|
Ammonia Borane
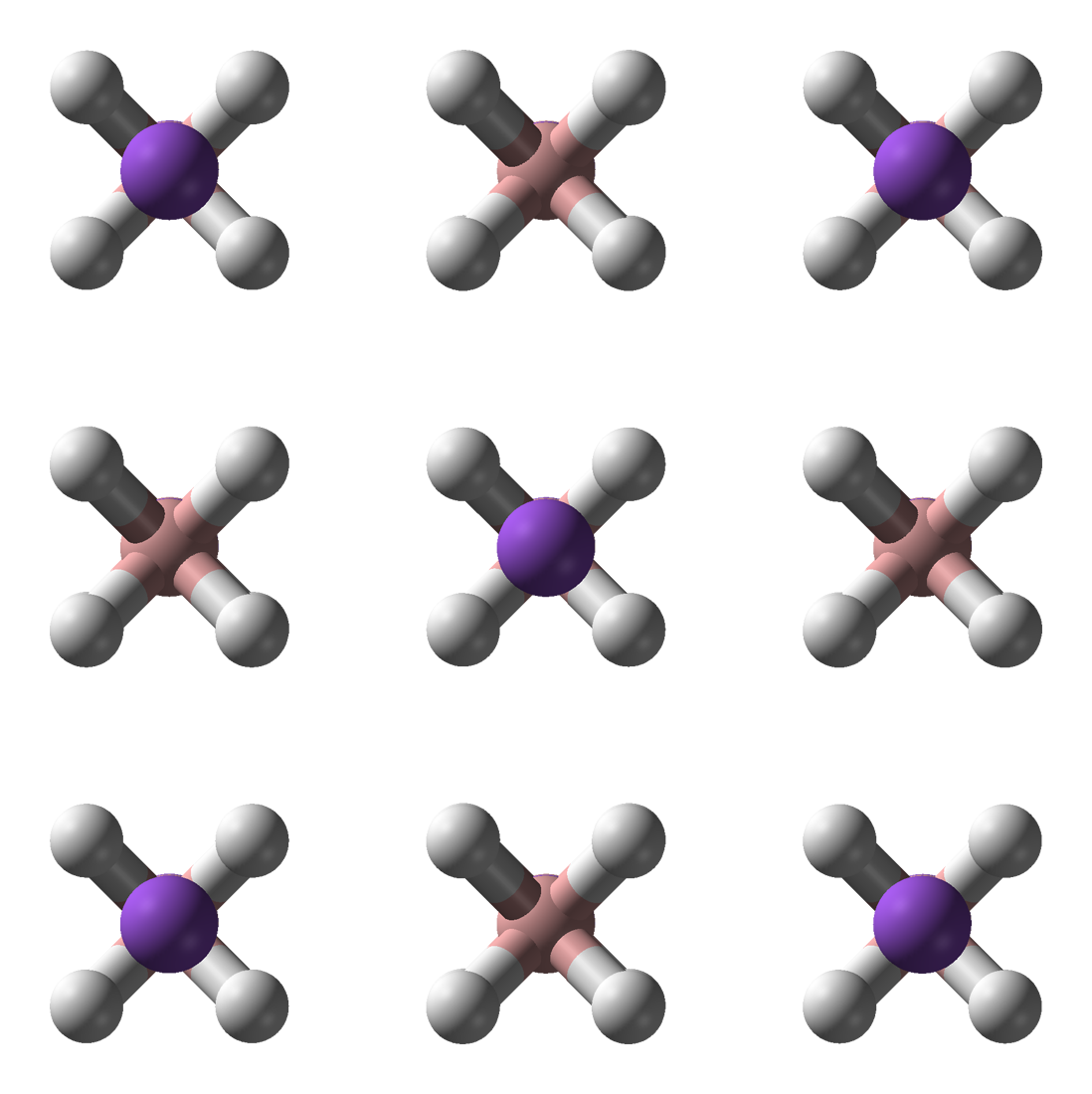
Ammonia borane (also systematically named amminetrihydridoboron), also called borazane, is the chemical compound with the formula H3NBH3. The colourless or white solid is the simplest molecular boron-nitrogen-hydride compound. It has attracted attention as a source of hydrogen fuel, but is otherwise primarily of academic interest. Synthesis Reaction of diborane with ammonia mainly gives the diammoniate salt 2B(NH3)2sup>+ (BH4)−. Ammonia borane is the main product when an adduct of borane is employed in place of diborane: :BH3(THF) + NH3 → BH3NH3 + THF Properties and structure The molecule adopts a structure similar to that of ethane, with which it is isoelectronic. The B−N distance is 1.58(2) Å. The B−H and N−H distances are 1.15 and 0.96 Å, respectively. Its similarity to ethane is tenuous since ammonia borane is a solid and ethane is a gas: their melting points differing by 284 °C. This difference is consistent with the highly polar natur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Borohydride
Sodium borohydride, also known as sodium tetrahydridoborate and sodium tetrahydroborate, is an inorganic compound with the formula Na BH4. This white solid, usually encountered as an aqueous basic solution, is a reducing agent that finds application in papermaking and dye industries. It is also used as a reagent in organic synthesis. The compound was discovered in the 1940s by H. I. Schlesinger, who led a team seeking volatile uranium compounds.Hermann I Schlesinger and Herbert C Brown (1945)Preparation of alkali metal compounds. US Patent 2461661. Granted on 1949-02-15; expired on 1966-02-15. Results of this wartime research were declassified and published in 1953. Properties The compound is soluble in alcohols, certain ethers, and water, although it slowly hydrolyzes. Sodium borohydride is an odorless white to gray-white microcrystalline powder that often forms lumps. It can be purified by recrystallization from warm (50 °C) diglyme. Sodium borohydride is soluble ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Journal Of The American Chemical Society
The ''Journal of the American Chemical Society'' is a weekly peer-reviewed scientific journal that was established in 1879 by the American Chemical Society. The journal has absorbed two other publications in its history, the ''Journal of Analytical and Applied Chemistry'' (July 1893) and the ''American Chemical Journal'' (January 1914). It covers all fields of chemistry. Since 2021, the editor-in-chief is Erick M. Carreira (ETH Zurich). In 2014, the journal moved to a hybrid open access publishing model. Abstracting and indexing The journal is abstracted and indexed in Chemical Abstracts Service, Scopus, EBSCO databases, ProQuest databases, Index Medicus/MEDLINE/PubMed, and the Science Citation Index Expanded. According to the ''Journal Citation Reports'', the journal has a 2021 impact factor of 16.383. Editors-in-chief The following people are or have been editor-in-chief: * 1879–1880 – Hermann Endemann * 1880–1881 – Gideon E. Moore * 1881–1882 – Hermann Endemann ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond. Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, and Biological Chemistry'. 1232 pages. Two general types of monoalkenes are distinguished: terminal and internal. Also called α-olefins, terminal alkenes are more useful. However, the International Union of Pure and Applied Chemistry (IUPAC) recommends using the name "alkene" only for acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkane
In organic chemistry, an alkane, or paraffin (a historical trivial name that also has other meanings), is an acyclic saturated hydrocarbon. In other words, an alkane consists of hydrogen and carbon atoms arranged in a tree structure in which all the carbon–carbon bonds are single. Alkanes have the general chemical formula . The alkanes range in complexity from the simplest case of methane (), where ''n'' = 1 (sometimes called the parent molecule), to arbitrarily large and complex molecules, like pentacontane () or 6-ethyl-2-methyl-5-(1-methylethyl) octane, an isomer of tetradecane (). The International Union of Pure and Applied Chemistry (IUPAC) defines alkanes as "acyclic branched or unbranched hydrocarbons having the general formula , and therefore consisting entirely of hydrogen atoms and saturated carbon atoms". However, some sources use the term to denote ''any'' saturated hydrocarbon, including those that are either monocyclic (i.e. the cycloalkanes) or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylene
Acetylene (systematic name: ethyne) is the chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in its pure form and thus is usually handled as a solution. Pure acetylene is odorless, but commercial grades usually have a marked odor due to impurities such as divinyl sulfide and phosphine.Compressed Gas Association (1995Material Safety and Data Sheet – Acetylene As an alkyne, acetylene is unsaturated because its two carbon atoms are bonded together in a triple bond. The carbon–carbon triple bond places all four atoms in the same straight line, with CCH bond angles of 180°. Discovery Acetylene was discovered in 1836 by Edmund Davy, who identified it as a "new carburet of hydrogen". It was an accidental discovery while attempting to isolate potassium metal. By heating potassium carbonate with carbon at very high temperatures, he produced a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylene
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon-carbon double bonds). Ethylene is widely used in the chemical industry, and its worldwide production (over 150 million tonnes in 2016) exceeds that of any other organic compound. Much of this production goes toward polyethylene, a widely used plastic containing polymer chains of ethylene units in various chain lengths. Ethylene is also an important natural plant hormone and is used in agriculture to force the ripening of fruits. The hydrate of ethylene is ethanol. Structure and properties This hydrocarbon has four hydrogen atoms bound to a pair of carbon atoms that are connected by a double bond. All six atoms that comprise ethylene are coplanar. The H-C-H angle is 117.4°, close to the 120° for ideal sp² hybridized carbon. The molecule is also relatively weak: rota ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iminoborane
Iminoboranes comprise a group of organoboron compounds with the formula RB=NR'. They are electronically related to acetylenes but are usually more reactive due to the polarity. Structure and bonding The parent iminoborane, HB=NH, is produced by the photolysis of H3BNH3. Bonding in iminoboranes can be described by two resonance structures In chemistry, resonance, also called mesomerism, is a way of describing bonding in certain molecules or polyatomic ions by the combination of several contributing structures (or ''forms'', also variously known as ''resonance structures'' or '' ...: :R-\overset\overset-R R-B=\ddot-R The stability is dramatically affected by bulky substituents. One isolable iminoborane is (''tert''-Bu)B=N(''tert''-Bu). Synthesis Elimination of fluoro- or chlorosilanes provides a well-tested route. Bulky substituents such as (Me3Si)3Si stabilize the iminoborane with respect to oligomerization: :(Me3Si)3SiB(F)-N(SiMe3)2 → (Me3Si)3Si-B=N-SiMe3 + F ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amine-borane
Ammonia borane (also systematically named amminetrihydridoboron), also called borazane, is the chemical compound with the formula H3NBH3. The colourless or white solid is the simplest molecular boron-nitrogen-hydride compound. It has attracted attention as a source of hydrogen fuel, but is otherwise primarily of academic interest. Synthesis Reaction of diborane with ammonia mainly gives the diammoniate salt + (BH4)−. Ammonia borane is the main product when an adduct of borane is employed in place of diborane: :BH3(THF) + NH3 → BH3NH3 + THF Properties and structure The molecule adopts a structure similar to that of ethane, with which it is isoelectronic. The B−N distance is 1.58(2) Å. The B−H and N−H distances are 1.15 and 0.96 Å, respectively. Its similarity to ethane is tenuous since ammonia borane is a solid and ethane is a gas: their melting points differing by 284 °C. This difference is consistent with the highly polar nature of ammonia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iminoborane
Iminoboranes comprise a group of organoboron compounds with the formula RB=NR'. They are electronically related to acetylenes but are usually more reactive due to the polarity. Structure and bonding The parent iminoborane, HB=NH, is produced by the photolysis of H3BNH3. Bonding in iminoboranes can be described by two resonance structures In chemistry, resonance, also called mesomerism, is a way of describing bonding in certain molecules or polyatomic ions by the combination of several contributing structures (or ''forms'', also variously known as ''resonance structures'' or '' ...: :R-\overset\overset-R R-B=\ddot-R The stability is dramatically affected by bulky substituents. One isolable iminoborane is (''tert''-Bu)B=N(''tert''-Bu). Synthesis Elimination of fluoro- or chlorosilanes provides a well-tested route. Bulky substituents such as (Me3Si)3Si stabilize the iminoborane with respect to oligomerization: :(Me3Si)3SiB(F)-N(SiMe3)2 → (Me3Si)3Si-B=N-SiMe3 + F ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Journal Of Molecular Spectroscopy
''Journal of Molecular Spectroscopy'' is a peer-reviewed scientific journal that deals with experimental and theoretical articles on all subjects relevant to molecular spectroscopy and its modern applications. Indexing and abstracting According to the ''Journal Citation Reports'', the journal has a 2020 impact factor of 1.507. The journal in indexing in the following bibliographic databases: * AGRICOLA * Chemical Abstracts * Current Contents/Physics, Chemical, & Earth Sciences * Research Alert * Science Abstracts * Science Citation Index * Biological Abstracts * Scopus Scopus is Elsevier's abstract and citation database launched in 2004. Scopus covers nearly 36,377 titles (22,794 active titles and 13,583 inactive titles) from approximately 11,678 publishers, of which 34,346 are peer-reviewed journals in top-l ... References External links * {{Official website, https://www.journals.elsevier.com/journal-of-molecular-spectroscopy/ Optics journals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |




.png)
