|
1-Aminoethanol
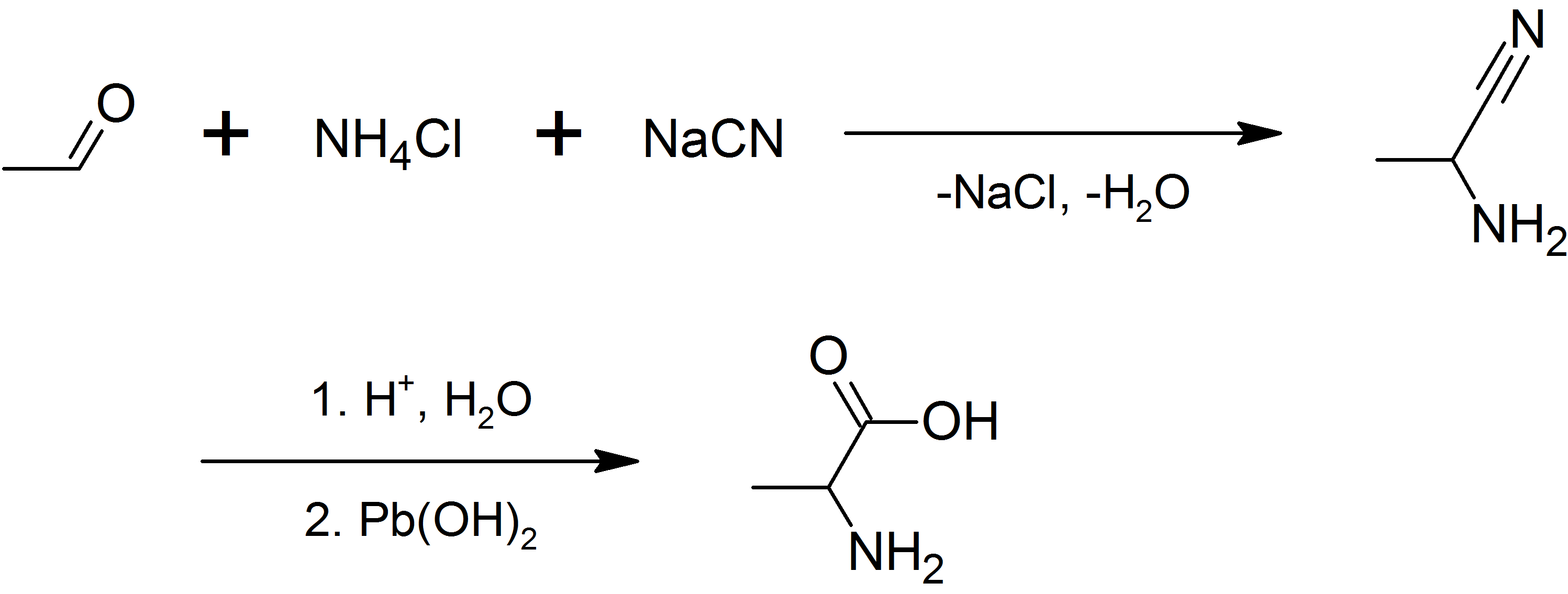
1-Aminoethanol is an organic compound with the formula CHCH(NH)OH. It is classified as an alkanolamine. Specifically, it is a structural isomer of 2-aminoethanol (ethanolamine). These two compounds differ in the position of the amino group. Since the central carbon atom in 1-aminoethanol has four different substituents, the compound has two stereoisomers. Unlike 2-aminoethanol, which is of considerable importance in commerce, 1-aminoethanol is not encountered as a pure material and is mainly of theoretical interest. 1-Aminoethanol exists in a solution of acetaldehyde and aqueous ammonia. 1-Aminoethanol is suggested as intermediate in Strecker reaction of alanine synthesis. 1-Aminoethanol was first prepared in 1833 by the German chemist Johann Wolfgang Döbereiner; its empirical formula was first determined by the German chemist Justus von Liebig in 1835. The structure of 1-aminoethanol remained unproven until 1877, when the German-Italian chemist Robert Schiff Robert Schi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkanolamine
In organic chemistry, alkanolamines are organic compounds that contain both hydroxyl () and amino (, , and ) functional groups on an alkane backbone. The term alkanolamine is a broad class term that is sometimes used as a subclassification. Methanolamine.svg, methanolamine, an intermediate in the reaction of ammonia with formaldehyde Ethanolamine.png, Ethanolamine 2-amino-2-methyl-1-propanol.svg, 2-amino-2-methyl-1-propanol is a precursor to oxazolines valinol.svg, valinol is derived from the amino acid valine Sphingosine structure.svg, Sphingosine is a component of some cell membrane. 1-Aminoalcohols 1-Aminoalcohols are better known as hemiaminals. Methanolamine is the simplest member. 2-Aminoalcohols Key members: ethanolamine, dimethylethanolamine, ''N''-methylethanolamine, Aminomethyl propanol Two popular drugs, often called alkanolamine beta blockers, are members of this structural class: propranolol, pindolol. Isoetarine is yet another medicinally useful derivative o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2-Aminoethanol
Ethanolamine (2-aminoethanol, monoethanolamine, ETA, or MEA) is an organic chemical compound with the formula or . The molecule is bifunctional, containing both a primary amine and a primary alcohol. Ethanolamine is a colorless, viscous liquid with an odor reminiscent of ammonia.. ETA molecules are a component in the formation of cellular membranes and are thus a molecular building block for life. It was thought to exist only on Earth and on certain asteroids, but in 2021 evidence was found that ETA molecules exist in interstellar space. Derivatives of ethanolamine are widespread in nature; e.g., lipids, as precursor of a variety of ''N''-acylethanolamines (NAEs), that modulate several animal and plant physiological processes such as seed germination, plant–pathogen interactions, chloroplast development and flowering, as well as precursor, combined with arachidonic acid 20: 4, ω-6), to form the endocannabinoid anandamide (AEA: ; 20:4, ω-6). The ethanolamines comprise a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stereoisomers
In stereochemistry, stereoisomerism, or spatial isomerism, is a form of isomerism in which molecules have the same molecular formula and sequence of bonded atoms (constitution), but differ in the three-dimensional orientations of their atoms in space. This contrasts with structural isomers, which share the same molecular formula, but the bond connections or their order differs. By definition, molecules that are stereoisomers of each other represent the same structural isomer. Enantiomers Enantiomers, also known as optical isomers, are two stereoisomers that are related to each other by a reflection: they are mirror images of each other that are non-superposable. Human hands are a macroscopic analog of this. Every stereogenic center in one has the opposite configuration in the other. Two compounds that are enantiomers of each other have the same physical properties, except for the direction in which they rotate polarized light and how they interact with different optical is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Strecker Reaction
The Strecker amino acid synthesis, also known simply as the Strecker synthesis, is a method for the synthesis of amino acids by the reaction of an aldehyde with ammonia in the presence of potassium cyanide. The condensation reaction yields an α-aminonitrile, which is subsequently hydrolyzed to give the desired amino acid. The method is used commercially for the production of racemic methionine from methional. While usage of ammonium salts gives unsubstituted amino acids, primary and secondary amines also give substituted amino acids. Likewise, the usage of ketones, instead of aldehydes, gives α,α-disubstituted amino acids. Reaction mechanism In the first part of the reaction, the carbonyl group, carbonyl oxygen of an aldehyde is protonated, followed by a nucleophilic addition, nucleophilic attack of ammonia to the carbonyl carbon. After subsequent proton exchange, water is cleaved from the iminium ion intermediate. A cyanide ion then attacks the iminium carbon yielding an am ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alanine
Alanine (symbol Ala or A), or α-alanine, is an α-amino acid that is used in the biosynthesis of proteins. It contains an amine group and a carboxylic acid group, both attached to the central carbon atom which also carries a methyl group side chain. Consequently, its IUPAC systematic name is 2-aminopropanoic acid, and it is classified as a nonpolar, aliphatic α-amino acid. Under biological conditions, it exists in its zwitterionic form with its amine group protonated (as −NH3+) and its carboxyl group deprotonated (as −CO2−). It is non-essential to humans as it can be synthesised metabolically and does not need to be present in the diet. It is encoded by all codons starting with GC (GCU, GCC, GCA, and GCG). The L-isomer of alanine (left-handed) is the one that is incorporated into proteins. L-alanine is second only to leucine in rate of occurrence, accounting for 7.8% of the primary structure in a sample of 1,150 proteins. The right-handed form, D-alanine, occurs in p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beilstein Journal Of Organic Chemistry
The ''Beilstein Journal of Organic Chemistry'' is a peer-reviewed open-access scientific journal established in 2005. It is published by the Beilstein Institute for the Advancement of Chemical Sciences, a German non-profit foundation. The editor-in-chief is Peter Seeberger (Max Planck Institute of Colloids and Interfaces). According to the ''Journal Citation Reports'', the journal has a 2020 impact factor The impact factor (IF) or journal impact factor (JIF) of an academic journal is a scientometric index calculated by Clarivate that reflects the yearly mean number of citations of articles published in the last two years in a given journal, as i ... of 2.88. Scientific videos are available for selected articles of the journal. References External links * Organic chemistry journals Open access journals Creative Commons Attribution-licensed journals Publications established in 2005 English-language journals BioMed Central academic journals {{chem-journal-stu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Johann Wolfgang Döbereiner
Johann Wolfgang Döbereiner (13 December 1780 – 24 March 1849) was a German chemist who is best known for work that foreshadowed the periodic law for the chemical elements, and for inventing the first lighter, which was known as the Döbereiner's lamp. He became a professor of chemistry and pharmacy at the University of Jena. Life and work As a coachman's son, Döbereiner had little opportunity for formal schooling. Thus, he was apprenticed to an apothecary, and began to read widely and to attend science lectures. He eventually became a professor at the University of Jena in 1810 and also studied chemistry at Strasbourg. In work published in 1829, Döbereiner reported trends in certain properties of selected groups of elements. For example, the average of the atomic masses of lithium and potassium was close to the atomic mass of sodium. A similar pattern was found with calcium, strontium, and barium; with sulfur, selenium, tellurium; and with chlorine, bromine, and iodine. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Justus Von Liebig
Justus Freiherr von Liebig (12 May 1803 – 20 April 1873) was a German scientist who made major contributions to agricultural and biological chemistry, and is considered one of the principal founders of organic chemistry. As a professor at the University of Giessen, he devised the modern laboratory-oriented teaching method, and for such innovations, he is regarded as one of the greatest chemistry teachers of all time. He has been described as the "father of the fertilizer industry" for his emphasis on nitrogen and trace minerals as essential plant nutrients, and his formulation of the law of the minimum, which described how plant growth relied on the scarcest nutrient resource, rather than the total amount of resources available. He also developed a manufacturing process for beef extracts, and with his consent a company, called Liebig Extract of Meat Company, was founded to exploit the concept; it later introduced the Oxo brand beef bouillon cube. He popularized an earlier ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Robert Schiff
Robert Schiff (July 25, 1854 in Frankfurt am Main, Germany – 1940 in Massa, Tuscany, Massa, Italy) was a German-born, Italian chemist. He was the son of physiologist Moritz Schiff and Claudia Trier. He successively was a student at the University of Heidelberg, then at the University of Zürich, where he obtained a doctorate in 1876. He then became an assistant to Stanislao Cannizzaro at Sapienza University of Rome, and in 1879 was appointed professor of chemistry at the University of Modena. In 1892 he relocated as a professor to the University of Pisa.The Jewish Encyclopedia: A De ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Primary Alcohols
A primary alcohol is an alcohol in which the hydroxy group In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy ... is bonded to a primary carbon atom. It can also be defined as a molecule containing a “–CH2OH” group. In contrast, a secondary alcohol has a formula “–CHROH” and a tertiary alcohol has a formula “–CR2OH”, where “R” indicates a carbon-containing group. Examples of primary alcohols include ethanol and n-Butanol, 1-butanol. Methanol is also generally regarded as a primary alcohol, including the 1911 edition of the Encyclopædia Britannica,. See also * Alcohol (chemistry), Alcohol (especially Nomenclature section for discussion on Secondary and Tertiary alcohols.) * Oxidation of primary alcohols to carboxylic acids References Primary alcohols, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |