|
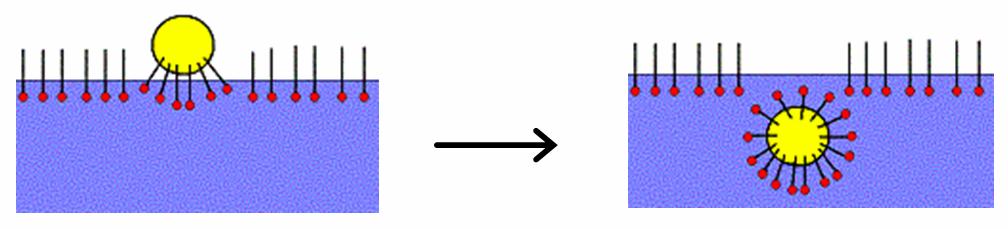
Solubilize
Micellar solubilization (solubilization) is the process of incorporating the solubilizate (the component that undergoes solubilization) into or onto micelles. Solubilization may occur in a system consisting of a solvent, an association colloid (a colloid that forms micelles), and at least one other solubilizate. Usage of the term Solubilization is distinct from dissolution because the resulting fluid is a colloidal dispersion involving an association colloid. This suspension is distinct from a true solution, and the amount of the solubilizate in the micellar system can be different (often higher) than the regular solubility of the solubilizate in the solvent. In non-chemical literature and in everyday language, the term "solubilization" is sometimes used in a broader meaning as "to bring to a solution or (non- sedimenting) suspension" by any means, e.g., leaching by a reaction with an acid. Application Micellar solubilization is widely utilized, e.g. in laundry washing usin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Micelle
A micelle () or micella () ( or micellae, respectively) is an aggregate (or supramolecular assembly) of surfactant amphipathic lipid molecules dispersed in a liquid, forming a colloidal suspension (also known as associated colloidal system). A typical micelle in water forms an aggregate, with the hydrophilic "head" regions in contact with surrounding solvent, sequestering the hydrophobic single-tail regions in the micelle centre. This phase is caused by the packing behavior of single-tail lipids in a bilayer. The difficulty in filling the volume of the interior of a bilayer, while accommodating the area per head group forced on the molecule by the hydration of the lipid head group, leads to the formation of the micelle. This type of micelle is known as a normal-phase micelle (or oil-in-water micelle). Inverse micelles have the head groups at the centre with the tails extending out (or water-in-oil micelle). Micelles are approximately spherical in shape. Other shapes, such ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dissolution (chemistry)
Solvations describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the solute, including solubility, reactivity, and color, as well as influencing the properties of the solvent such as its viscosity and density. If the attractive forces between the solvent and solute particles are greater than the attractive forces holding the solute particles together, the solvent particles pull the solute particles apart and surround them. The surrounded solute particles then move away from the solid solute and out into the solution. Ions are surrounded by a concentric shell of solvent. Solvation is the process of reorganizing solvent and solute molecules into solvation complexes and involves bond formation, hydrogen bonding, and van der Waals forces. Solvation of a solute by water is called hydration. Solubility of solid compounds de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrotrope
A hydrotrope is a compound that solubilizes hydrophobic compounds in aqueous solutions by means other than micellar solubilization. Typically, hydrotropes consist of a hydrophilic part and a hydrophobic part (similar to surfactants), but the hydrophobic part is generally too small to cause spontaneous self-aggregation. Hydrotropes do not have a critical concentration above which self-aggregation spontaneously starts to occur (as found for micelle- and vesicle-forming surfactants, which have a critical micelle concentration (cmc) and a critical vesicle concentration (cvc)). Instead, some hydrotropes aggregate in a step-wise self-aggregation process, gradually increasing aggregation size. However, many hydrotropes do not seem to self-aggregate at all, unless a solubilizate has been added. Examples of hydrotropes include urea, tosylate, cumenesulfonate and xylenesulfonate. The term ''hydrotropy'' was originally put forward by Carl Neuberg to describe the increase in the solubility ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solvent
A solvent (from the Latin language, Latin ''wikt:solvo#Latin, solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a Solution (chemistry), solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for Chemical polarity#Polarity of molecules, polar molecules, and the most common solvent used by living things; all the ions and proteins in a Cell (biology), cell are dissolved in water within the cell. Major uses of solvents are in paints, paint removers, inks, and dry cleaning. Specific uses for Organic compound, organic solvents are in dry cleaning (e.g. tetrachloroethylene); as paint thinners (toluene, turpentine); as nail polish removers and solvents of glue (acetone, methyl acetate, ethyl acetate); in spot removers (hexane, petrol ether); in detergents (D-limonene, citrus terpenes); and in perfumes (ethanol). Solvents find various applications in chemical, pharmaceutical, oil, and gas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Colloid
A colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout another substance. Some definitions specify that the particles must be dispersed in a liquid, while others extend the definition to include substances like aerosols and gels. The term colloidal suspension refers unambiguously to the overall mixture (although a narrower sense of the word '' suspension'' is distinguished from colloids by larger particle size). A colloid has a dispersed phase (the suspended particles) and a continuous phase (the medium of suspension). The dispersed phase particles have a diameter of approximately 1 nanometre to 1 micrometre. Some colloids are translucent because of the Tyndall effect, which is the scattering of light by particles in the colloid. Other colloids may be opaque or have a slight color. Colloidal suspensions are the subject of interface and colloid science. This field of study began in 1845 by Franc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solution (chemistry)
In chemistry, a solution is defined by IUPAC as "A liquid or solid phase containing more than one substance, when for convenience one (or more) substance, which is called the solvent, is treated differently from the other substances, which are called solutes. When, as is often but not necessarily the case, the sum of the mole fractions of solutes is small compared with unity, the solution is called a dilute solution. A superscript attached to the ∞ symbol for a property of a solution denotes the property in the limit of infinite dilution." One important parameter of a solution is the concentration, which is a measure of the amount of solute in a given amount of solution or solvent. The term " aqueous solution" is used when one of the solvents is water. Types ''Homogeneous'' means that the components of the mixture form a single phase. ''Heterogeneous'' means that the components of the mixture are of different phase. The properties of the mixture (such as concentration, temp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solubility
In chemistry, solubility is the ability of a chemical substance, substance, the solute, to form a solution (chemistry), solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution. The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a wikt:saturated#Chemistry, saturated solution, one in which no more solute can be dissolved. At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be "miscibility, miscible in all proportions" (or just "miscible"). The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,J. de Swaan Arons and G. A. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sedimentation
Sedimentation is the deposition of sediments. It takes place when particles in suspension settle out of the fluid in which they are entrained and come to rest against a barrier. This is due to their motion through the fluid in response to the forces acting on them: these forces can be due to gravity, centrifugal acceleration, or electromagnetism. Settling is the falling of suspended particles through the liquid, whereas sedimentation is the final result of the settling process. In geology, sedimentation is the deposition of sediments which results in the formation of sedimentary rock. The term is broadly applied to the entire range of processes that result in the formation of sedimentary rock, from initial erosion through sediment transport and settling to the lithification of the sediments. However, the strict geological definition of sedimentation is the mechanical deposition of sediment particles from an initial suspension in air or water. Sedimentation may pertain to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Leaching (chemistry)
Leaching is the process of a solute becoming detached or extracted from its carrier substance by way of a solvent. Leaching is a naturally occurring process which scientists have adapted for a variety of applications with a variety of methods. Specific extraction methods depend on the soluble characteristics relative to the sorbent material such as concentration, distribution, nature, and size. Leaching can occur naturally seen from plant substances (inorganic and organic), solute leaching in soil, and in the decomposition of organic materials. Leaching can also be applied affectedly to enhance water quality and contaminant removal, as well as for disposal of hazardous waste products such as fly ash, or rare earth elements (REEs). Understanding leaching characteristics is important in preventing or encouraging the leaching process and preparing for it in the case where it is inevitable. In an ideal leaching equilibrium stage, all the solute is dissolved by the solvent, lea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Laundry
Laundry is the washing of clothing and other textiles, and, more broadly, their drying and ironing as well. Laundry has been part of history since humans began to wear clothes, so the methods by which different cultures have dealt with this universal human need are of interest to several branches of scholarship. Laundry work has traditionally been highly gendered, with the responsibility in most cultures falling to women (formerly known as laundresses or washerwomen). The Industrial Revolution gradually led to mechanized solutions to laundry work, notably the washing machine and later the tumble dryer. Laundry, like cooking and child care, is still done both at home and by commercial establishments outside the home. The word "laundry" may refer to the clothing itself, or to the place where the cleaning happens. An individual home may have a laundry room; a utility room includes, but is not restricted to, the function of washing clothes. An apartment building or student ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Washing
Washing is a method of cleaning, usually with water and soap or detergent. Regularly washing and then rinsing both body and clothing is an essential part of good hygiene and health. Often people use soaps and detergents to assist in the emulsification of oils and dirt particles so they can be washed away. The soap can be applied directly, or with the aid of a Towel, washcloth or assisted with Sponge (tool), sponges or similar List of cleaning tools, cleaning tools. In social contexts, washing refers to the act of bathing, or washing different parts of the body, such as Hand washing, hands, Hair washing, hair, or Face washing, faces. Excessive washing may damage the hair, causing dandruff, or cause rough skin/skin lesions. Some washing of the body is done ritually in religions like Christianity and Judaism, as an Ritual purification, act of purification. Washing can also refer to washing objects. For example, laundry, washing of clothing or other cloth items, like bedsheets, or Di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |