|
Tetrapyrrole
Tetrapyrroles are a class of chemical compounds that contain four pyrrole or pyrrole-like rings. The pyrrole/pyrrole derivatives are linked by ( or units), in either a linear or a cyclic fashion. Pyrroles are a five-atom ring with four carbon atoms and one nitrogen atom. Tetrapyrroles are common cofactors in biochemistry and their biosynthesis and degradation feature prominently in the chemistry of life. Some tetrapyrroles form the active core of compounds with crucial biochemical roles in living systems, such as hemoglobin and chlorophyll. In these two molecules, in particular, the pyrrole macrocycle ring frames a metal atom, that forms a coordination compound with the pyrroles and plays a central role in the biochemical function of those molecules. Structure Linear tetrapyrroles (called bilanes) include: *Heme breakdown products (e.g., bilirubin, biliverdin) * Phycobilins (found in cyanobacteria) * Luciferins as found in dinoflagellates and euphausiid shrimps (krill) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorin
In organic chemistry, chlorins are tetrapyrrole pigments that are partially hydrogenation, hydrogenated porphyrins. The parent chlorin is an unstable compound which undergoes air oxidation to porphine. The name chlorin derives from chlorophyll. Chlorophylls are magnesium-containing chlorins and occur as photosynthetic pigments in chloroplasts. The term "chlorin" strictly speaking refers to only compounds with the same ring oxidation state as chlorophyll. Chlorins are excellent photosensitizing agents. Various synthetic chlorins analogues such as Temoporfin, m-tetrahydroxyphenylchlorin (mTHPC) and Talaporfin, mono-L-aspartyl chlorin e6 are effectively employed in experimental photodynamic therapy as photosensitizer. Chlorophylls The most abundant chlorin is the Photosynthesis, photosynthetic pigment chlorophyll. Chlorophylls have a fifth, ketone-containing ring unlike the chlorins. Diverse chlorophylls exists, such as Chlorophyll a, chlorophyll ''a'', Chlorophyll b, chlorophyll ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heme
Heme (American English), or haem (Commonwealth English, both pronounced /Help:IPA/English, hi:m/ ), is a ring-shaped iron-containing molecule that commonly serves as a Ligand (biochemistry), ligand of various proteins, more notably as a Prosthetic group, component of hemoglobin, which is necessary to bind oxygen in the bloodstream. It is composed of four pyrrole rings with 2 Vinyl group, vinyl and 2 propionic acid side chains. Heme is biosynthesized in both the bone marrow and the liver. Heme plays a critical role in multiple different redox reactions in mammals, due to its ability to carry the oxygen molecule. Reactions include oxidative metabolism (cytochrome c oxidase, succinate dehydrogenase), xenobiotic detoxification via cytochrome P450 pathways (including Drug metabolism, metabolism of some drugs), gas sensing (Guanylate cyclase, guanyl cyclases, nitric oxide synthase), and microRNA processing (DGCR8). Heme is a coordination complex "consisting of an iron ion coordinated ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bilane
In organic chemistry, bilane is a compound with the formula or . It is a tetrapyrrole, a class of compounds with four independent pyrrole rings. Specifically, the molecule can be described as four pyrrole molecules connected in an open chain by three methylene bridges at carbons adjacent to the nitrogens, replacing the respective hydrogens. The name is also used for the class of compounds formally derived from bilane proper by replacement of some additional hydrogen atoms by various functional groups. Natural bilanes usually have side chains substituted on the two carbons in each pyrrole ring that are not adjacent to the nitrogens. Artificial bilanes may be substituted on the bridging carbons (called ''meso'' positions). The parent (unsubstituted) bilane is difficult to prepare and unstable,Claudia Ryppa, Mathias O. Senge, Sabine S. Hatscher, Erich Kleinpeter, Philipp Wacker, Uwe Schilde, and Arno Wiehe (2005): "Synthesis of Mono- and Disubstituted Porphyrins: A- and 5,10- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Porphin
Porphine or porphin is an organic compound of empirical formula . It is heterocyclic and aromatic. The molecule is a flat macrocycle, consisting of four pyrrole-like rings joined by four methine bridges, which makes it the simplest of the tetrapyrroles. The nonpolar tetrapyrrolic ring structure of porphine means it is poorly soluble in most organic solvents and hardly water soluble. As a result, porphine is mostly of theoretical interest. It has been detected in GC-MS of certain fractions of '' Piper betle''. Porphine derivatives: porphyrins Substituted derivatives of porphine are called porphyrins. Many porphyrins are found in nature with the dominant example being protoporphyrin IX. Many synthetic porphyrins are also known, including octaethylporphyrin and tetraphenylporphyrin. File:PPIXtransH.png, Derivatives of protoporphyrin IX are common in nature, the precursor to heme Heme (American English), or haem (Commonwealth English, both pronounced /Help:IPA/Englis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Porphyrin
Porphyrins ( ) are heterocyclic, macrocyclic, organic compounds, composed of four modified pyrrole subunits interconnected at their α carbon atoms via methine bridges (). In vertebrates, an essential member of the porphyrin group is heme, which is a component of hemoproteins, whose functions include carrying oxygen in the bloodstream. In plants, an essential porphyrin derivative is chlorophyll, which is involved in light harvesting and electron transfer in photosynthesis. The parent of porphyrins is porphine, a rare chemical compound of exclusively theoretical interest. Substituted porphines are called porphyrins. With a total of 26 π-electrons the porphyrin ring structure is a coordinated aromatic system. One result of the large conjugated system is that porphyrins absorb strongly in the visible region of the electromagnetic spectrum, i.e. they are deeply colored. The name "porphyrin" derives . Structure Porphyrin complexes consist of a square planar MN4 core. The p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Porphin
Porphine or porphin is an organic compound of empirical formula . It is heterocyclic and aromatic. The molecule is a flat macrocycle, consisting of four pyrrole-like rings joined by four methine bridges, which makes it the simplest of the tetrapyrroles. The nonpolar tetrapyrrolic ring structure of porphine means it is poorly soluble in most organic solvents and hardly water soluble. As a result, porphine is mostly of theoretical interest. It has been detected in GC-MS of certain fractions of '' Piper betle''. Porphine derivatives: porphyrins Substituted derivatives of porphine are called porphyrins. Many porphyrins are found in nature with the dominant example being protoporphyrin IX. Many synthetic porphyrins are also known, including octaethylporphyrin and tetraphenylporphyrin. File:PPIXtransH.png, Derivatives of protoporphyrin IX are common in nature, the precursor to heme Heme (American English), or haem (Commonwealth English, both pronounced /Help:IPA/Englis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uroporphyrinogen III
Uroporphyrinogen III is a tetrapyrrole, the first macrocycle, macrocyclic intermediate in the biosynthesis of heme, chlorophyll, vitamin B12, and siroheme. It is a colorless compound, like other porphyrinogens. Structure The molecular structure of uroporphyrinogen III can be described as a hexahydroporphine core, where each pyrrole ring has the hydrogen atoms on its two outermost carbons replaced by an acetic acid group (, "A") and a propionic acid group (, "P"). The groups are attached in an asymmetric way: going around the macrocycle, the order is AP-AP-AP-PA. Biosynthesis and metabolism In the general porphyrin biosynthesis pathway, uroporphyrinogen III is derived from the linear tetrapyrrole preuroporphyrinogen (a substituted hydroxymethylbilane) by the action of the enzyme uroporphyrinogen-III synthase, uroporphyrinogen-III cosynthase. The conversion entails a reversal of the last pyrrole unit (thus swapping the acetic and propionic acid groups) and a condensation reacti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
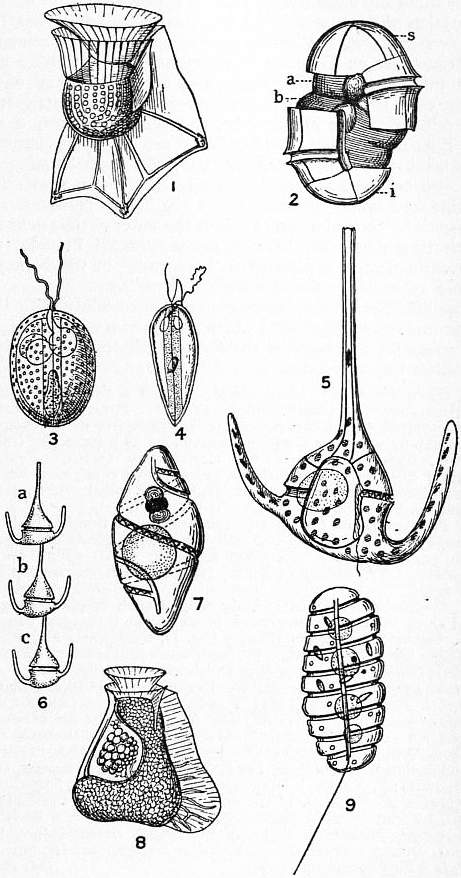
Dinoflagellate
The Dinoflagellates (), also called Dinophytes, are a monophyletic group of single-celled eukaryotes constituting the phylum Dinoflagellata and are usually considered protists. Dinoflagellates are mostly marine plankton, but they are also common in freshwater habitats. Their populations vary with sea surface temperature, salinity, and depth. Many dinoflagellates are photosynthetic, but a large fraction of these are in fact mixotrophic, combining photosynthesis with ingestion of prey ( phagotrophy and myzocytosis). In terms of number of species, dinoflagellates are one of the largest groups of marine eukaryotes, although substantially smaller than diatoms. Some species are endosymbionts of marine animals and play an important part in the biology of coral reefs. Other dinoflagellates are unpigmented predators on other protozoa, and a few forms are parasitic (for example, '' Oodinium'' and '' Pfiesteria''). Some dinoflagellates produce resting stages, called dinoflagellate cys ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phycobilin
Phycobilins (from Greek: '' (phykos)'' meaning "alga", and from Latin: ''bilis'' meaning "bile") are light-capturing bilins found in cyanobacteria and in the chloroplasts of red algae, glaucophytes and some cryptomonads (though not in green algae and plants). Most of their molecules consist of a chromophore which makes them coloured. They are unique among the photosynthetic pigments in that they are bonded to certain water-soluble proteins, known as phycobiliproteins. Phycobiliproteins then pass the light energy to chlorophylls for photosynthesis. The phycobilins are especially efficient at absorbing red, orange, yellow, and green light (in the range 520 to 630 nm), wave lengths that are not well absorbed by chlorophyll ''a''. Organisms growing in shallow waters tend to contain phycobilins that can capture yellow/red light, while those at greater depth often contain more of the phycobilins that can capture green light, which is relatively more abundant there. The phycobili ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phycoerythrobilin
Phycoerythrobilin is a red phycobilin, i.e. an open tetrapyrrole chromophore found in cyanobacteria and in the chloroplasts of red algae, glaucophytes and some cryptomonads. Phycoerythrobilin is present in the phycobiliprotein phycoerythrin, of which it is the terminal acceptor of energy. The amount of phycoerythrobilin in phycoerythrins varies a lot, depending on the considered organism. In some Rhodophytes and oceanic cyanobacteria, phycoerythrobilin is also present in the phycocyanin Phycocyanin is a pigment-protein complex from the light-harvesting phycobiliprotein family, along with allophycocyanin and phycoerythrin. It is an accessory pigment to chlorophyll. All phycobiliproteins are water-soluble, so they cannot exist ..., then termed R-phycocyanin. Like all phycobilins, phycoerythrobilin is covalently linked to these phycobiliproteins by a thioether bond. References External links Chemical Structure of phycoerythrobilin Tetrapyrroles Photosynthetic pigment ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dinoflagellates
The Dinoflagellates (), also called Dinophytes, are a monophyletic group of single-celled eukaryotes constituting the phylum Dinoflagellata and are usually considered protists. Dinoflagellates are mostly marine plankton, but they are also common in freshwater habitats. Their populations vary with sea surface temperature, salinity, and depth. Many dinoflagellates are photosynthetic, but a large fraction of these are in fact mixotrophic, combining photosynthesis with ingestion of prey ( phagotrophy and myzocytosis). In terms of number of species, dinoflagellates are one of the largest groups of marine eukaryotes, although substantially smaller than diatoms. Some species are endosymbionts of marine animals and play an important part in the biology of coral reefs. Other dinoflagellates are unpigmented predators on other protozoa, and a few forms are parasitic (for example, '' Oodinium'' and '' Pfiesteria''). Some dinoflagellates produce resting stages, called dinoflagellate cysts ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |