Dioxolanes on:
[Wikipedia]
[Google]
[Amazon]
Dioxolane is a
 (+)-''cis''-Dioxolane is the
(+)-''cis''-Dioxolane is the
 NaBArF4 can also be used for deprotection of acetal or ketal-protected carbonyl compounds. For example, deprotection of 2-phenyl-1,3-dioxolane to
NaBArF4 can also be used for deprotection of acetal or ketal-protected carbonyl compounds. For example, deprotection of 2-phenyl-1,3-dioxolane to -> ce\text] PhCHO + ethylene glycol, HOCH2CH2OH
 A similar approach is used in the total synthesis of sporol, with the dioxolane ring later expanded to a dioxane system.
A similar approach is used in the total synthesis of sporol, with the dioxolane ring later expanded to a dioxane system.
environmental and toxicological data
{{Muscarinic acetylcholine receptor modulators Muscarinic agonists Solvents Protecting groups Formals
heterocyclic
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic organic chemistry is the branch of organic chemistry dealing with the synthesis, proper ...
acetal
In organic chemistry, an acetal is a functional group with the connectivity . Here, the R groups can be organic fragments (a carbon atom, with arbitrary other atoms attached to that) or hydrogen, while the R' groups must be organic fragments n ...
with the chemical formula
A chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, such as pare ...
(CH2)2O2CH2. It is related to tetrahydrofuran
Tetrahydrofuran (THF), or oxolane, is an organic compound with the formula (CH2)4O. The compound is classified as heterocyclic compound, specifically a cyclic ether. It is a colorless, water- miscible organic liquid with low viscosity. It is ...
(THF) by replacement of the methylene group (CH2) at the 2-position with an oxygen atom. The corresponding saturated 6-membered C4O2 rings are called dioxanes. The isomer
In chemistry, isomers are molecules or polyatomic ions with identical molecular formula – that is, the same number of atoms of each element (chemistry), element – but distinct arrangements of atoms in space. ''Isomerism'' refers to the exi ...
ic 1,2-dioxolane (wherein the two oxygen centers are adjacent) is a peroxide
In chemistry, peroxides are a group of Chemical compound, compounds with the structure , where the R's represent a radical (a portion of a complete molecule; not necessarily a free radical) and O's are single oxygen atoms. Oxygen atoms are joined ...
. 1,3-dioxolane is used as a solvent
A solvent (from the Latin language, Latin ''wikt:solvo#Latin, solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a Solution (chemistry), solution. A solvent is usually a liquid but can also be a solid, a gas ...
and as a comonomer
A monomer ( ; ''mono-'', "one" + '' -mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or two- or three-dimensional network in a process called polymerization.
Classification
Chemis ...
in polyacetals.
As a class of compounds
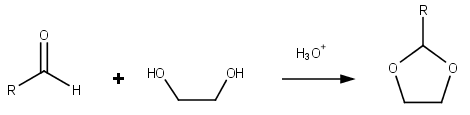
Dioxolanes are a group oforganic compound
Some chemical authorities define an organic compound as a chemical compound that contains a carbon–hydrogen or carbon–carbon bond; others consider an organic compound to be any chemical compound that contains carbon. For example, carbon-co ...
s containing the dioxolane ring. Dioxolanes can be prepared by acetalization of aldehyde
In organic chemistry, an aldehyde () (lat. ''al''cohol ''dehyd''rogenatum, dehydrogenated alcohol) is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred ...
s and ketalization of ketone
In organic chemistry, a ketone is an organic compound with the structure , where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond C=O). The simplest ketone is acetone ( ...
s with ethylene glycol
Ethylene glycol ( IUPAC name: ethane-1,2-diol) is an organic compound (a vicinal diol) with the formula . It is mainly used for two purposes: as a raw material in the manufacture of polyester fibers and for antifreeze formulations. It is an odo ...
.
 (+)-''cis''-Dioxolane is the
(+)-''cis''-Dioxolane is the trivial name
In chemistry, a trivial name is a non-systematic name for a chemical substance. That is, the name is not recognized according to the rules of any formal system of chemical nomenclature such as IUPAC inorganic or IUPAC organic nomenclature. A ...
for which is a muscarinic acetylcholine receptor
Muscarinic acetylcholine receptors (mAChRs) are acetylcholine receptors that form G protein-coupled receptor, G protein-coupled receptor complexes in the cell membranes of certain neurons and other Cell (biology), cells. They play several role ...
agonist
An agonist is a chemical that activates a Receptor (biochemistry), receptor to produce a biological response. Receptors are Cell (biology), cellular proteins whose activation causes the cell to modify what it is currently doing. In contrast, an R ...
.
Protecting groups
Organic compounds containingcarbonyl group
In organic chemistry, a carbonyl group is a functional group with the formula , composed of a carbon atom double-bonded to an oxygen atom, and it is divalent at the C atom. It is common to several classes of organic compounds (such as aldehydes ...
s sometimes need protection
Protection is any measure taken to guard something against damage caused by outside forces. Protection can be provided to physical objects, including organisms, to systems, and to intangible things like civil and political rights. Although ...
so that they do not undergo reactions during transformations of other functional group
In organic chemistry, a functional group is any substituent or moiety (chemistry), moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions r ...
s that may be present. A variety of approaches to protection and deprotection of carbonyls including as dioxolanes are known. For example, consider the compound methyl cyclohexanone-4-carboxylate, where lithium aluminium hydride
Lithium aluminium hydride, commonly abbreviated to LAH, is an inorganic compound with the chemical formula or . It is a white solid, discovered by Finholt, Bond and Schlesinger in 1947. This compound is used as a reducing agent in organic synthe ...
reduction will produce 4-hydroxymethylcyclohexanol. The ester functional group can be reduced without affecting the ketone
In organic chemistry, a ketone is an organic compound with the structure , where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond C=O). The simplest ketone is acetone ( ...
by protecting the ketone as a ketal
In organic chemistry, an acetal is a functional group with the connectivity . Here, the R groups can be organic fragments (a carbon atom, with arbitrary other atoms attached to that) or hydrogen, while the R' groups must be organic fragments n ...
. The ketal is produced by acid catalysed reaction with ethylene glycol
Ethylene glycol ( IUPAC name: ethane-1,2-diol) is an organic compound (a vicinal diol) with the formula . It is mainly used for two purposes: as a raw material in the manufacture of polyester fibers and for antifreeze formulations. It is an odo ...
, the reduction reaction carried out, and the protecting group removed by hydrolysis to produce 4-hydroxymethylcyclohexanone.
 NaBArF4 can also be used for deprotection of acetal or ketal-protected carbonyl compounds. For example, deprotection of 2-phenyl-1,3-dioxolane to
NaBArF4 can also be used for deprotection of acetal or ketal-protected carbonyl compounds. For example, deprotection of 2-phenyl-1,3-dioxolane to benzaldehyde
Benzaldehyde (C6H5CHO) is an organic compound consisting of a benzene ring with a formyl substituent. It is among the simplest aromatic aldehydes and one of the most industrially useful.
It is a colorless liquid with a characteristic almond-li ...
can be achieved in water in five minutes at 30 °C.
::PhCH(OCH2)2 + H2O Natural products
Neosporol is anatural product
A natural product is a natural compound or substance produced by a living organism—that is, found in nature. In the broadest sense, natural products include any substance produced by life. Natural products can also be prepared by chemical s ...
that includes a 1,3-dioxolane moiety, and is an isomer of sporol which has a 1,3-dioxane ring. The total synthesis
Total synthesis, a specialized area within organic chemistry, focuses on constructing complex organic compounds, especially those found in nature, using laboratory methods. It often involves synthesizing natural products from basic, commercially ...
of both compounds has been reported, and each includes a step in which a dioxolane system is formed using trifluoroperacetic acid (TFPAA), prepared by the hydrogen peroxide – urea method. This method involves no water, so it gives a completely anhydrous
A substance is anhydrous if it contains no water. Many processes in chemistry can be impeded by the presence of water; therefore, it is important that water-free reagents and techniques are used. In practice, however, it is very difficult to achie ...
peracid, necessary in this case as the presence of water would lead to unwanted side reaction
A side reaction is a chemical reaction that occurs at the same time as the actual main reaction, but to a lesser extent. It leads to the formation of by-product, so that the Yield (chemistry), yield of main product is reduced:
: + B ->[] P1
: + C ...
s.
:trifluoroacetic anhydride, + hydrogen peroxide - urea, → trifluoroperacetic acid, + trifluoroacetic acid, + urea,
In the case of neosporol, a Prilezhaev reaction
The Prilezhaev reaction, also known as the Prileschajew reaction or Prilezhaev epoxidation, is the chemical reaction of an alkene with a peroxy acid to form epoxides. It is named after Nikolai Prilezhaev, who first reported this reaction in 1909 ...
with trifluoroperacetic acid is used to convert a suitable allyl alcohol precursor to an epoxide
In organic chemistry, an epoxide is a cyclic ether, where the ether forms a three-atom ring: two atoms of carbon and one atom of oxygen. This triangular structure has substantial ring strain, making epoxides highly reactive, more so than other ...
, which then undergoes a ring-expansion reaction with a proximate carbonyl
In organic chemistry, a carbonyl group is a functional group with the formula , composed of a carbon atom double bond, double-bonded to an oxygen atom, and it is divalent at the C atom. It is common to several classes of organic compounds (such a ...
functional group to form the dioxolane ring.
 A similar approach is used in the total synthesis of sporol, with the dioxolane ring later expanded to a dioxane system.
A similar approach is used in the total synthesis of sporol, with the dioxolane ring later expanded to a dioxane system.
See also
* DioxaneReferences
External links
environmental and toxicological data
{{Muscarinic acetylcholine receptor modulators Muscarinic agonists Solvents Protecting groups Formals