|
Amyloid-beta
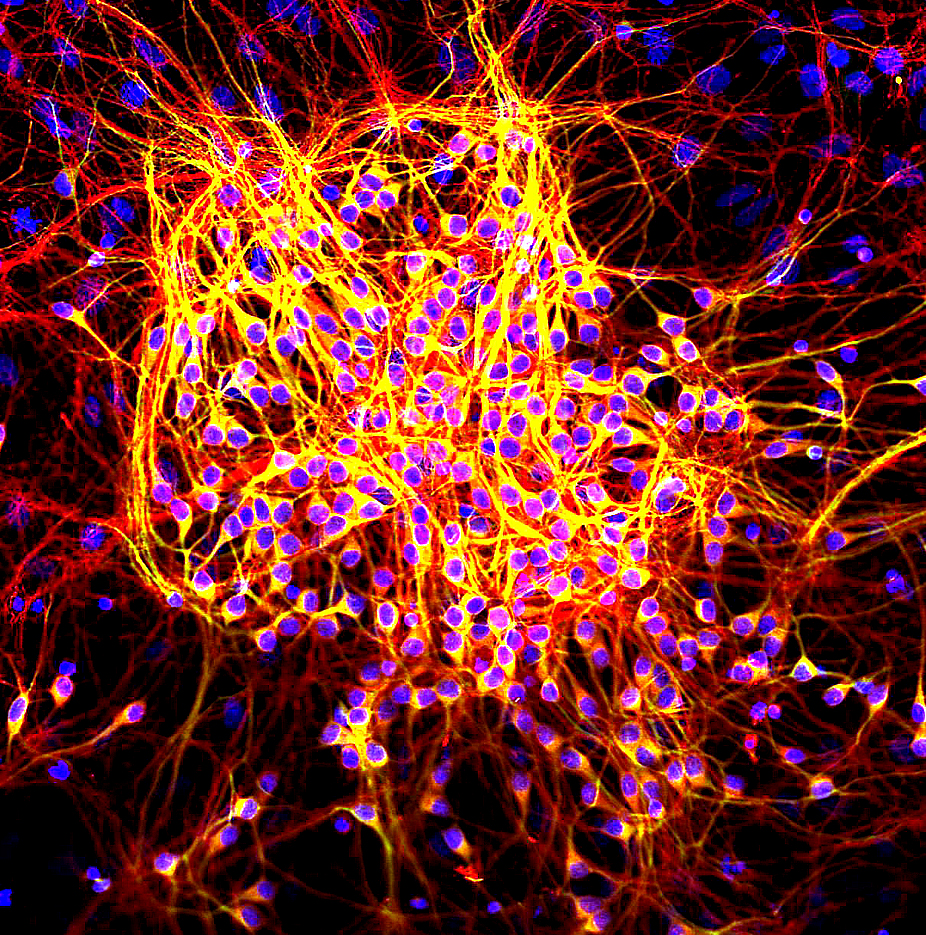
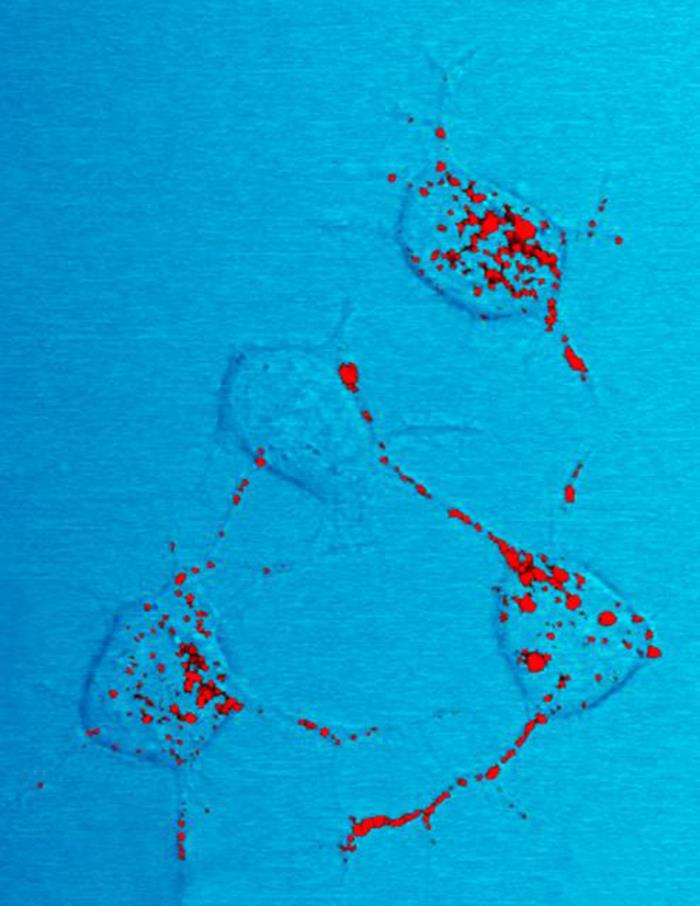
Amyloid beta (Aβ or Abeta) denotes peptides of 36–43 amino acids that are the main component of the amyloid plaques found in the brains of people with Alzheimer's disease. The peptides derive from the amyloid precursor protein (APP), which is cleaved by beta secretase and gamma secretase to yield Aβ in a cholesterol-dependent process and substrate presentation. Aβ molecules can aggregate to form flexible soluble oligomers which may exist in several forms. It is now believed that certain misfolded oligomers (known as "seeds") can induce other Aβ molecules to also take the misfolded oligomeric form, leading to a chain reaction akin to a prion infection. The oligomers are toxic to nerve cells. The other protein implicated in Alzheimer's disease, tau protein, also forms such prion-like misfolded oligomers, and there is some evidence that misfolded Aβ can induce tau to misfold. A study has suggested that APP and its amyloid potential is of ancient origins, dating as far back as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amyloid Precursor Protein
Amyloid-beta precursor protein (APP) is an integral membrane protein expressed in many biological tissue, tissues and concentrated in the synapses of neurons. It functions as a cell surface receptor and has been implicated as a regulator of synapse formation, neural plasticity, antimicrobial activity, and iron export. It is coded for by the gene ''APP'' and regulated by substrate presentation. APP is best known as the precursor molecule whose proteolysis generates amyloid beta (Aβ), a polypeptide containing 37 to 49 amino acid residues, whose Amyloid#Structure, amyloid fibrillar form is the primary component of amyloid plaques found in the brains of Alzheimer's disease patients. Genetics Amyloid-beta precursor protein is an ancient and highly Conserved sequence, conserved protein. In humans, the gene ''APP'' is located on chromosome 21 and contains 18 exons spanning 290 kilobases. Several alternative splicing isoforms of APP have been observed in humans, ranging in len ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amyloid Precursor Protein
Amyloid-beta precursor protein (APP) is an integral membrane protein expressed in many biological tissue, tissues and concentrated in the synapses of neurons. It functions as a cell surface receptor and has been implicated as a regulator of synapse formation, neural plasticity, antimicrobial activity, and iron export. It is coded for by the gene ''APP'' and regulated by substrate presentation. APP is best known as the precursor molecule whose proteolysis generates amyloid beta (Aβ), a polypeptide containing 37 to 49 amino acid residues, whose Amyloid#Structure, amyloid fibrillar form is the primary component of amyloid plaques found in the brains of Alzheimer's disease patients. Genetics Amyloid-beta precursor protein is an ancient and highly Conserved sequence, conserved protein. In humans, the gene ''APP'' is located on chromosome 21 and contains 18 exons spanning 290 kilobases. Several alternative splicing isoforms of APP have been observed in humans, ranging in len ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glymphatic System
The glymphatic system (or glymphatic clearance pathway, or paravascular system) was described and named in 2013 as a system for waste clearance in the central nervous system (CNS) of vertebrates. According to this model, cerebrospinal fluid (CSF) flows into the paravascular space around cerebral arteries, combining with interstitial fluid (ISF) and parenchymal solutes, and exiting down venous paravascular spaces. The pathway consists of a para-arterial influx route for CSF to enter the brain parenchyma, coupled to a clearance mechanism for the removal of interstitial fluid (ISF) and extracellular solutes from the interstitial compartments of the brain and spinal cord. Exchange of solutes between CSF and ISF is driven primarily by arterial pulsation and regulated during sleep by the expansion and contraction of brain extracellular space. Clearance of soluble proteins, waste products, and excess extracellular fluid is accomplished through convective bulk flow of ISF, facilitated by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alzheimer's Disease
Alzheimer's disease (AD) is a neurodegeneration, neurodegenerative disease that usually starts slowly and progressively worsens. It is the cause of 60–70% of cases of dementia. The most common early symptom is difficulty in short-term memory, remembering recent events. As the disease advances, symptoms can include primary progressive aphasia, problems with language, Orientation (mental), disorientation (including easily getting lost), mood swings, loss of motivation, self-neglect, and challenging behaviour, behavioral issues. As a person's condition declines, they often withdraw from family and society. Gradually, bodily functions are lost, ultimately leading to death. Although the speed of progression can vary, the typical life expectancy following diagnosis is three to nine years. The cause of Alzheimer's disease is poorly understood. There are many environmental and genetic risk factors associated with its development. The strongest genetic risk factor is from an alle ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cerebral Amyloid Angiopathy
Cerebral amyloid angiopathy (CAA) is a form of angiopathy in which amyloid beta peptide deposits in the walls of small to medium blood vessels of the central nervous system and meninges. The term ''congophilic'' is sometimes used because the presence of the abnormal aggregations of amyloid can be demonstrated by microscopic examination of brain tissue after staining with Congo red. The amyloid material is only found in the brain and as such the disease is not related to other forms of amyloidosis. Signs and symptoms CAA is associated with brain hemorrhages, particularly microhemorrhages. Since CAA can be caused by the same amyloid protein that is associated with Alzheimer's dementia, brain bleeds are more common in people who have a diagnosis of Alzheimer's disease. However, they can also occur in those who have no history of dementia. The bleeding within the brain is usually confined to a particular lobe and this is slightly different compared to brain bleeds which occur as a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amyloid
Amyloids are aggregates of proteins characterised by a Fibril, fibrillar morphology of 7–13 Nanometer, nm in diameter, a beta sheet (β-sheet) Secondary structure of proteins, secondary structure (known as cross-β) and ability to be Staining, stained by particular dyes, such as Congo red. In the human body, amyloids have been linked to the development of various diseases. Pathogenic amyloids form when previously healthy proteins lose their normal Protein structure, structure and physiology, physiological functions (Protein misfolding, misfolding) and form fibrous deposits in amyloid plaques around cells which can disrupt the healthy function of tissues and organs. Such amyloids have been associated with (but not necessarily as the cause of) more than 50 human diseases, known as amyloidosis, and may play a role in some neurodegenerative diseases. Some of these diseases are mainly sporadic and only a few cases are Genetic disorder, familial. Others are only Genetic disorder, fam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tau Protein
The tau proteins (abbreviated from tubulin associated unit) are a group of six highly soluble protein isoforms produced by alternative splicing from the gene ''MAPT'' (microtubule-associated protein tau). They have roles primarily in maintaining the stability of microtubules in axons and are abundant in the neurons of the central nervous system (CNS), where the cerebral cortex has the highest abundance. They are less common elsewhere but are also expressed at very low levels in CNS astrocytes and oligodendrocytes. Pathologies and dementias of the nervous system such as Alzheimer's disease and Parkinson's disease are associated with tau proteins that have become hyperphosphorylated insoluble aggregates called neurofibrillary tangles. The tau proteins were identified in 1975 as heat-stable proteins essential for microtubule assembly, and since then they have been characterized as intrinsically disordered proteins. Function Microtubule stabilization Tau proteins are found mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Prion
Prions are misfolded proteins that have the ability to transmit their misfolded shape onto normal variants of the same protein. They characterize several fatal and transmissible neurodegenerative diseases in humans and many other animals. It is not known what causes a normal protein to misfold, but the resulting abnormal three-dimensional structure confers infectious properties by collapsing nearby protein molecules into the same shape. The word ''prion'' is derived from the term, "proteinaceous infectious particle". In comparison to all other known infectious agents such as viroids, viruses, bacteria, fungi, and parasites, all of which contain nucleic acids ( DNA, RNA, or both), the hypothesized role of a protein as an infectious agent stands in contrast. Prion isoforms of the prion protein (PrP), whose specific function is uncertain, are hypothesized as the cause of transmissible spongiform encephalopathies (TSEs), including scrapie in sheep, chronic wasting disease (CWD) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ion Channel Hypothesis Of Alzheimer's Disease
The ion channel hypothesis of Alzheimer’s disease (AD), also known as the channel hypothesis or the amyloid beta ion channel hypothesis, is a more recent variant of the amyloid hypothesis of AD, which identifies amyloid beta (Aβ) as the underlying cause of neurotoxicity seen in AD. While the traditional formulation of the amyloid hypothesis pinpoints insoluble, fibrillar aggregates of Aβ as the basis of disruption of calcium ion homeostasis and subsequent apoptosis in AD, the ion channel hypothesis in 1993 introduced the possibility of an ion-channel-forming oligomer of soluble, non-fibrillar Aβ as the cytotoxic species allowing unregulated calcium influx into neurons in AD. The ion channel hypothesis is broadly supported as an explanation for the calcium ion influx that disrupts calcium ion homeostasis and induces apoptosis in neurons. Because the extracellular deposition of Aβ fibrils in senile plaques is not sufficient to predict risk or onset of AD, and clinical ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beta-secretase 1
Beta-secretase 1, also known as beta-site amyloid precursor protein cleaving enzyme 1, beta-site APP cleaving enzyme 1 (BACE1), membrane-associated aspartic protease 2, memapsin-2, aspartyl protease 2, and ASP2, is an enzyme that in humans is encoded by the ''BACE1'' gene. Expression of BACE1 is observed mainly in neurons. BACE1 is an aspartic acid protease important in the formation of myelin sheaths in peripheral nerve cells: in mice the expression of BACE1 is high in the postnatal stages, when myelination occurs. * The transmembrane protein contains two active site aspartate residues in its extracellular protein domain and may function as a dimer, its cytoplasmic tail is required for the correct maturation and an efficient intracellular trafficking, but doesn't affect the activity. It is produced as a pro-enzyme, the endoproteolitc removal occurs after BACE leaves Endoplasmic reticulum, in the Golgi apparatus. In addition the pro-peptide receives additional sugars to increase t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amyloid Plaques
Amyloid plaques (also known as neuritic plaques, amyloid beta plaques or senile plaques) are extracellular deposits of the amyloid beta (Aβ) protein mainly in the grey matter of the brain. Degenerative neuronal elements and an abundance of microglia and astrocytes can be associated with amyloid plaques. Some plaques occur in the brain as a result of aging, but large numbers of plaques and neurofibrillary tangles are characteristic features of Alzheimer's disease. Abnormal neurites in amyloid plaques are tortuous, often swollen axons and dendrites. The neurites contain a variety of organelles and cellular debris, and many of them include characteristic paired helical filaments, the ultrastructural component of neurofibrillary tangles. The plaques are highly variable in shape and size; in tissue sections immunostained for Aβ, they comprise a log-normal size distribution curve with an average plaque area of 400-450 square micrometers (µm²). The smallest plaques (less than 200 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amyloid Plaque
Amyloid plaques (also known as neuritic plaques, amyloid beta plaques or senile plaques) are extracellular deposits of the amyloid beta (Aβ) protein mainly in the grey matter of the brain. Degenerative neuronal elements and an abundance of microglia and astrocytes can be associated with amyloid plaques. Some plaques occur in the brain as a result of aging, but large numbers of plaques and neurofibrillary tangles are characteristic features of Alzheimer's disease. Abnormal neurites in amyloid plaques are tortuous, often swollen axons and dendrites. The neurites contain a variety of organelles and cellular debris, and many of them include characteristic paired helical filaments, the ultrastructural component of neurofibrillary tangles. The plaques are highly variable in shape and size; in tissue sections immunostained for Aβ, they comprise a log-normal size distribution curve with an average plaque area of 400-450 square micrometers (µm²). The smallest plaques (less than 200 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |