Silicon Hyperconjugation on:
[Wikipedia]
[Google]
[Amazon]
Negative hyperconjugation is a theorized phenomenon in
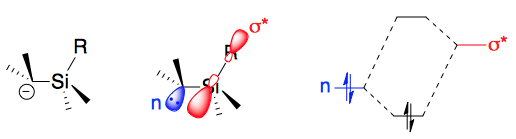 The silicon α and β effects arise because 3rd period heteroatoms can stabilize adjacent carbanions charges via ( negative) hyperconjugation.
In the α effect, reactions that develop negative charge adjacent to the silicon, such as metalations, exhibit accelerated rates. The C–M σ orbital partially overlaps the C–Si σ* anti-bonding orbital, which stabilizes the C–M bond. More generally, (i.e. even for "naked" carbanions) the Si σ* orbitals help stabilize the electrons on the α carbon.
In the β effect, reactions that develop positive charge on carbon atoms β to the silicon accelerate. The C–Si σ orbital partially overlaps the with the C–X (leaving group) σ* orbital (2b):
The silicon α and β effects arise because 3rd period heteroatoms can stabilize adjacent carbanions charges via ( negative) hyperconjugation.
In the α effect, reactions that develop negative charge adjacent to the silicon, such as metalations, exhibit accelerated rates. The C–M σ orbital partially overlaps the C–Si σ* anti-bonding orbital, which stabilizes the C–M bond. More generally, (i.e. even for "naked" carbanions) the Si σ* orbitals help stabilize the electrons on the α carbon.
In the β effect, reactions that develop positive charge on carbon atoms β to the silicon accelerate. The C–Si σ orbital partially overlaps the with the C–X (leaving group) σ* orbital (2b):  This electron-density donation into the anti-bonding orbital weakens the C–X bond, decreasing the barrier to the cleavage indicated 3, and favoring formation of the
This electron-density donation into the anti-bonding orbital weakens the C–X bond, decreasing the barrier to the cleavage indicated 3, and favoring formation of the
 The silicon α‑effect described above is mainly focused on carbon. In fact, the most industrially-important silicon α‑effect instead occurs with
The silicon α‑effect described above is mainly focused on carbon. In fact, the most industrially-important silicon α‑effect instead occurs with
Zheorthocresol
an
NotEvans
(17 Jan 2022).
Answers to "Stabilisation of anions by silicon"
(Accessed 2022-11-29.) ''Chemistry Stack Exchange''. Stack Exchange. {{webarchive, url=https://web.archive.org/web/20221129050341/https://chemistry.stackexchange.com/questions/77376/stabilisation-of-anions-by-silicon, date=2022-11-29, ref={{harvid, Dealon, NotEvans, 2017
{{Cite book , url=https://onlinelibrary.wiley.com/doi/book/10.1002/9783527619535 , title=Metal-Catalyzed Cross-Coupling Reactions , date=2004-08-25 , publisher=Wiley , isbn=978-3-527-30518-6 , editor-last=de Meijere , editor-first=Armin , edition=1 , language=en , doi=10.1002/9783527619535 , editor-last2=Diederich , editor-first2=François
{{Cite journal , last1=Mitzel , first1=Norbert W. , last2=Losehand , first2=Udo , last3=Richardson , first3=Alan , date=1999-07-01 , title=Two Successive Steps of Hypercoordination at Tin: The Gas-Phase and Solid-State Structures of ( N,N- Dimethylaminoxy)trimethylstannane , url=https://pubs.acs.org/doi/10.1021/om990219q , journal=Organometallics , language=en , volume=18 , issue=14 , pages=2610–2614 , doi=10.1021/om990219q , issn=0276-7333
orthocresol
(Jul 6 2017).
Answer
to {{harvnb, Dealon, NotEvans, 2017. {{Cite journal , last1=Berkefeld , first1=André , last2=Guerra , first2=Célia Fonseca , last3=Bertermann , first3=Rüdiger , last4=Troegel , first4=Dennis , last5=Daiß , first5=Jürgen O. , last6=Stohrer , first6=Jürgen , last7=Bickelhaupt , first7=F. Matthias , last8=Tacke , first8=Reinhold , date=2014-06-09 , title=Silicon α-Effect: A Systematic Experimental and Computational Study of the Hydrolysis of C α - and C γ -Functionalized Alkoxytriorganylsilanes of the Formula Type ROSiMe 2 (CH 2 ) n X (R = Me, Et; n = 1, 3; X = Functional Group) , url=https://pubs.acs.org/doi/10.1021/om500073m , journal=Organometallics , language=en , volume=33 , issue=11 , pages=2721–2737 , doi=10.1021/om500073m , issn=0276-7333 Whitmore, Frank C.; Sommer, Leo H. (March 1946) * "Organo-silicon compounds II: Silicon analogs of neopentyl chloride and neopentyl iodide: The alpha silicon effect." {{doi, 10.1021/ja01207a036. {{issn, 0002-7863. {{PMID, 21015745. * "——— III: α- and β-chloroalkyl silanes and the unusual reactivity of the latter." {{doi, 10.1021/ja01207a037. '' Journal of the American Chemical Society'', volume 68 issue 3. pp. 481–487.
organosilicon compounds
Organosilicon compounds are organometallic compounds containing carbon–silicon bonds. Organosilicon chemistry is the corresponding science of their preparation and properties. Most organosilicon compounds are similar to the ordinary organic co ...
, in which hyperconjugation stabilizes or destabilizes certain accumulations of positive charge. The phenomenon explains corresponding peculiarities in the stereochemistry
Stereochemistry, a subdiscipline of chemistry, involves the study of the relative spatial arrangement of atoms that form the structure of molecules and their manipulation. The study of stereochemistry focuses on the relationships between stereois ...
and rate of hydrolysis.
Second-row elements generally stabilize adjacent carbanion
In organic chemistry, a carbanion is an anion in which carbon is trivalent (forms three bonds) and bears a formal negative charge (in at least one significant resonance form).
Formally, a carbanion is the conjugate base of a carbon acid:
:R3C ...
s more effectively than their first-row congeners; conversely they destabilize adjacent carbocation
A carbocation is an ion with a positively charged carbon atom. Among the simplest examples are the methenium , methanium and vinyl cations. Occasionally, carbocations that bear more than one positively charged carbon atom are also encountere ...
s, and these effects reverse one atom over. For phosphorus and later elements, these phenomena are easily ascribed to the element's greater electronegativity than carbon. However, Si has lower electronegativity than carbon, polarizing the electron density onto carbon.
The continued presence of second-row type stability in certain organosilicon compounds is known as the silicon α and β effects, after the corresponding locants. These stabilities occur because of a partial overlap between the C–Si σ orbital and the σ* antibonding orbital
In chemical bonding theory, an antibonding orbital is a type of molecular orbital that weakens the chemical bond between two atoms and helps to raise the energy of the molecule relative to the separated atoms. Such an orbital has one or more no ...
at the β position, lowering the ''SN'' reaction transition state's energy. This hyperconjugation requires an antiperiplanar
In organic chemistry, anti-periplanar, or antiperiplanar, describes the bond angle in a molecule. In this conformer, the dihedral angle of the bond and the bond is greater than +150° or less than −150° (Figures 1 and 2). Anti-periplanar i ...
relationship between the Si group and the leaving group to maximize orbital overlap.
Moreover, there is also another kind of silicon α effect, which is mainly about the hydrolysis on the silicon atom.
Experimental evidence
In 1946, Leo Sommer andFrank C. Whitmore
Frank Clifford Whitmore (October 1, 1887 – June 24, 1947), nicknamed "Rocky", was a prominent chemist who submitted significant evidence for the existence of carbocation mechanisms in organic chemistry.
He was born in 1887 in the town of North ...
reported that radically chlorinating liquid ethyltrichlorosilane gave an isomeric mixture with exhibited unexpected reactivity in aqueous base. All chlorides pendant to silicon hydrolyze, but the geminal chlorine on carbon failed to hydrolyze, and the vicinal chlorine eliminated to ethene: The same behavior appeared with ''n''-propyltrichlorosilane. The α and γ isomers resisted hydrolysis, but a hydroxyl group replaced the β chlorine: They concluded that silicon inhibits electrofugal activity at the α carbon.
The silicon effect also manifests in certain compound properties. Trimethylsilylmethylamine (Me3SiCH2NH2) is a stronger base ( conjugate pKa 10.96) than neopentylamine (conjugate pKa 10.21); trimethylsilylacetic acid (pKa 5.22) is a poorer acid than trimethylacetic acid
Pivalic acid is a carboxylic acid with a molecular formula of (CH3)3CCO2H. This colourless, odiferous organic compound is solid at room temperature. A common abbreviation for the pivalyl or pivaloyl group (''t''-BuC(O)) is Piv and for pivalic acid ...
(pKa 5.00).
In 1994, Yong and coworkers compared the free-energy effects of α- and β- Si(CH3)3 moieties on C–H homo- and heterolysis. They, too, concluded that the β silicon atom could stabilize carbocations and the α silicon destabilize carbocations.
Orbital structure
carbenium
A carbenium ion is a positive ion with the structure RR′R″C+, that is, a chemical species with a trivalent carbon that bears a +1 formal charge.
In older literature the name carbonium ion was used for this class, but now it refers exclusivel ...
4.
In silyl ethers
silyl ether
Silyl ethers are a group of chemical compounds which contain a silicon atom covalently bonded to an alkoxy group. The general structure is R1R2R3Si−O−R4 where R4 is an alkyl group or an aryl group. Silyl ethers are usually used as protecting g ...
s. Under hydrolysis condition, certain α-silane-terminated prepolymers crosslink 10-1000 times faster than the corresponding prepolymers produced from conventional Cγ-functionalized trialkoxypropylsilanes and dialkoxymethylpropylsilanes.
History
This silicon α-effect was first observed in the late 1960s by researchers at Bayer AG as an increase in reactivity at the silicon atom for hydrolysis and was used for cross-linking of α-silane-terminated prepolymers. For a long time after that, people attributed this reactivity as silicon α-effect. However, the real mechanism beneath it had been debated for many years after this discovery. Generally, this effect has been rationalized as an intramolecular donor-acceptor interaction between the lone pair of the organofunctional group (such as NR2, OC(O)R, N(H)COOMe) and the silicon atom. However, this hypothesis has been proved incorrect by Mitzel and coworkers and more experiments are needed to interpret this effect.Mechanism study
Reinhold and coworkers performed a systematical experiment to study the kinetics and mechanisms of hydrolysis of such compounds. They prepared a series of α-silanes and γ-silanes and tested their reactivity in different pH (acidic and basic regime), functional group X and the spacer between the silicon atom and the functional group X. In general, they find that under basic conditions, the rate of hydrolysis is mainly controlled by the electrophilicity of the silicon center and the rate of the hydrolysis of the γ-silanes is less influenced by the generally electronegative functional groups than α-silanes. More electronegative the functional groups are, the higher the rate of hydrolysis. However, under acidic conditions, the rate of hydrolysis depends on both the electrophilicity of the silicon center (determining the molecular reactivity) and the concentration of the (protonated) reactive species. Under acidic conditions, the nucleophile changes from OH- to H2O, so it involves the process of protonation and the atoms are protonated could be either silicon or the functional group X. As a result, the general trend in acidic solution is more complicated.References
{{reflist, 30em, refs= {{Cite journal , last1=Bausch , first1=M. J. , author2=Gong Yong , date=June 1994 , title=Effects of α- and β-silicon atoms on the free energies of C-H homolysis and heterolysis , url=https://pubs.acs.org/doi/abs/10.1021/ja00092a055 , journal=Journal of the American Chemical Society , language=en , volume=116 , issue=13 , pages=5963–5964 , doi=10.1021/ja00092a055 , issn=0002-7863 Colvin, E. (1981) ''Silicon in Organic Synthesis''. Butterworth: London. {{wikicitZhe
an
NotEvans
(17 Jan 2022).
Answers to "Stabilisation of anions by silicon"
(Accessed 2022-11-29.) ''Chemistry Stack Exchange''. Stack Exchange. {{webarchive, url=https://web.archive.org/web/20221129050341/https://chemistry.stackexchange.com/questions/77376/stabilisation-of-anions-by-silicon, date=2022-11-29, ref={{harvid, Dealon, NotEvans, 2017
(Jul 6 2017).
Answer
to {{harvnb, Dealon, NotEvans, 2017. {{Cite journal , last1=Berkefeld , first1=André , last2=Guerra , first2=Célia Fonseca , last3=Bertermann , first3=Rüdiger , last4=Troegel , first4=Dennis , last5=Daiß , first5=Jürgen O. , last6=Stohrer , first6=Jürgen , last7=Bickelhaupt , first7=F. Matthias , last8=Tacke , first8=Reinhold , date=2014-06-09 , title=Silicon α-Effect: A Systematic Experimental and Computational Study of the Hydrolysis of C α - and C γ -Functionalized Alkoxytriorganylsilanes of the Formula Type ROSiMe 2 (CH 2 ) n X (R = Me, Et; n = 1, 3; X = Functional Group) , url=https://pubs.acs.org/doi/10.1021/om500073m , journal=Organometallics , language=en , volume=33 , issue=11 , pages=2721–2737 , doi=10.1021/om500073m , issn=0276-7333 Whitmore, Frank C.; Sommer, Leo H. (March 1946) * "Organo-silicon compounds II: Silicon analogs of neopentyl chloride and neopentyl iodide: The alpha silicon effect." {{doi, 10.1021/ja01207a036. {{issn, 0002-7863. {{PMID, 21015745. * "——— III: α- and β-chloroalkyl silanes and the unusual reactivity of the latter." {{doi, 10.1021/ja01207a037. '' Journal of the American Chemical Society'', volume 68 issue 3. pp. 481–487.
Sommer, Leo H.; Dorfman, Edwin; Goldberg, Gershon M.; and Whitmore, Frank C. "The reactivity with alkali of chlorine-carbon bonds alpha, beta and gamma to silicon." ''Ibid'', pp. 488–489. {{doi, 10.1021/ja01207a038. {{issn, 0002-7863. {{PMID, 21015747.
Silicon Physical organic chemistry