|
ACOT1
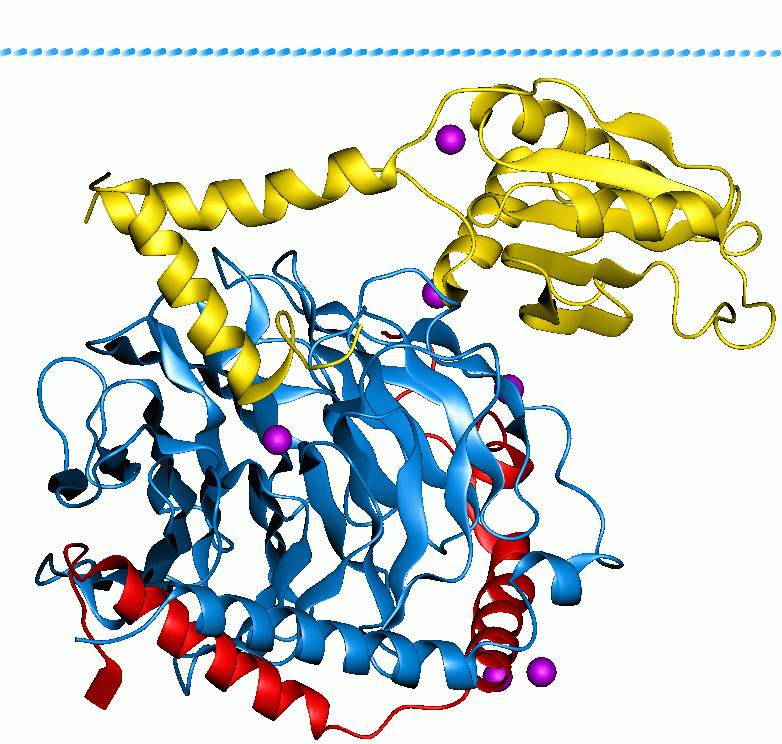
Acyl-CoA thioesterase 1 is a protein that in humans is encoded by the ACOT1 gene. Structure The ACOT1 gene is located on the 14th chromosome, with its specific localization being 14q24.3. It contains 7 exons. The protein encoded by this gene contains 410 amino acids, and forms a homodimer with another chain. The protein contains a StAR-related transfer domain, which is a domain responsible for binding to lipids. There are 4 known ligands that bind to this homodimer: polyethylene glycol, chlorine, glycerol, and a form of TCEP. Function The protein encoded by the ACOT1 gene is part of a family of Acyl-CoA thioesterases, which catalyze the hydrolysis of various Coenzyme A esters of various molecules to the free acid plus CoA. These enzymes have also been referred to in the literature as acyl-CoA hydrolases, acyl-CoA thioester hydrolases, and palmitoyl-CoA hydrolases. The reaction carried out by these enzymes is as follows: CoA ester + H2O → free acid + coenzyme A These enzy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thioesterase
Thioesterases are enzymes which belong to the esterase family. Esterases, in turn, are one type of the several hydrolases known. Thioesterases exhibit esterase activity (splitting of an ester into acid and alcohol, in the presence of water) specifically at a thiol group. Thioesterases or thiolester hydrolases are identified as members of EC 3.1.2. Family The thioesterase activity is performed by members of the acyl-CoA thioesterase (ACOT) family. The regulatory role of ACOT in fatty acid metabolism depends on their substrate specificity, tissue expression and subcellular localization. For example, deactivation of fatty acids at the ER may traffic fatty acids away from pathways associated with the ER membrane, such as glycerolipid biosynthesis. Two structurally different ACOT types lead to a similar enzymatic activity in vitro, dividing the family into type I and type II ACOTs. Type I ACOTs (ACOT1–6) contain the α/β-hydrolase domain, which is also present in many lipases an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
StAR-related Transfer Domain
START (StAR-related lipid-transfer) is a lipid-binding domain in StAR, HD-ZIP and signalling proteins. The archetypical domain is found in StAR (Steroidogenic acute regulatory protein), a mitochondrial protein that is synthesized in steroid-producing cells. StAR initiates steroid production by mediating the delivery of cholesterol to the first enzyme in steroidogenic pathway. The START domain is critical for this activity, perhaps through the binding of cholesterol. Following the discovery of StAR, 15 START-domain-containing proteins (termed STARD1 through STARD15) were subsequently identified in vertebrates as well as other that are related. Thousands of proteins containing at least one START domain have been determined in invertebrates, bacteria and plants to form a larger superfamily, variously known as START, Bet v1-like or SRPBCC (START/RHOalphaC/ PITP/Bet v1/CoxG/CalC) domain proteins, all of which bind hydrophobic ligands. In the case of plants, many of the START pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, responding to stimuli, providing structure to cells and organisms, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in protein folding into a specific 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called peptides. The individual amino acid residues are bonded together by peptide bonds and adjacent amino acid residues. The sequence of amino acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ATP-sensitive Potassium Channel
An ATP-sensitive potassium channel (or KATP channel) is a type of potassium channel that is gated by intracellular nucleotides, ATP and ADP. ATP-sensitive potassium channels are composed of Kir6.x-type subunits and sulfonylurea receptor (SUR) subunits, along with additional components. KATP channels are found in the plasma membrane; however some may also be found on subcellular membranes. These latter classes of KATP channels can be classified as being either sarcolemmal ("sarcKATP"), mitochondrial ("mitoKATP"), or nuclear ("nucKATP"). Discovery and structure KATP channels were first identified in cardiac myocytes by the Akinori Noma group in Japan. They have also been found in pancreas where they control insulin secretion, but are in fact widely distributed in plasma membranes. SarcKATP are composed of eight protein subunits (octamer). Four of these are members of the inward-rectifier potassium ion channel family Kir6.x (either Kir6.1 or Kir6.2), while the other four ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
NADH
Nicotinamide adenine dinucleotide (NAD) is a coenzyme central to metabolism. Found in all living cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine nucleobase and the other nicotinamide. NAD exists in two forms: an oxidized and reduced form, abbreviated as NAD and NADH (H for hydrogen), respectively. In metabolism, nicotinamide adenine dinucleotide is involved in redox reactions, carrying electrons from one reaction to another. The cofactor is, therefore, found in two forms in cells: NAD is an oxidizing agent – it accepts electrons from other molecules and becomes reduced. This reaction, also with H+, forms NADH, which can then be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD. However, it is also used in other cellular processes, most notably as a substrate of enzymes in adding or removing chemical groups to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amino Acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha amino acids appear in the genetic code. Amino acids can be classified according to the locations of the core structural functional groups, as Alpha and beta carbon, alpha- , beta- , gamma- or delta- amino acids; other categories relate to Chemical polarity, polarity, ionization, and side chain group type (aliphatic, Open-chain compound, acyclic, aromatic, containing hydroxyl or sulfur, etc.). In the form of proteins, amino acid '' residues'' form the second-largest component (water being the largest) of human muscles and other tissues. Beyond their role as residues in proteins, amino acids participate in a number of processes such as neurotransmitter transport and biosynthesis. It is thought that they played a key role in enabling l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dehydrogenase
A dehydrogenase is an enzyme belonging to the group of oxidoreductases that oxidizes a substrate by reducing an electron acceptor, usually NAD+/NADP+ or a flavin coenzyme such as FAD or FMN. Like all catalysts, they catalyze reverse as well as forward reactions, and in some cases this has physiological significance: for example, alcohol dehydrogenase catalyzes the oxidation of ethanol to acetaldehyde in animals, but in yeast it catalyzes the production of ethanol from acetaldehyde. IUBMB Classification Oxidoreductases, enzymes that catalyze oxidation-reduction reactions, constitute Class EC 1 of the IUBMB classification of enzyme-catalyzed reactions. Any of these may be called dehydrogenases, especially those in which NAD+ is the electron acceptor (oxidant), but reductase is also used when the physiological emphasis on reduction of the substrate, and oxidase is used ''only'' when O2 is the electron acceptor. The systematic name of an oxidoreductase is "donor:acceptor oxidore ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
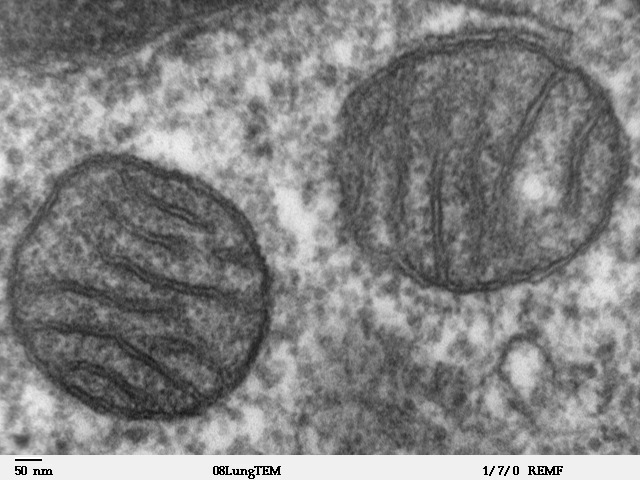
Mitochondria
A mitochondrion (; ) is an organelle found in the cells of most Eukaryotes, such as animals, plants and fungi. Mitochondria have a double membrane structure and use aerobic respiration to generate adenosine triphosphate (ATP), which is used throughout the cell as a source of chemical energy. They were discovered by Albert von Kölliker in 1857 in the voluntary muscles of insects. The term ''mitochondrion'' was coined by Carl Benda in 1898. The mitochondrion is popularly nicknamed the "powerhouse of the cell", a phrase coined by Philip Siekevitz in a 1957 article of the same name. Some cells in some multicellular organisms lack mitochondria (for example, mature mammalian red blood cells). A large number of unicellular organisms, such as microsporidia, parabasalids and diplomonads, have reduced or transformed their mitochondria into other structures. One eukaryote, ''Monocercomonoides'', is known to have completely lost its mitochondria, and one multicellular organism, ' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
G Alpha Subunit
G alpha subunits are one of the three types of subunit of guanine nucleotide binding proteins, which are membrane-associated, heterotrimeric G proteins. Background G proteins and their receptors (GPCRs) form one of the most prevalent signalling systems in mammalian cells, regulating systems as diverse as sensory perception, cell growth and hormonal regulation. At the cell surface, the binding of ligands such as hormones and neurotransmitters to a GPCR activates the receptor by causing a conformational change, which in turn activates the bound G protein on the intracellular-side of the membrane. The activated receptor promotes the exchange of bound GDP for GTP on the G protein alpha subunit. GTP binding changes the conformation of switch regions within the alpha subunit, which allows the bound trimeric G protein (inactive) to be released from the receptor, and to dissociate into active alpha subunit (GTP-bound) and beta/gamma dimer. The alpha subunit and the beta/gamma dimer go ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
G Protein
G proteins, also known as guanine nucleotide-binding proteins, are a family of proteins that act as molecular switches inside cells, and are involved in transmitting signals from a variety of stimuli outside a cell to its interior. Their activity is regulated by factors that control their ability to bind to and hydrolyze guanosine triphosphate (GTP) to guanosine diphosphate (GDP). When they are bound to GTP, they are 'on', and, when they are bound to GDP, they are 'off'. G proteins belong to the larger group of enzymes called GTPases. There are two classes of G proteins. The first function as monomeric small GTPases (small G-proteins), while the second function as heterotrimeric G protein complexes. The latter class of complexes is made up of ''alpha'' (α), ''beta'' (β) and ''gamma'' (γ) subunits. In addition, the beta and gamma subunits can form a stable dimeric complex referred to as the beta-gamma complex . Heterotrimeric G proteins located within the cell are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Endomembrane System
The endomembrane system is composed of the different membranes (endomembranes) that are suspended in the cytoplasm within a eukaryotic cell. These membranes divide the cell into functional and structural compartments, or organelles. In eukaryotes the organelles of the endomembrane system include: the nuclear membrane, the endoplasmic reticulum, the Golgi apparatus, lysosomes, vesicles, endosomes, and plasma (cell) membrane among others. The system is defined more accurately as the set of membranes that forms a single functional and developmental unit, either being connected directly, or exchanging material through vesicle transport. Importantly, the endomembrane system does not include the membranes of plastids or mitochondria, but might have evolved partially from the actions of the latter (see below). The nuclear membrane contains a lipid bilayer that encompasses the contents of the nucleus. The endoplasmic reticulum (ER) is a synthesis and transport organelle that branches int ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Retinoic Acid
Retinoic acid (used simplified here for all-''trans''-retinoic acid) is a metabolite of vitamin A1 (all-''trans''-retinol) that mediates the functions of vitamin A1 required for growth and development. All-''trans''-retinoic acid is required in chordate animals, which includes all higher animals from fish to humans. During early embryonic development, all-''trans''-retinoic acid generated in a specific region of the embryo helps determine position along the embryonic anterior/posterior axis by serving as an intercellular signaling molecule that guides development of the posterior portion of the embryo. It acts through Hox genes, which ultimately control anterior/posterior patterning in early developmental stages. All-''trans''-retinoic acid (ATRA) is the major occurring retinoic acid, while isomers like 13-''cis''- and 9-''cis''-retinoic acid are also present in much lower levels. The key role of all-''trans''-retinoic acid in embryonic development mediates the high teratogeni ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |