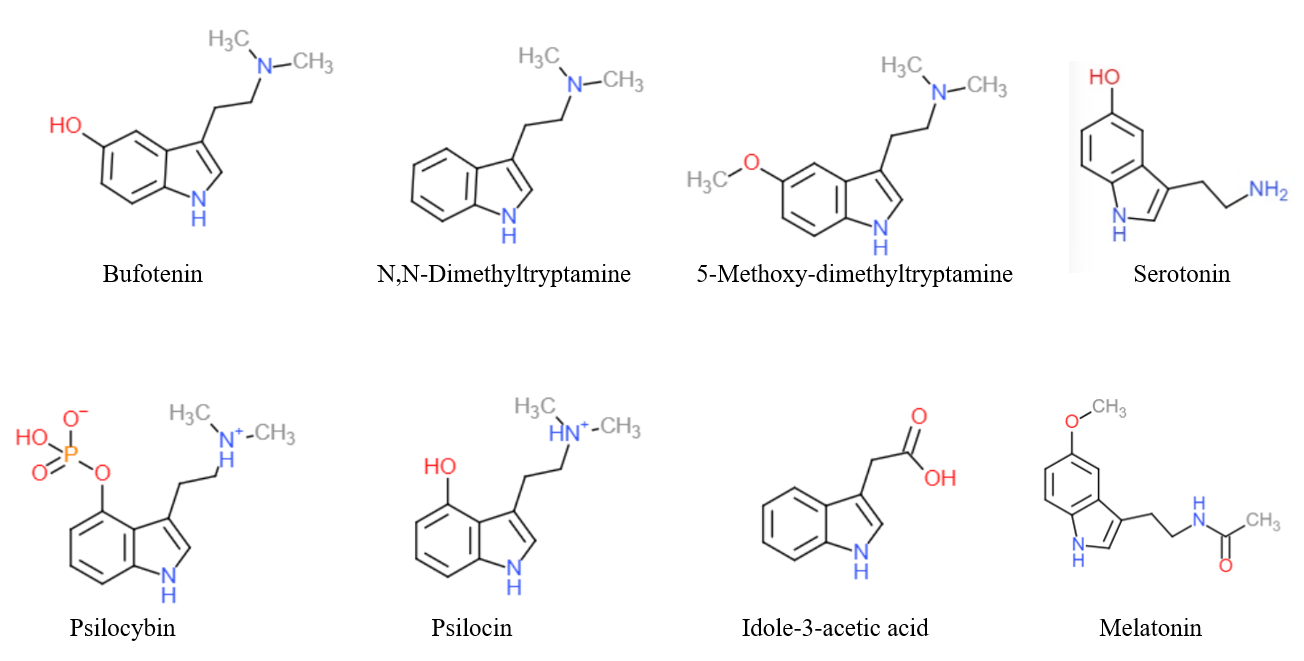
|
4-MeO-DMT
4-MeO-DMT (4-methoxy-''N'',''N''-dimethyltryptamine) is a tryptamine derivative which has some central activity in animal tests similar to that of related psychedelic tryptamine drugs, although with significantly lower potency than either 5-MeO-DMT or 4-hydroxy-DMT (psilocin).Nichols DE, Glennon RA. Medicinal Chemistry and Structure-Activity Relationships of Hallucinogens, in Hallucinogens, Neurochemical, Behavioral, and Clinical Perspectives (Raven Press 1984), pp 95-142. See also * 4-Acetoxy-DMT *Alexander Shulgin *4-MeO-MiPT 4-MeO-MiPT, or 4-methoxy-''N''-methyl-''N''-isopropyltryptamine, is a lesser-known Psychedelics, dissociatives and deliriants, psychedelic drug. It is the 4-methoxy analog (chemistry), analog of MiPT. 4-MeO-MiPT was first synthesized by Alexander ... References {{Tryptamines Serotonin receptor agonists Tryptamines Indole ethers at the benzene ring Methoxy compounds Dimethylamino compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tryptamines
Substituted tryptamines, or serotonin analogues, are organic compounds which may be thought of as being derived from tryptamine itself. The molecular structures of all tryptamines contain an indole ring, joined to an amino (NH2) group via an ethyl (−CH2–CH2−) sidechain. In substituted tryptamines, the indole ring, sidechain, and/or amino group are modified by substituting another group for one of the hydrogen (H) atoms. Well-known tryptamines include serotonin, an important neurotransmitter, and melatonin, a hormone involved in regulating the sleep-wake cycle. Tryptamine alkaloids are found in fungi, plants and animals; and sometimes used by humans for the neurological or psychotropic effects of the substance. Prominent examples of tryptamine alkaloids include psilocybin (from "psilocybin mushrooms") and DMT. In South America, dimethyltryptamine is obtained from numerous plant sources, like chacruna, and it is often used in ayahuasca brews. Many synthetic tryptamines ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-Acetoxy-DMT
''O''-Acetylpsilocin (also known as psilacetin, 4-acetoxy-DMT, 4-AcO-DMT, or synthetic shrooms) is a semi-synthetic psychoactive drug that has been suggested by David Nichols to be a potentially useful alternative to psilocybin for pharmacological studies, as they are both believed to be prodrugs of psilocin. However, some users report that ''O''-acetylpsilocin's subjective effects differ from those of psilocybin and psilocin. Additionally, some users prefer 4-AcO-DMT to natural psilocybin mushrooms due to feeling fewer adverse side effects such as nausea and heavy body load, which are more frequently reported in experiences involving natural mushrooms. It is the acetylated form of the psilocybin mushroom alkaloid psilocin and is a lower homolog of 4-AcO-MET, 4-AcO-DET, 4-AcO-MiPT and 4-AcO-DiPT. History ''O''-Acetylpsilocin (psilacetin) and several other esters of psilocin were patented on January 16, 1963 by Sandoz Ltd via Albert Hofmann & Franz Troxler. Despite ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
5-MeO-DMT
5-MeO-DMT (5-methoxy-''N'',''N''-dimethyltryptamine) or O-methyl-bufotenin is a psychedelic of the tryptamine class. It is found in a wide variety of plant species, and also is secreted by the glands of at least one toad species, the Colorado River toad. Like its close relatives DMT and bufotenin (5-HO-DMT), it has been used as an entheogen in South America. Slang terms include Five-methoxy, The power, and Toad venom. Chemistry 5-MeO-DMT was first synthesized in 1936, and in 1959 it was isolated as one of the psychoactive ingredients of '' Anadenanthera peregrina'' seeds used in preparing Yopo snuff. It was once believed to be a major component of the psychoactive effects of the snuff, although this has recently been shown to be unlikely, due to the limited or sometimes even non-existent quantity contained within the seeds, which instead achieve their psychoactivity from the ''O''-demethylated metabolite of 5-MeO-DMT, bufotenin. It is metabolized mainly by CYP2D6. Effects D ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-MeO-MiPT
4-MeO-MiPT, or 4-methoxy-''N''-methyl-''N''-isopropyltryptamine, is a lesser-known Psychedelics, dissociatives and deliriants, psychedelic drug. It is the 4-methoxy analog (chemistry), analog of MiPT. 4-MeO-MiPT was first synthesized by Alexander Shulgin and is mentioned in his book ''TiHKAL'' (''Tryptamines i Have Known And Loved''). Subsequent testing by Shulgin on human test subjects showed the effective dose (pharmacology), effective dose as 20-30 Milligram, mg (or 0.4 mg per Kg body weight of subject); the onset time between ingestion and the first noticeable effects was 45-60 min, with sensations lasting between 2-2.5 hours. The sensation were significantly milder than those of 4-HO-MiPT, with 4-MeO-MiPT producing erotic-enhancing effects, and few of the visuals common with tryptamines. Very little data exists about the pharmacological properties, metabolism, and toxicity of 4-MeO-MiPT. See also * 4-MeO-DMT * Tryptamine * Psychedelics, dissociatives and deliriants E ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tryptamine
Tryptamine is an indolamine metabolite of the essential amino acid, tryptophan. The chemical structure is defined by an indole ─ a fused benzene and pyrrole ring, and a 2-aminoethyl group at the second carbon (third aromatic atom, with the first one being the heterocyclic nitrogen). The structure of tryptamine is a shared feature of certain aminergic neuromodulators including melatonin, serotonin, bufotenin and psychedelic derivatives such as dimethyltryptamine (DMT), psilocybin, psilocin and others. Tryptamine has been shown to activate trace amine-associated receptors expressed in the mammalian brain, and regulates the activity of dopaminergic, serotonergic and glutamatergic systems. In the human gut, symbiotic bacteria convert dietary tryptophan to tryptamine, which activates 5-HT4 receptors and regulates gastrointestinal motility. Multiple tryptamine-derived drugs have been developed to treat migraines, while trace amine-associated receptors are being explored as a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Derivative
In chemistry, a derivative is a compound that is derived from a similar compound by a chemical reaction. In the past, derivative also meant a compound that ''can be imagined to'' arise from another compound, if one atom or group of atoms is replaced with another atom or group of atoms, but modern chemical language now uses the term structural analog for this meaning, thus eliminating ambiguity. The term "structural analogue" is common in organic chemistry. In biochemistry, the word is used for compounds that at least theoretically can be formed from the precursor compound. Chemical derivatives may be used to facilitate analysis. For example, melting point (MP) analysis can assist in identification of many organic compounds. A crystalline derivative may be prepared, such as a semicarbazone or 2,4-dinitrophenylhydrazone (derived from aldehydes or ketones), as a simple way of verifying the identity of the original compound, assuming that a table of derivative MP values is available ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Central Nervous System
The central nervous system (CNS) is the part of the nervous system consisting primarily of the brain and spinal cord. The CNS is so named because the brain integrates the received information and coordinates and influences the activity of all parts of the bodies of bilaterally symmetric and triploblastic animals—that is, all multicellular animals except sponges and diploblasts. It is a structure composed of nervous tissue positioned along the rostral (nose end) to caudal (tail end) axis of the body and may have an enlarged section at the rostral end which is a brain. Only arthropods, cephalopods and vertebrates have a true brain (precursor structures exist in onychophorans, gastropods and lancelets). The rest of this article exclusively discusses the vertebrate central nervous system, which is radically distinct from all other animals. Overview In vertebrates, the brain and spinal cord are both enclosed in the meninges. The meninges provide a barrier to chemicals dissolv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Psychedelic Drug
Psychedelics are a subclass of hallucinogenic drugs whose primary effect is to trigger non-ordinary states of consciousness (known as psychedelic experiences or "trips").Pollan, Michael (2018). ''How to Change Your Mind: What the New Science of Psychedelics Teaches Us About Consciousness, Dying, Addiction, Depression, and Transcendence'' Sometimes, they are called classic hallucinogens, serotonergic hallucinogens, or serotonergic psychedelics, and the term ''psychedelics'' is used more broadly to include all hallucinogens; this article uses the narrower definition of ''psychedelics''. Psychedelics cause specific psychological, visual, and auditory changes, and often a substantially altered state of consciousness.Leary, Timothy; Metzner, Ralph (1964). ''The Psychedelic Experience: A Manual Based on The Tibetan Book of the Dead'' Psychedelic states are often compared to meditative, psychodynamic or transcendental types of alterations of mind. The "classical" psychedelics, the psy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Psilocin
Psilocin (also known as 4-HO-DMT, 4-hydroxy DMT, psilocine, psilocyn, or psilotsin) is a substituted tryptamine alkaloid and a serotonergic psychedelic substance. It is present in most psychedelic mushrooms together with its phosphorylated counterpart psilocybin. Psilocin is a Schedule I drug under the Convention on Psychotropic Substances. Acting on the 5-HT2A receptors, psilocin modulates the production and reuptake of serotonin. The mind-altering effects of psilocin are highly variable and subjective and resemble those of LSD and DMT. Chemistry Psilocin and its phosphorylated cousin, psilocybin, were first isolated and named in 1958 by Swiss chemist Albert Hofmann. Hofmann obtained the chemicals from laboratory-grown specimens of the entheogenic mushroom ''Psilocybe mexicana''. Hofmann also succeeded in finding synthetic routes to these chemicals. Psilocin can be obtained by dephosphorylation of natural psilocybin under strongly acidic or under alkaline conditions (hyd ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alexander Shulgin
Alexander Theodore "Sasha" Shulgin (June 17, 1925 – June 2, 2014) was an American medicinal chemist, biochemist, organic chemist, pharmacologist, psychopharmacologist, and author. He is credited with introducing 3,4-methylenedioxymethamphetamine (MDMA, commonly known as "ecstasy") to psychologists in the late 1970s for psychopharmaceutical use and for the discovery, synthesis and personal bioassay of over 230 psychoactive compounds for their psychedelic and entactogenic potential. In 1991 and 1997, he and his wife Ann Shulgin compiled the books '' PiHKAL'' and ''TiHKAL'' (standing for ''Phenethylamines'' and ''Tryptamines I Have Known And Loved''), from notebooks that extensively described their work and personal experiences with these two classes of psychoactive drugs. Shulgin performed seminal work into the descriptive synthesis of many of these compounds. Some of Shulgin's noteworthy discoveries include compounds of the 2C* family (such as 2C-B) and compounds of t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Serotonin Receptor Agonists
Serotonin () or 5-hydroxytryptamine (5-HT) is a monoamine neurotransmitter. Its biological function is complex and multifaceted, modulating mood, cognition, reward, learning, memory, and numerous physiological processes such as vomiting and vasoconstriction. Approximately 90% of the serotonin that the body produces is in the intestinal tract. Biochemically, the indoleamine molecule derives from the amino acid tryptophan, via the (rate-limiting) hydroxylation of the 5 position on the ring (forming the intermediate 5-hydroxytryptophan), and then decarboxylation to produce serotonin. Serotonin is primarily found in the enteric nervous system located in the gastrointestinal tract (GI tract). However, it is also produced in the central nervous system (CNS), specifically in the raphe nuclei located in the brainstem, Merkel cells located in the skin, pulmonary neuroendocrine cells and taste receptor cells in the tongue. Additionally, serotonin is stored in blood platelets and is rel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Indole Ethers At The Benzene Ring
Indole is an aromatic heterocyclic organic compound with the formula C8 H7 N. It has a bicyclic structure, consisting of a six-membered benzene ring fused to a five-membered pyrrole ring. Indole is widely distributed in the natural environment and can be produced by a variety of bacteria. As an intercellular signal molecule, indole regulates various aspects of bacterial physiology, including spore formation, plasmid stability, resistance to drugs, biofilm formation, and virulence. The amino acid tryptophan is an indole derivative and the precursor of the neurotransmitter serotonin. General properties and occurrence Indole is a solid at room temperature. It occurs naturally in human feces and has an intense fecal odor. At very low concentrations, however, it has a flowery smell, and is a constituent of many perfumes. It also occurs in coal tar. The corresponding substituent is called indolyl. Indole undergoes electrophilic substitution, mainly at position 3 (see diagram i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |