|
Acid Red 88
Acid red 88 is an azo dye. Due to its intense colour, solid samples appear almost black. It is used to dye cotton textiles red. A closely related acid dye is Acid Red 13. Preparation and use It can be obtained by azo coupling of naphthionic acid and 2-naphthol. Instead of crystallization, crystallising, it vitrification, vitrifies when cooled or salting out, salted out of the solution. : This compound is used in the textile industry as a dye. It can also be used for research in photocatalysis (as degradation object). References {{Reflist External links echo Chemical Database: 1-Naphthalenesulfonic acid, 4-((2-hydroxy-1-naphthalenyl)azo)-, monosodium salt (EnvironmentalChemistry.com) This page contains information on the chemical 1-Naphthalenesulfonic acid, 4-((2-hydroxy-1-naphthalenyl)azo)-, monosodium salt including: 72 synonyms/identifiers. Azo dyes 2-Naphthols ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Azo Dye
Azo dyes are organic compounds bearing the functional group R−N=N−R′, in which R and R′ are usually aryl and substituted aryl groups. They are a commercially important family of azo compounds, i.e. compounds containing the C-N=N-C linkage. Azo dyes are synthetic dyes and do not occur naturally. Most azo dyes contain only one azo group, but some dyes called "disazo dyes" contain two azo groups, some dyes called "trisazo dyes" contain three azo groups and are or more. Azo dyes comprise 60-70% of all dyes used in food and textile industries. Azo dyes are widely used to treat textiles, leather articles, and some foods. Chemically related derivatives of azo dyes include azo pigments, which are insoluble in water and other solvents. Classes Many kinds of azo dyes are known, and several classification systems exist. Some classes include disperse dyes, metal-complex dyes, reactive dyes, and substantive dyes. Also called direct dyes, substantive dyes are employed for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ullmann's Encyclopedia Of Industrial Chemistry
''Ullmann's Encyclopedia of Industrial Chemistry'' is a major reference work related to industrial chemistry by Chemist Fritz Ullmann, first published in 1914, and exclusively in German as "Enzyklopädie der Technischen Chemie" until 1984. History Ullmann's Encyclopedia of Industrial Chemistry is a major reference work related to industrial chemistry by chemist Fritz Ullmann. Its 1st edition was published in German by Fritz Ullmann in 1914. The 4th edition, published 1972 to 1984, already contained 25 volumes. The 5th edition, published 1985 to 1996, was the first version available in English. In 1997, the first online version was published. 2014 marked its centenary. As of 2016, Ullmann's Encyclopedia was in its 7th edition, in 40 volumes including one index volume and more than 1,050 articles (200 more than the 6th edition), approx. 30,000 pages, 22,000 images, 8,000 tables, 19,000 references and 85,000 indices. Editions * 1914–1922: 1st edition in 12 volumes, which can ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wiley-VCH
Wiley-VCH is a German publisher owned by John Wiley & Sons. It was founded in 1921 as Verlag Chemie (meaning "Chemistry Press": VCH stands for ''Verlag Chemie'') by two German learned societies. Later, it was merged into the German Chemical Society (GDCh). In 1991, VCH acquired Akademie Verlag :''There also were unrelated publishing houses in Stuttgart and in (East-)Berlin, and there is the (JAVG).'' Akademie Verlag (AV) is a German scientific and academic publishing company, founded in 1946 in the Soviet-occupied eastern part .... It has been owned by John Wiley & Sons since 1996. The humanities section of Akademie Verlag and the Akademie brand were sold in 1997 to R. Oldenbourg Verlag, while VCH retained the natural sciences catalog. References External links * Wiley (publisher) Publishing companies of Germany Publishing companies established in 1921 Weinheim German companies established in 1921 {{publish-company-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid Dye
Acid dyes are anionic, soluble in water and are essentially applied from acidic bath. These dyes possess acidic groups, such as SO3H and COOH and are applied on wool, silk and nylon when ionic bond is established between protonated –NH2 group of fibre and acid group of dye. Overall wash fastness is poor although lightfastness is quite good. As dye and fibre contain opposite electrical nature, strike rate and uptake of acid dye on these fibres is faster; electrolyte at higher concentration is added to retard dye uptake and to form levelled shades. Acid generates cation on fibre and temperature helps to substitute negative part of acid with anionic dye molecules. An acid dye is a dye that is typically applied to a textile at low pH. They are mainly used to dye wool, not cotton fabrics. Some acid dyes are used as food colorants, and some can also be used to stain organelles in the medical field. Description Acid dyes are generally divided into three classes according to their fast ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid Red 13
Acid Red 13 is an azo dye Azo dyes are organic compounds bearing the functional group R−N=N−R′, in which R and R′ are usually aryl and substituted aryl groups. They are a commercially important family of azo compounds, i.e. compounds containing the C-N=N ..... It is produced as a sodium salt. Solid samples appear dark red. References {{reflist Azo dyes Organic sodium salts Naphthalenesulfonates 2-Naphthols ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Azo Coupling
In organic chemistry, an azo coupling is an organic reaction between a diazonium compound () and another aromatic compound that produces an azo compound (). In this electrophilic aromatic substitution reaction, the aryldiazonium cation is the electrophile and the activated arene is a nucleophile. In most cases, including the examples below, the diazonium compound is also aromatic. Diazotization The process of conversion of primary aromatic amines into its diazonium salt is called diazotization. Diazonium salts are important synthetic intermediates that can undergo coupling reactions to form azo dyes and electrophilic substitution reactions to introduce functional groups. Uses of the reaction Aromatic azo compounds tend to be brightly colored due to the extended conjugated systems. Many are used as dyes (see azo dye). Important azo dyes include methyl red and pigment red 170. Azo printing exploits this reaction as well. Azo coupling is also used to produce prontosil and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Naphthionic Acid
Naphthionic acid is an organic compound with the formula C10H6(SO3H)(NH2). It is one of several aminonaphthalenesulfonic acids, derivatives of naphthalene containing both amine and sulfonic acid functional groups. It is a white solid, although commercial samples can appear gray.''4-Amino-1-naphthalenesulfonic acid;'' MSDS No. 250619; Sigma–Aldrich Chemie GmbH: Steinheim, 29 Dec 2011. It is used in the synthesis of azo dyes such as Rocceline (a. k. a. Solid Red A), during which the amino group of the acid (in the form of a salt) is diazotated and then coupled with, in the case mentioned, β-naphthol. It is prepared by treating 1-aminonaphthalene with sulfuric acid Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid ( Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular formu ....Gerald Booth "Naphthalene Derivatives" in Ullmann's Encyclopedia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystallization
Crystallization is the process by which solid forms, where the atoms or molecules are highly organized into a structure known as a crystal. Some ways by which crystals form are precipitating from a solution, freezing, or more rarely deposition directly from a gas. Attributes of the resulting crystal depend largely on factors such as temperature, air pressure, and in the case of liquid crystals, time of fluid evaporation. Crystallization occurs in two major steps. The first is nucleation, the appearance of a crystalline phase from either a supercooled liquid or a supersaturated solvent. The second step is known as crystal growth, which is the increase in the size of particles and leads to a crystal state. An important feature of this step is that loose particles form layers at the crystal's surface and lodge themselves into open inconsistencies such as pores, cracks, etc. The majority of minerals and organic molecules crystallize easily, and the resulting crystals are g ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vitrification
Vitrification (from Latin ''vitreum'', "glass" via French ''vitrifier'') is the full or partial transformation of a substance into a glass, that is to say, a non-crystalline amorphous solid. Glasses differ from liquids structurally and glasses possess a higher degree of connectivity with the same Hausdorff dimensionality of bonds as crystals: dimH = 3. In the production of ceramics, vitrification is responsible for its impermeability to water. Vitrification is usually achieved by heating materials until they liquidize, then cooling the liquid, often rapidly, so that it passes through the glass transition to form a glassy solid. Certain chemical reactions also result in glasses. In terms of chemistry, vitrification is characteristic for amorphous materials or disordered systems and occurs when bonding between elementary particles (atoms, molecules, forming blocks) becomes higher than a certain threshold value. Thermal fluctuations break the bonds; therefore, the low ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
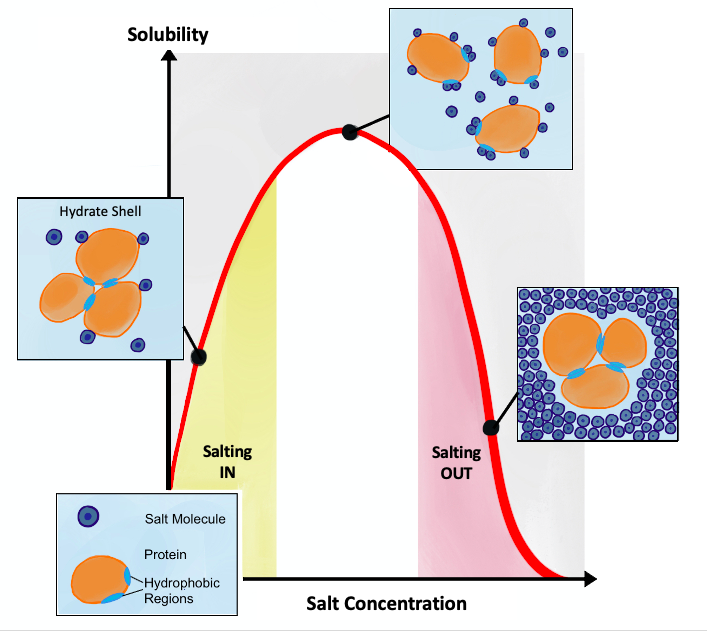
Salting Out
Salting out (also known as salt-induced precipitation, salt fractionation, anti-solvent crystallization, precipitation crystallization, or drowning out) is a purification technique that utilizes the reduced solubility of certain molecules in a solution of very high ionic strength. Salting out is typically used to precipitate large biomolecules, such as proteins or DNA. Because the salt concentration needed for a given protein to precipitate out of the solution differs from protein to protein, a specific salt concentration can be used to precipitate a target protein. This process is also used to concentrate dilute solutions of proteins. Dialysis can be used to remove the salt if needed. Principle Salt compounds dissociate in aqueous solutions. This property is exploited in the process of salting out. When the salt concentration is increased, some of the water molecules are attracted by the salt ions, which decreases the number of water molecules available to interact with the c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Naphthol Red Preparation
Naphthol may refer to: * 1-Naphthol 1-Naphthol, or α-naphthol, is a fluorescent organic compound with the formula . It is a white solid. It is an isomer of 2-naphthol differing by the location of the hydroxyl group on the naphthalene ring. The naphthols are naphthalene homologues ... * 2-Naphthol {{Short pages monitor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |