|
(benzylidene)iron Tricarbonyl
(Benzylideneacetone)iron tricarbonyl is the organoiron compound with the formula (CHCH=CHC(O)CH)Fe(CO). It is a reagent for transferring the Fe(CO) unit. This red-colored compound is commonly abbreviated (bda)Fe(CO). Structure and bonding (bda)Fe(CO) is an example of a complex of an η-ketone. It is a piano stool complex. The compound is characterized by IR bands at 2065, 2005, and 1985 cm (cyclohexane solution), the three bands being indicative of the low symmetry of the complex, which is chiral. Synthesis, reactions, related reagents It is prepared by the reaction of Fe(CO) with benzylideneacetone. (bda)Fe(CO) sometimes reacts with Lewis bases to give adducts without displacement of the bda. The reagents of the type (bda)Fe(CO)(PR) function as sources of "Fe(CO)(PR)" (R = aryl, etc.). Other sources of Fe(CO) are Fe(CO) and Fe(CO)(cyclooctene). The latter is highly reactive and thermally sensitive. Imine derivatives of cinnamaldehyde, e.g. CHCH=CHC(H)=NCH, also form r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organoiron Compound
Organoiron chemistry is the chemistry of iron compounds containing a carbon-to-iron chemical bond. Organoiron compounds are relevant in organic synthesis as reagents such as iron pentacarbonyl, diiron nonacarbonyl and disodium tetracarbonylferrate. While iron adopts oxidation states from Fe(−II) through to Fe(VII), Fe(IV) is the highest established oxidation state for organoiron species. Although iron is generally less active in many catalytic applications, it is less expensive and "Green chemistry, greener" than other metals. Organoiron compounds feature a wide range of ligands that support the Fe-C bond; as with other organometals, these supporting ligands prominently include phosphines, carbon monoxide, and cyclopentadienyl, but HSAB theory, hard ligands such as amines are employed as well. Iron(0) and more reduced states Carbonyl complexes Important metal carbonyls, iron carbonyls are the three neutral binary carbonyls, iron pentacarbonyl, diiron nonacarbonyl, and triiron d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lewis Acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any species that has a filled orbital containing an electron pair which is not involved in bonding but may form a dative bond with a Lewis acid to form a Lewis adduct. For example, NH3 is a Lewis base, because it can donate its lone pair of electrons. Trimethylborane (Me3B) is a Lewis acid as it is capable of accepting a lone pair. In a Lewis adduct, the Lewis acid and base share an electron pair furnished by the Lewis base, forming a dative bond. In the context of a specific chemical reaction between NH3 and Me3B, a lone pair from NH3 will form a dative bond with the empty orbital of Me3B to form an adduct NH3•BMe3. The terminology refers to the contributions of Gilbert N. Lewis. From p. 142: "We are inclined to think of substances as po ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonyl Complexes
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a carbonyl group is often referred to as a carbonyl compound. The term carbonyl can also refer to carbon monoxide as a ligand in an inorganic or organometallic complex (a metal carbonyl, e.g. nickel carbonyl). The remainder of this article concerns itself with the organic chemistry definition of carbonyl, where carbon and oxygen share a double bond. Carbonyl compounds In organic chemistry, a carbonyl group characterizes the following types of compounds: Other organic carbonyls are urea and the carbamates, the derivatives of acyl chlorides chloroformates and phosgene, carbonate esters, thioesters, lactones, lactams, hydroxamates, and isocyanates. Examples of inorganic carbonyl compounds are carbon dioxide and carbonyl sulfide. A sp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organoiron Compounds
Organoiron chemistry is the chemistry of iron compounds containing a carbon-to-iron chemical bond. Organoiron compounds are relevant in organic synthesis as reagents such as iron pentacarbonyl, diiron nonacarbonyl and disodium tetracarbonylferrate. While iron adopts oxidation states from Fe(−II) through to Fe(VII), Fe(IV) is the highest established oxidation state for organoiron species. Although iron is generally less active in many catalytic applications, it is less expensive and " greener" than other metals. Organoiron compounds feature a wide range of ligands that support the Fe-C bond; as with other organometals, these supporting ligands prominently include phosphines, carbon monoxide, and cyclopentadienyl, but hard ligands such as amines are employed as well. Iron(0) and more reduced states Carbonyl complexes Important iron carbonyls are the three neutral binary carbonyls, iron pentacarbonyl, diiron nonacarbonyl, and triiron dodecacarbonyl. One or more carbonyl ligands ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organometallics
''Organometallics'' is a biweekly journal published by the American Chemical Society. Its area of focus is organometallic and organometalloid chemistry. This peer-reviewed journal has an impact factor of 3.837 as reported by the 2021 Journal Citation Reports by Thomson Reuters. Since 2015 Paul Chirik is the editor-in-chief of ''Organometallics''. He is an American chemist and the Edwards S. Sanford Professor of Chemistry at Princeton University, and associate director for external partnerships of the Andlinger Center for Energy and the Environment. He writes about the catalysis of hydrocarbons. Past editors-in-chief are Dietmar Seyferth and John Gladysz. Retrieved on 2014-07-30. This journal is indexed in |
Adduct
An adduct (from the Latin ''adductus'', "drawn toward" alternatively, a contraction of "addition product") is a product of a direct addition of two or more distinct molecules, resulting in a single reaction product containing all atoms of all components. The resultant is considered a distinct molecular species. Examples include the addition of sodium bisulfite to an aldehyde to give a sulfonate. It can just be considered as a single product resulting from the direct combination of different molecules which comprises all the reactant molecules' atoms. Adducts often form between Lewis acids and Lewis bases. A good example is the formation of adducts between the Lewis acid borane and the oxygen atom in the Lewis bases, tetrahydrofuran (THF): BH3·O(CH2)4 or diethyl ether: BH3·O(CH3CH2)2. Many Lewis acids and Lewis bases reacting in the gas phase or in non-aqueous solvents to form adducts have been examined in the ECW model. Trimethylboron, trimethyltin chloride and bis(hexaflu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
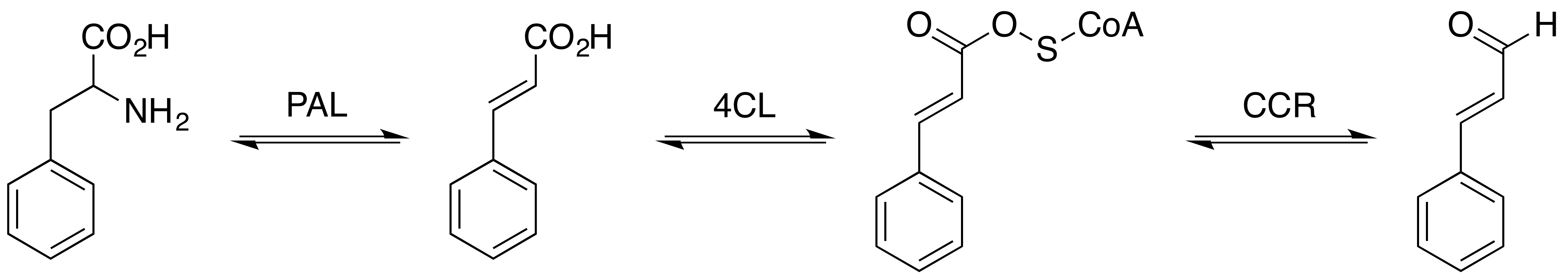
Cinnamaldehyde
Cinnamaldehyde is an organic compound with the formula(C9H8O) C6H5CH=CHCHO. Occurring naturally as predominantly the ''trans'' (''E'') isomer, it gives cinnamon its flavor and odor. It is a phenylpropanoid that is naturally synthesized by the shikimate pathway. This pale yellow, viscous liquid occurs in the bark of cinnamon trees and other species of the genus ''Cinnamomum''. The essential oil of cinnamon bark is about 90% cinnamaldehyde. Cinnamaldehyde decomposes to styrene because of oxidation as a result of bad storage or transport conditions. Styrene especially forms in high humidity and high temperatures. This is the reason why cinnamon contains small amounts of styrene. Structure and synthesis Cinnamaldehyde was isolated from cinnamon essential oil in 1834 by Jean-Baptiste Dumas and Eugène-Melchior Péligot and synthesized in the laboratory by the Italian chemist Luigi Chiozza in 1854. The natural product is ''trans''-cinnamaldehyde. The molecule consists of a benzene rin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Imine
In organic chemistry, an imine ( or ) is a functional group or organic compound containing a carbon–nitrogen double bond (). The nitrogen atom can be attached to a hydrogen or an organic group (R). The carbon atom has two additional single bonds. Imines are common in synthetic and naturally occurring compounds and they participate in many reactions. Structure For ketimines and aldimines, respectively, the five core atoms (C2C=NX and C(H)C=NX, X = H or C) are coplanar. Planarity results from the sp2-hybridization of the mutually double-bonded carbon and the nitrogen atoms. The C=N distance is 1.29-1.31 Å for nonconjugated imines and 1.35 Å for conjugated imines. By contrast, C-N distances in amines and nitriles are 1.47 and 1.16 Å, respectively. Rotation about the C=N bond is slow. Using NMR spectroscopy, both E- and Z-isomers of aldimines have been detected. Owing to steric effects, the E isomer is favored. Nomenclature and classification The term "imine" was coine ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclooctene
Cyclooctene is the cycloalkene with a formula . Its molecule has a ring of 8 carbon atoms, connected by seven single bonds and one double bond. Cyclooctene is notable because it is the smallest cycloalkene that can exist stably as either the ''cis'' or ''trans'' stereoisomer, with ''cis''-cyclooctene being the most common. Theoretical analysis implies a total of 16 conformational and configurational isomers, all chiral, forming 8 enantiomeric In chemistry, an enantiomer ( /ɪˈnænti.əmər, ɛ-, -oʊ-/ ''ih-NAN-tee-ə-mər''; from Ancient Greek ἐνάντιος ''(enántios)'' 'opposite', and μέρος ''(méros)'' 'part') – also called optical isomer, antipode, or optical ant ... pairs. The ''cis'' isomer can adopt various conformations, the most stable one being shaped like a ribbon. The most stable conformation of ''trans''-cyclooctene is shaped like the 8-carbon equivalent of the chair conformation of cyclohexane. Longer cycloalkene rings such as the ten-c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzylideneacetone
Benzylideneacetone is the organic compound described by the formula C6H5CH=CHC(O)CH3. Although both ''cis''- and ''trans''-isomers are possible for the α,β-unsaturated ketone, only the trans isomer is observed. Its original preparation demonstrated the scope of condensation reactions to construct new, complex organic compounds. Benzylideneacetone is used as a flavouring ingredient in food and perfumes. Preparation Benzylideneacetone can be efficiently prepared by the base-induced condensation of acetone and benzaldehyde: : However, the benzylideneacetone formed via this reaction can undergo another Claisen-Schmidt condensation with another molecule of benzaldehyde to form dibenzylideneacetone. Because relatively weak bases such as NaOH make very little of the enolate ion at equilibrium, there is still a lot of unreacted base left in the reaction mixture, which can go on and remove protons from the alpha carbon of benzylideneacetone, allowing it to undergo another Claisen- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Encyclopedia Of Reagents For Organic Synthesis
The ''Encyclopedia of Reagents for Organic Synthesis'' is published in print and online by John Wiley & Sons Ltd. The online version is also known as e-EROS. The encyclopedia contains a description of the use of reagents used in organic chemistry. The eight-volume print version includes 3500 alphabetically arranged articles and the online version is regularly updated to include new reagents and catalyst Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...s. References External links *Print version Encyclopedias of science Chemistry books {{encyclopedia-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diiron Nonacarbonyl
Diiron nonacarbonyl is an organometallic compound with the formula Fe2(CO)9. This metal carbonyl is an important reagent in organometallic chemistry and of occasional use in organic synthesis. It is a more reactive source of Fe(0) than Fe(CO)5. This micaceous orange solid is virtually insoluble in all common solvents. Synthesis and structure Following the original method, photolysis of an acetic acid solution of Fe(CO)5 produces Fe2(CO)9 in good yield:King, R. B. Organometallic Syntheses. Volume 1 Transition-Metal Compounds; Academic Press: New York, 1965. . :2 Fe(CO)5 → Fe2(CO)9 + CO Fe2(CO)9 consists of a pair of Fe(CO)3 centers linked by three bridging CO ligands. Although older textbooks show an Fe-Fe bond consistent with the 18 electron rule (8 valence electrons from Fe, two each from the terminal carbonyls, one each from the bridging carbonyls and one from the other Fe atom in the metal-metal bond), theoretical analyses have consistently indicated the absence of a d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fe(CO)3-Crystals.jpg)


iron-tricarbonyl-3D-balls.png)