|
Sorbate
Sorbic acid, or 2,4-hexadienoic acid, is a natural organic compound used as a food preservative. It has the chemical formula and the structure . It is a colourless solid that is slightly soluble in water and sublimes readily. It was first isolated from the unripe berries of the ''Sorbus aucuparia'' (rowan tree), hence its name. Production The traditional route to sorbic acid involves condensation of malonic acid and crotonaldehyde. It can also be prepared from isomeric hexadienoic acids, which are available via a nickel-catalyzed reaction of allyl chloride, acetylene, and carbon monoxide. The route used commercially, however, is from crotonaldehyde and ketene. An estimated 30,000 tons are produced annually. History Sorbic acid was isolated in 1859 by distillation of rowanberry oil by A. W. von Hofmann. This affords parasorbic acid, the lactone of sorbic acid, which he converted to sorbic acid by hydrolysis. Its antimicrobial activities were discovered in the late 1930s and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Sorbate
Potassium sorbate is the potassium salt of sorbic acid, chemical formula CH3CH=CH−CH=CH−CO2K. It is a white salt that is very soluble in water (58.2% at 20 °C). It is primarily used as a food preservative (E number 202). Potassium sorbate is effective in a variety of applications including food, wine, and personal-care products. While sorbic acid occurs naturally in rowan berries, virtually all of the world's supply of sorbic acid, from which potassium sorbate is derived, is manufactured synthetically. Production Potassium sorbate is produced industrially by neutralizing sorbic acid with potassium hydroxide. The precursor sorbic acid is produced in a two-step process via the condensation of crotonaldehyde and ketene. Uses Potassium sorbate is used to inhibit molds and yeasts in many foods, such as cheese, wine, yogurt, dried meats, apple cider, dehydrated fruits, soft drinks and fruit drinks, and baked goods.Erich Lück, Martin Jager and Nico Raczek "Sorbic Acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Sorbate
Potassium sorbate is the potassium salt of sorbic acid, chemical formula CH3CH=CH−CH=CH−CO2K. It is a white salt that is very soluble in water (58.2% at 20 °C). It is primarily used as a food preservative (E number 202). Potassium sorbate is effective in a variety of applications including food, wine, and personal-care products. While sorbic acid occurs naturally in rowan berries, virtually all of the world's supply of sorbic acid, from which potassium sorbate is derived, is manufactured synthetically. Production Potassium sorbate is produced industrially by neutralizing sorbic acid with potassium hydroxide. The precursor sorbic acid is produced in a two-step process via the condensation of crotonaldehyde and ketene. Uses Potassium sorbate is used to inhibit molds and yeasts in many foods, such as cheese, wine, yogurt, dried meats, apple cider, dehydrated fruits, soft drinks and fruit drinks, and baked goods.Erich Lück, Martin Jager and Nico Raczek "Sorbic Acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
E Number
E numbers ("E" stands for "Europe") are codes for substances used as food additives, including those found naturally in many foods such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly found on food labels, their safety assessment and approval are the responsibility of the European Food Safety Authority (EFSA). The fact that an additive has an E number implies that its use was at one time permitted in products for sale in the European Single Market; some of these additives are no longer allowed today. Having a single unified list for food additives was first agreed upon in 1962 with food colouring. In 1964, the directives for preservatives were added, in 1970 antioxidants were added, in 1974 emulsifiers, stabilisers, thickeners and gelling agents were added as well. Numbering schemes The numbering scheme follows that of the International Numbering System (INS) as determined by the ''Codex Alimentarius'' committee, t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Sorbate
Calcium sorbate is the calcium salt of sorbic acid. Calcium sorbate is a polyunsaturated fatty acid salt. It is a commonly used food preservative; its E number E numbers ("E" stands for "Europe") are codes for substances used as food additives, including those found naturally in many foods such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly ... is E203, but it is no longer allowed to be used in the European Union. References External links Calcium sorbate UKfoodguide.net Calcium compounds Sorbates E-number additives {{organic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Sorbate
Sodium sorbate is the sodium salt of sorbic acid. It is an unstable white solid. Unlike other sorbic acid salts such as potassium sorbate (E202) and calcium sorbate (E203), the use of sodium sorbate as a food additive is not allowed in the EU due to potential genotoxic effects. Its E-number E numbers ("E" stands for "Europe") are codes for substances used as food additives, including those found naturally in many foods such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly f ... is E201. References Organic sodium salts Sorbates {{organic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Sorbate
Calcium sorbate is the calcium salt of sorbic acid. Calcium sorbate is a polyunsaturated fatty acid salt. It is a commonly used food preservative; its E number E numbers ("E" stands for "Europe") are codes for substances used as food additives, including those found naturally in many foods such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly ... is E203, but it is no longer allowed to be used in the European Union. References External links Calcium sorbate UKfoodguide.net Calcium compounds Sorbates E-number additives {{organic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Preservative
A preservative is a substance or a chemical that is added to products such as food products, beverages, pharmaceutical drugs, paints, biological samples, cosmetics, wood, and many other products to prevent decomposition by microbial growth or by undesirable chemical changes. In general, preservation is implemented in two modes, chemical and physical. Chemical preservation entails adding chemical compounds to the product. Physical preservation entails processes such as refrigeration or drying.Erich Lück and Gert-Wolfhard von Rymon Lipinski "Foods, 3. Food Additives" in ''Ullmann's Encyclopedia of Industrial Chemistry'', 2002, Wiley-VCH, Weinheim. Preservative food additives reduce the risk of foodborne infections, decrease microbial spoilage, and preserve fresh attributes and nutritional quality. Some physical techniques for food preservation include dehydration, UV-C radiation, freeze-drying, and refrigeration. Chemical preservation and physical preservation techniques are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fungi
A fungus ( : fungi or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and molds, as well as the more familiar mushrooms. These organisms are classified as a kingdom, separately from the other eukaryotic kingdoms, which by one traditional classification include Plantae, Animalia, Protozoa, and Chromista. A characteristic that places fungi in a different kingdom from plants, bacteria, and some protists is chitin in their cell walls. Fungi, like animals, are heterotrophs; they acquire their food by absorbing dissolved molecules, typically by secreting digestive enzymes into their environment. Fungi do not photosynthesize. Growth is their means of mobility, except for spores (a few of which are flagellated), which may travel through the air or water. Fungi are the principal decomposers in ecological systems. These and other differences place fungi in a single group of related organisms, named the ''Eumycota'' (''t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Yeast
Yeasts are eukaryotic, single-celled microorganisms classified as members of the fungus kingdom. The first yeast originated hundreds of millions of years ago, and at least 1,500 species are currently recognized. They are estimated to constitute 1% of all described fungal species. Yeasts are unicellular organisms that evolved from multicellular ancestors, with some species having the ability to develop multicellular characteristics by forming strings of connected budding cells known as pseudohyphae or false hyphae. Yeast sizes vary greatly, depending on species and environment, typically measuring 3–4 µm in diameter, although some yeasts can grow to 40 µm in size. Most yeasts reproduce asexually by mitosis, and many do so by the asymmetric division process known as budding. With their single-celled growth habit, yeasts can be contrasted with molds, which grow hyphae. Fungal species that can take both forms (depending on temperature or other conditions) a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mold (fungus)
A mold () or mould () is one of the structures certain fungi can form. The dust-like, colored appearance of molds is due to the formation of spores containing fungal secondary metabolites. The spores are the dispersal units of the fungi. Not all fungi form molds. Some fungi form mushrooms; others grow as single cells and are called microfungi (for example yeasts). A large and taxonomically diverse number of fungal species form molds. The growth of hyphae results in discoloration and a fuzzy appearance, especially on food. The network of these tubular branching hyphae, called a mycelium, is considered a single organism. The hyphae are generally transparent, so the mycelium appears like very fine, fluffy white threads over the surface. Cross-walls (septa) may delimit connected compartments along the hyphae, each containing one or multiple, genetically identical nuclei. The dusty texture of many molds is caused by profuse production of asexual spores (conidia) formed by dif ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrosamines
In organic chemistry, nitrosamines (or more formally ''N''-Nitrosamines) are organic compounds with the chemical structure , where R is usually an alkyl group. They feature a nitroso group () bonded to a deprotonated amine. Most nitrosamines are carcinogenic in nonhuman animals. A 2006 systematic review supports a "positive association between nitrite and nitrosamine intake and gastric cancer, between meat and processed meat intake and gastric cancer and oesophageal cancer, and between preserved fish, vegetable and smoked food intake and gastric cancer, but is not conclusive". Chemistry The organic chemistry of nitrosamines is well developed with regard to their syntheses, their structures, and their reactions. They usually are produced by the reaction of nitrous acid () and secondary amines. :HONO + R2NH -> R2N-NO + H2O The nitrous acid usually arises from protonation of a nitrite. This synthesis method is relevant to the generation of nitrosamines under some biological cond ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
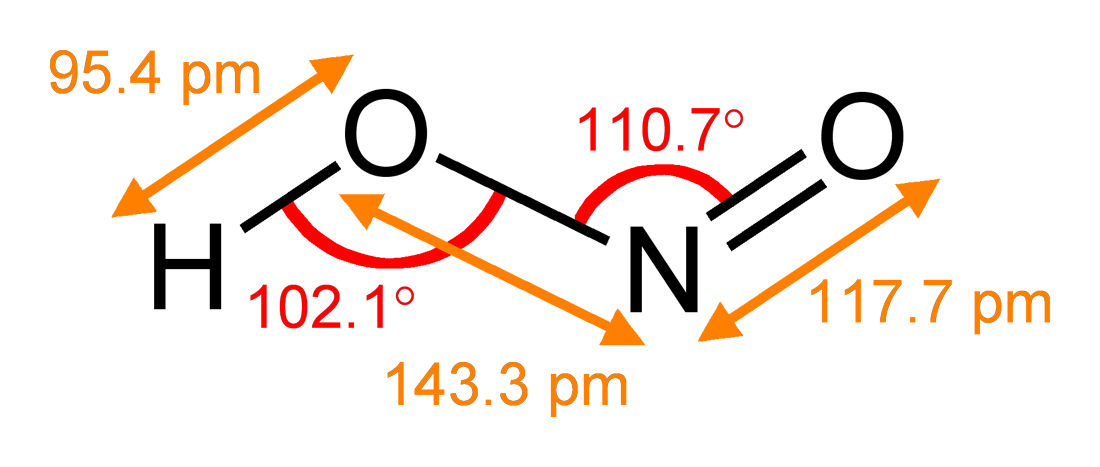
Nitrites
The nitrite ion has the chemical formula . Nitrite (mostly sodium nitrite) is widely used throughout chemical and pharmaceutical industries. The nitrite anion is a pervasive intermediate in the nitrogen cycle in nature. The name nitrite also refers to organic compounds having the –ONO group, which are esters of nitrous acid. Production Sodium nitrite is made industrially by passing a mixture of nitrogen oxides into aqueous sodium hydroxide or sodium carbonate solution: : The product is purified by recrystallization. Alkali metal nitrites are thermally stable up to and beyond their melting point (441 °C for KNO2). Ammonium nitrite can be made from dinitrogen trioxide, N2O3, which is formally the anhydride of nitrous acid: :2 NH3 + H2O + N2O3 → 2 NH4NO2 Structure The nitrite ion has a symmetrical structure (C2v symmetry), with both N–O bonds having equal length and a bond angle of about 115°. In valence bond theory, it is described as a resonance hybrid with equa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |