetrahydrocannabinol; in some references it is called 1,1-dimethylheptyl- 11-hydroxytetrahydrocannabinol. The abbreviation "HU" stands for Hebrew University. Effects and research HU-210, the (–) enantiomer of 11-OH-D8-THC-DMH, has almost all of the cannabinoid activity, while the (+) enantiomer, known as HU-211, is inactive as a cannabinoid and instead acts as an NMDA antagonist having neuroprotective effects. HU-210 promotes cell proliferation, pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aliphatic
In organic chemistry, hydrocarbons ( compounds composed solely of carbon and hydrogen) are divided into two classes: aromatic compounds and aliphatic compounds (; G. ''aleiphar'', fat, oil). Aliphatic compounds can be saturated, like hexane, or unsaturated, like hexene and hexyne. Open-chain compounds, whether straight or branched, and which contain no rings of any type, are always aliphatic. Cyclic compounds can be aliphatic if they are not aromatic. Structure Aliphatic compounds can be saturated, joined by single bonds (alkanes), or unsaturated, with double bonds (alkenes) or triple bonds (alkynes). If other elements (heteroatoms) are bound to the carbon chain, the most common being oxygen, nitrogen, sulfur, and chlorine, it is no longer a hydrocarbon, and therefore no longer an aliphatic compound. The least complex aliphatic compound is methane (CH4). Properties Most aliphatic compounds are flammable, allowing the use of hydrocarbons as fuel, such as methane in Buns ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy groups. Both the negatively charged anion , called hydroxide, and the neutral radical , known as the hydroxyl radical, consist of an unbonded hydroxy group. According to IUPAC definitions, the term ''hydroxyl'' refers to the hydroxyl radical () only, while the functional group is called a ''hydroxy group''. Properties Water, alcohols, carboxylic acids, and many other hydroxy-containing compounds can be readily deprotonated due to a large difference between the electronegativity of oxygen (3.5) and that of hydrogen (2.1). Hydroxy-containing compounds engage in intermolecular hydrogen bonding increasing the electrostatic attraction between molecules and thus to higher boiling and melting points than found for compounds that lack this f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CP Cannabinoids
Many synthetic cannabinoids were designed by Pfizer in the 1970s and 1980s, and feature an alphanumeric code beginning with the prefix "CP" (after Charles Pfizer). Recently, several members of this class of cannabinoids have been discovered in recreational drug products. * CP 47,497 — * (C6)-CP 47,497 — * (C7)-CP 47,497 (CP 47,497 itself) — * (C8)-CP 47,497 (Cannabicyclohexanol) — * (C9)-CP 47,497 — * CP 50,556-1 (Levonantradol) — * CP 55,244 — * CP 55,940 — * (±)-CP 55,940 — (±)-CP 55,940 is a widely used cannabinoid research tool. * (+)-CP 55,940 — * (-)-CP 55,940 — * CP-945,598 (Otenabant) — See also * List of AM cannabinoids * List of HU cannabinoids * List of JWH cannabinoids * List of miscellaneous designer cannabinoids Since the first synthetic cannabinoids were discovered in recreational drug products in 2008, new synthetic cannabinoids with no precedent in the scientific literature conti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dibenzopyran
Xanthene (9''H''-xanthene, 10''H''-9-oxaanthracene) is the organic compound with the formula CH2 6H4sub>2O. It is a yellow solid that is soluble in common organic solvents. Xanthene itself is an obscure compound, but many of its derivatives are useful dyes. Xanthene dyes Dyes that contain a xanthene core include fluorescein, eosins, and rhodamines. Xanthene dyes tend to be fluorescent, yellow to pink to bluish red, brilliant dyes. Many xanthene dyes can be prepared by condensation of derivates of phthalic anhydride with derivates of resorcinol or 3-aminophenol. Further reading * See also * Xanthone Xanthone is an organic compound with the molecular formula O 6H4CO. It is a white solid. In 1939, xanthone was introduced as an insecticide and it currently finds uses as Insecticide, ovicide for codling moth eggs and as a larvicide. Xanthone ... * Xanthydrol References {{reflist Fungicides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cannabinoid
Cannabinoids () are several structural classes of compounds found in the cannabis plant primarily and most animal organisms (although insects lack such receptors) or as synthetic compounds. The most notable cannabinoid is the phytocannabinoid tetrahydrocannabinol (THC) (delta-9-THC), the primary intoxicating compound in cannabis. Cannabidiol (CBD) is a major constituent of temperate Cannabis plants and a minor constituent in tropical varieties. At least 113 distinct phytocannabinoids have been isolated from cannabis, although only four (i.e., THCA, CBDA, CBCA and their common precursor CBGA) have been demonstrated to have a biogenetic origin. It was reported in 2020 that phytocannabinoids can be found in other plants such as rhododendron, licorice and liverwort, and earlier in Echinacea. Phytocannabinoids are multi-ring phenolic compounds structurally related to THC, but endocannabinoids are fatty acid derivatives. Nonclassical synthetic cannabinoids (cannabimimetics) include amin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cannabinoid Receptor 1
Cannabinoid receptor type 1 (CB1), also known as cannabinoid receptor 1, is a G protein-coupled cannabinoid receptor that in humans is encoded by the ''CNR1'' gene. The human CB1 receptor is expressed in the peripheral nervous system and central nervous system. It is activated by: endocannabinoids, a group of retrograde neurotransmitters that include anandamide and 2-arachidonoylglycerol (2-AG); plant phytocannabinoids, such as the compound THC which is an active ingredient of the psychoactive drug cannabis; and, synthetic analogs of THC. CB1 is antagonized by the phytocannabinoid tetrahydrocannabivarin (THCV). The primary endogenous agonist of the human CB1 receptor is anandamide. Structure The CB1 receptor shares the structure characteristic of all G-protein-coupled receptors, possessing seven transmembrane domains connected by three extracellular and three intracellular loops, an extracellular N-terminal tail, and an intracellular C-terminal tail. The receptor may exist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cannabinoid Receptor 2
The cannabinoid receptor type 2, abbreviated as CB2, is a G protein-coupled receptor from the cannabinoid receptor family that in humans is encoded by the ''CNR2'' gene. It is closely related to the cannabinoid receptor type 1 (CB1), which is largely responsible for the efficacy of endocannabinoid-mediated presynaptic-inhibition, the psychoactive properties of tetrahydrocannabinol (THC), the active agent in cannabis, and other phytocannabinoids (plant cannabinoids). The principal endogenous ligand for the CB2 receptor is 2-Arachidonoylglycerol (2-AG). CB2 was cloned in 1993 by a research group from Cambridge looking for a second cannabinoid receptor that could explain the pharmacological properties of tetrahydrocannabinol. The receptor was identified among cDNAs based on its similarity in amino-acid sequence to the cannabinoid receptor type 1 (CB1) receptor, discovered in 1990. The discovery of this receptor helped provide a molecular explanation for the established effects of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |