|
Zwitterion
In chemistry, a zwitterion ( ; ), also called an inner salt or dipolar ion, is a molecule that contains an equal number of positively and negatively charged functional groups. : (1,2- dipolar compounds, such as ylides, are sometimes excluded from the definition.) Some zwitterions, such as amino acid zwitterions, are in chemical equilibrium with an uncharged "parent" molecule. Betaines are zwitterions that cannot isomerize to an all-neutral form, such as when the positive charge is located on a quaternary ammonium group. Similarly, a molecule containing a phosphonium group and a carboxylate group cannot isomerize. Amino acids Tautomerism of amino acids follows this stoichiometry: : The ratio of the concentrations of the two species in solution is independent of pH. It has been suggested, on the basis of theoretical analysis, that the zwitterion is stabilized in aqueous solution by hydrogen bonding with solvent water molecules. Analysis of neutron diffraction data for g ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amino Acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although over 500 amino acids exist in nature, by far the most important are the 22 α-amino acids incorporated into proteins. Only these 22 appear in the genetic code of life. Amino acids can be classified according to the locations of the core structural functional groups ( alpha- , beta- , gamma- amino acids, etc.); other categories relate to polarity, ionization, and side-chain group type ( aliphatic, acyclic, aromatic, polar, etc.). In the form of proteins, amino-acid '' residues'' form the second-largest component (water being the largest) of human muscles and other tissues. Beyond their role as residues in proteins, amino acids participate in a number of processes such as neurotransmitter transport and biosynthesis. It is thought that they played a key role in enabling life on Earth and its emergence. Amino acids are formally named by the IUPAC- IUBMB Joint Commi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid. Glycine is one of the proteinogenic amino acids. It is encoded by all the codons starting with GG (GGU, GGC, GGA, GGG). Glycine disrupts the formation of alpha-helices in secondary protein structure. Its small side chain causes it to favor random coils instead. Glycine is also an inhibitory neurotransmitter – interference with its release within the spinal cord (such as during a '' Clostridium tetani'' infection) can cause spastic paralysis due to uninhibited muscle contraction. It is the only achiral proteinogenic amino acid. It can fit into both hydrophilic and hydrophobic environments, due to its minimal side chain of only one hydrogen atom. History and etymology Glycine was discovered in 1820 by French chemist Henri Braconnot when he hydrolyzed gelatin by boiling it with sulfuric acid. He originally called it "sugar of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfamic Acid
Sulfamic acid, also known as amidosulfonic acid, amidosulfuric acid, aminosulfonic acid, sulphamic acid and sulfamidic acid, is a molecular compound with the formula H3NSO3. This colourless, water-soluble compound finds many applications. Sulfamic acid melts at 205 °C before decomposing at higher temperatures to water, sulfur trioxide, sulfur dioxide and nitrogen. Sulfamic acid (H3NSO3) may be considered an intermediate compound between sulfuric acid (H2SO4), and sulfamide (H4N2SO2), effectively replacing a hydroxyl (–OH) group with an amine (–NH2) group at each step. This pattern can extend no further in either direction without breaking down the sulfonyl (–SO2–) moiety. Sulfamates are derivatives of sulfamic acid. Production Sulfamic acid is produced industrially by treating urea with a mixture of sulfur trioxide and sulfuric acid (or oleum). The conversion is conducted in two stages, the first being sulfamation: :OC(NH2)2 + SO3 → OC(NH2)(NHSO3H) :OC(NH2)(NHS ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tautomer
In chemistry, tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert. The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hydrogen atom within the compound. The phenomenon of tautomerization is called tautomerism, also called desmotropism. Tautomerism is for example relevant to the behavior of amino acids and nucleic acids, two of the fundamental building blocks of life. Care should be taken not to confuse tautomers with depictions of "contributing structures" in chemical resonance. Tautomers are distinct chemical species that can be distinguished by their differing atomic connectivities, molecular geometries, and physicochemical and spectroscopic properties, whereas resonance forms are merely alternative Lewis structure (valence bond theory) depictions of a single chemical species, whose true structure is a quantum superposition, essentially the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dipolar Compound
In organic chemistry, a dipolar compound or simply dipole is an electrically neutral molecule carrying a positive and a negative charge in at least one canonical description. In most dipolar compounds the charges are Delocalized electron, delocalized. Unlike Salt (chemistry), salts, dipolar compounds have charges on separate atoms, not on positive and negative ions that make up the compound. Dipolar compounds exhibit a Dipole#Molecular dipoles, dipole moment. Dipolar compounds can be represented by a Resonance (chemistry), resonance structure. Contributing structures containing charged atoms are denoted as zwitterions. {{cite journal, last1=MacHiguchi, first1=Takahisa, last2=Okamoto, first2=Junko, last3=Takachi, first3=Junpei, last4=Hasegawa, first4=Toshio, last5=Yamabe, first5=Shinichi, last6=Minato, first6=Tsutomu, title=Exclusive Formation of α-Methyleneoxetanes in Ketene−Alkene Cycloadditions. Evidence for Intervention of Both an α-Methyleneoxetane and the Subsequent 1,4 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
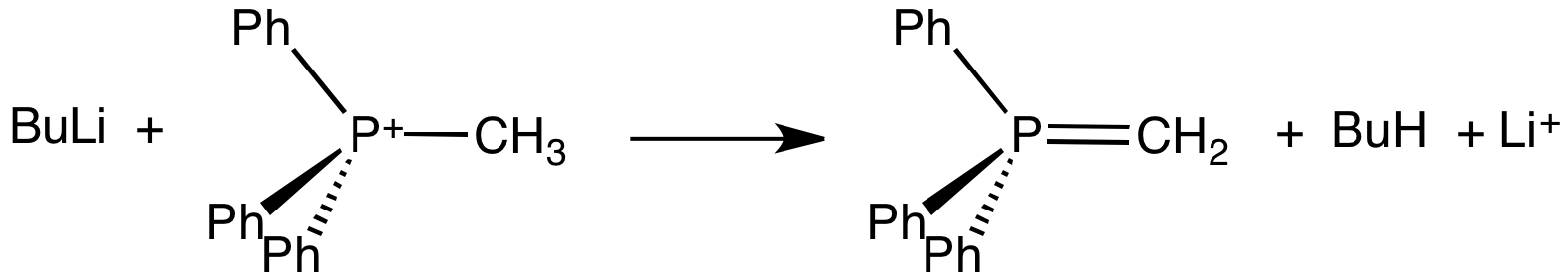
Ylide
An ylide () or ylid () is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent and an ionic bond; normally written X+–Y−. Ylides are thus 1,2- dipolar compounds, and a subclass of zwitterions. They appear in organic chemistry as reagents or reactive intermediates. The class name "ylide" for the compound should not be confused with the suffix "-ylide". Resonance structures Many ylides may be depicted by a multiply bonded form in a resonance structure, known as the ylene form, while the actual structure lies in between both forms: : The actual bonding picture of these types of ylides is strictly zwitterionic (the structure on the right) with the strong Coulombic attractio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Psilocybin
Psilocybin, also known as 4-phosphoryloxy-''N'',''N''-dimethyltryptamine (4-PO-DMT), is a natural product, naturally occurring tryptamine alkaloid and Investigational New Drug, investigational drug found in more than List of psilocybin mushroom species, 200 species of mushrooms, with Hallucinogen, hallucinogenic and Serotonin, serotonergic effects. Effects include euphoria, changes in perception, a distorted sense of time (via brain desynchronization), and perceived spiritual experiences. It can also cause adverse reactions such as nausea and panic attacks. Its effects depend on set and setting and one's subject-expectancy effect, expectations. Psilocybin is a prodrug of psilocin. That is, the compound itself is biologically inactive but quickly converted by the body to psilocin. Psilocybin is transformed into psilocin by dephosphorylation mediated via phosphatase enzymes. Psilocin is structural analog, chemically related to the neurotransmitter serotonin and acts as a binding ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proteinogenic Amino Acid
Proteinogenic amino acids are amino acids that are incorporated biosynthetically into proteins during translation from RNA. The word "proteinogenic" means "protein creating". Throughout known life, there are 22 genetically encoded (proteinogenic) amino acids, 20 in the standard genetic code and an additional 2 ( selenocysteine and pyrrolysine) that can be incorporated by special translation mechanisms. In contrast, non-proteinogenic amino acids are amino acids that are either not incorporated into proteins (like GABA, L-DOPA, or triiodothyronine), misincorporated in place of a genetically encoded amino acid, or not produced directly and in isolation by standard cellular machinery (like hydroxyproline). The latter often results from post-translational modification of proteins. Some non-proteinogenic amino acids are incorporated into nonribosomal peptides which are synthesized by non-ribosomal peptide synthetases. Both eukaryotes and prokaryotes can incorporate selenocysteine ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trimethylglycine
Trimethylglycine is an amino acid derivative with the formula . A colorless, water-soluble solid, it occurs in plants. Trimethylglycine is a zwitterion: the molecule contains both a quaternary ammonium cation, quaternary ammonium group and a carboxylate group. Trimethylglycine was the first betaine discovered; originally it was simply called betaine because it was discovered in sugar beets (''Beta vulgaris'' subsp. ''vulgaris''). Several other betaines are now known. Medical uses Betaine, sold under the brand name Cystadane, is indicated for the adjunctive treatment of homocystinuria, involving deficiencies or defects in cystathionine beta-synthase (CBS), 5,10-methylene-tetrahydrofolate reductase (MTHFR), or cobalamin cofactor metabolism (cbl). The most common side effect is elevated levels of methionine in the blood. The EU has authorized the health claim that betaine "contributes to normal homocysteine metabolism.". Biological function Biosynthesis In most organisms, gly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyridoxal-phosphate
Pyridoxal phosphate (PLP, pyridoxal 5'-phosphate, P5P), the active form of vitamin B6, is a coenzyme in a variety of enzymatic reactions. The International Union of Biochemistry and Molecular Biology has catalogued more than 140 PLP-dependent activities, corresponding to ~4% of all classified activities. The versatility of PLP arises from its ability to covalently bind the substrate, and then to act as an electrophilic catalyst, thereby stabilizing different types of carbanionic reaction intermediates. Role as a coenzyme PLP acts as a coenzyme in all transamination reactions, and in certain decarboxylation, deamination, and racemization reactions of amino acids. The aldehyde group of PLP forms a Schiff-base linkage (internal aldimine) with the ε-amino group of a specific lysine group of the aminotransferase enzyme. The α-amino group of the amino acid substrate displaces the ε-amino group of the active-site lysine residue in a process known as transaldimination. The result ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Betaine
A betaine () in chemistry is any neutral chemical compound with a positively charged cationic functional group that bears no hydrogen atom, such as a Quaternary ammonium cation, quaternary ammonium or phosphonium cation (generally: Onium compounds, onium ions), and with a negatively charged functional group, such as a carboxylate group that may not be adjacent to the cationic site. Historically, the term was reserved for trimethylglycine (TMG), which is involved in methylation reactions and detoxification of homocysteine. This is a modified amino acid consisting of glycine with three methyl groups serving as methyl donor for various metabolic pathways. Pronunciation The pronunciation of the compound reflects its origin and first isolation from sugar ''beets'' (''Beta vulgaris'' subsp. ''vulgaris''), and does not derive from the Greek letter beta (β). It is commonly pronounced ''beta-INE'' or ''BEE-tayn''. Glycine betaine The original betaine, ''N'',''N'',''N''-trimethyl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |