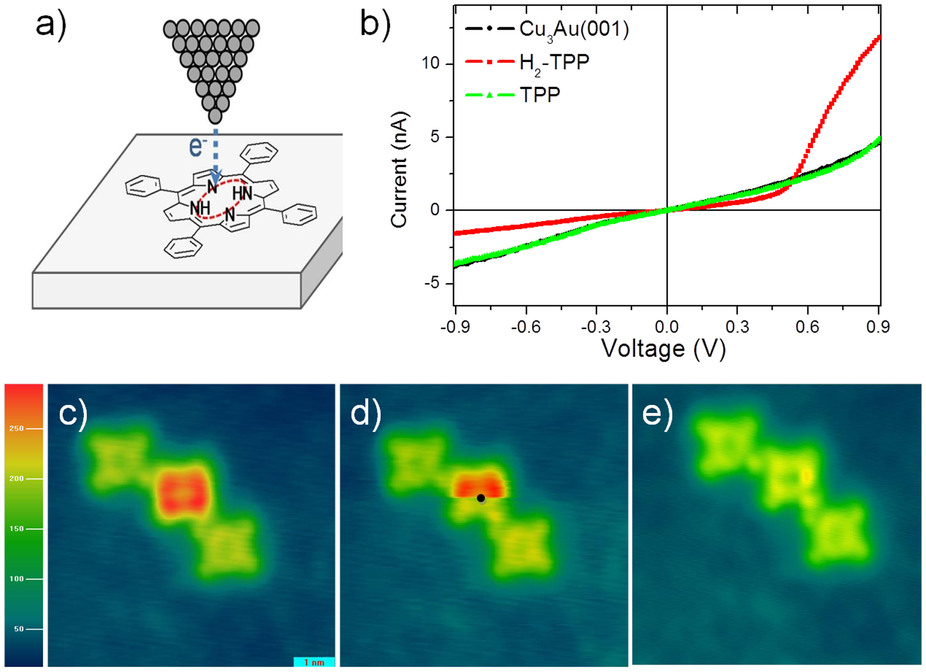
|
Porphine
Porphine or porphin is an organic chemical compound with formula . The molecule, which is flat, consists of four pyrrole-like rings joined by four methine (= C H−) groups to form a larger macrocycle ring, which makes it the simplest of the tetrapyrroles. It is classified as an aromatic and heterocyclic compound. Porphine is only of theoretical interest. It has been detected in GC-MS of certain fractions of ''Piper betle''. Porphine derivatives: porphyrins Substituted derivatives of porphine are called porphyrins. Many porphyrins are found in nature with the dominant example being protoporphyrin IX. Many synthetic porphyrins are also known, including octaethylporphyrin and tetraphenylporphyrin. File:PPIXtransH.png, Derivatives of protoporphyrin IX are common in nature, the precursor to heme Heme, or haem (pronounced / hi:m/ ), is a precursor to hemoglobin, which is necessary to bind oxygen in the bloodstream. Heme is biosynthesized in both the bone marrow and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Porphin Resonance Structures
Porphine or porphin is an organic chemical compound with formula . The molecule, which is flat, consists of four pyrrole-like rings joined by four methine (= C H−) groups to form a larger macrocycle ring, which makes it the simplest of the tetrapyrroles. It is classified as an aromatic and heterocyclic compound. Porphine is only of theoretical interest. It has been detected in GC-MS of certain fractions of ''Piper betle''. Porphine derivatives: porphyrins Substituted derivatives of porphine are called porphyrins. Many porphyrins are found in nature with the dominant example being protoporphyrin IX. Many synthetic porphyrins are also known, including octaethylporphyrin and tetraphenylporphyrin. File:PPIXtransH.png, Derivatives of protoporphyrin IX are common in nature, the precursor to hemes. File:H2octaethylporphyrin.png , Octaethylporphyrin (H2OEP) is a synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, OEP2− is highly symmetrical. Fil ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protoporphyrin IX
Protoporphyrin IX is an organic compound, classified as a porphyrin, that plays an important role in living organisms as a precursor to other critical compounds like heme (hemoglobin) and chlorophyll. It is a deeply colored solid that is not soluble in water. The name is often abbreviated as PPIX. Protoporphyrin IX contains a porphine core, a tetrapyrrole macrocycle with a marked aromatic character. Protoporphyrin IX is essentially planar, except for the N-H bonds that are bent out of the plane of the rings, in opposite (trans) directions. Nomenclature The general term protoporphyrin refers to porphine derivatives that have the outer hydrogen atoms in the four pyrrole rings replaced by other functional groups. The prefix proto often means 'first' in science nomenclature (such as carbon protoxide), hence Hans Fischer is thought to have coined the name protoporphyrin as the first class of porphyrins. Fischer described iron-deprived heme becoming the "proto-" porphyrin, parti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Octaethylporphyrin
Octaethylporphyrin (H2OEP) is an organic compound that is a relative of naturally occurring heme pigments. The compound is used in the preparation of models for the prosthetic group in heme proteins. It is a dark purple solid that is soluble in organic solvents. As its conjugate base OEP2-, it forms a range of transition metal porphyrin complexes. When treated with ferric chloride in hot acetic acid solution, it gives the square pyramidal complex Fe(OEP)Cl. It also forms the square planar complexes Ni(OEP) and Cu(OEP). Contrast with other porphyrins Unlike complexes of the naturally occurring porphyrins, OEP complexes have four-fold symmetry, which simplifies spectroscopic analysis. In contrast to tetraphenylporphyrin and related analogues, H2OEP features unprotected meso positions. In this way, it is a more accurate model for naturally occurring porphyrins. Synthesis H2OEP is prepared by condensation of 3,4-diethylpyrrole with formaldehyde Formaldehyde ( , ) ( system ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Octaethylporphyrin
Octaethylporphyrin (H2OEP) is an organic compound that is a relative of naturally occurring heme pigments. The compound is used in the preparation of models for the prosthetic group in heme proteins. It is a dark purple solid that is soluble in organic solvents. As its conjugate base OEP2-, it forms a range of transition metal porphyrin complexes. When treated with ferric chloride in hot acetic acid solution, it gives the square pyramidal complex Fe(OEP)Cl. It also forms the square planar complexes Ni(OEP) and Cu(OEP). Contrast with other porphyrins Unlike complexes of the naturally occurring porphyrins, OEP complexes have four-fold symmetry, which simplifies spectroscopic analysis. In contrast to tetraphenylporphyrin and related analogues, H2OEP features unprotected meso positions. In this way, it is a more accurate model for naturally occurring porphyrins. Synthesis H2OEP is prepared by condensation of 3,4-diethylpyrrole with formaldehyde Formaldehyde ( , ) ( system ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Liv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chelating Agents
Chelation is a type of bonding of ions and molecules to metal ions. It involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central metal atom. These ligands are called chelants, chelators, chelating agents, or sequestering agents. They are usually organic compounds, but this is not a necessity, as in the case of zinc and its use as a maintenance therapy to prevent the absorption of copper in people with Wilson's disease. Chelation is useful in applications such as providing nutritional supplements, in chelation therapy to remove toxic metals from the body, as contrast agents in MRI scanning, in manufacturing using homogeneous catalysts, in chemical water treatment to assist in the removal of metals, and in fertilizers. Chelate effect The chelate effect is the greater affinity of chelating ligands for a metal ion than that of similar nonchelating (monodentate) ligands for the same met ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biomolecules
A biomolecule or biological molecule is a loosely used term for molecules present in organisms that are essential to one or more typically biological processes, such as cell division, morphogenesis, or development. Biomolecules include large macromolecules (or polyelectrolytes) such as proteins, carbohydrates, lipids, and nucleic acids, as well as small molecules such as primary metabolites, secondary metabolites and natural products. A more general name for this class of material is biological materials. Biomolecules are an important element of living organisms, those biomolecules are often endogenous, produced within the organism but organisms usually need exogenous biomolecules, for example certain nutrients, to survive. Biology and its subfields of biochemistry and molecular biology study biomolecules and their reactions. Most biomolecules are organic compounds, and just four elements— oxygen, carbon, hydrogen, and nitrogen—make up 96% of the human ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetraphenylporphyrin
Tetraphenylporphyrin, abbreviated TPP or H2TPP, is a synthetic heterocyclic compound that resembles naturally occurring porphyrins. Porphyrins are dyes and cofactors found in hemoglobin and cytochromes and are related to chlorophyll and vitamin B12. The study of naturally occurring porphyrins is complicated by their low symmetry and the presence of polar substituents. Tetraphenylporphyrin is hydrophobic, symmetrically substituted, and easily synthesized. The compound is a dark purple solid that dissolves in nonpolar organic solvents such as chloroform and benzene. Synthesis and structure Tetraphenylporphyrin was first synthesized in 1935 by Rothemund, who caused benzaldehyde and pyrrole to react in a sealed bomb at 150 °C for 24 h. Adler and Longo modified the Rothemund method by allowing benzaldehyde and pyrrole to react for 30 min in refluxing propionic acid (141 °C) open to the air: :8 C4H4NH + 8 C6H5CHO + 3 O2 → 2 (C6H5C)4(C4H2N)2(C4H2NH)2 + 14 H2O De ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heme
Heme, or haem (pronounced / hi:m/ ), is a precursor to hemoglobin, which is necessary to bind oxygen in the bloodstream. Heme is biosynthesized in both the bone marrow and the liver. In biochemical terms, heme is a coordination complex "consisting of an iron ion coordinated to a porphyrin acting as a tetradentate ligand, and to one or two axial ligands." The definition is loose, and many depictions omit the axial ligands. Among the metalloporphyrins deployed by metalloproteins as prosthetic groups, heme is one of the most widely used and defines a family of proteins known as hemoproteins. Hemes are most commonly recognized as components of hemoglobin, the red pigment in blood, but are also found in a number of other biologically important hemoproteins such as myoglobin, cytochromes, catalases, heme peroxidase, and endothelial nitric oxide synthase. The word ''haem'' is derived from Greek ''haima'' meaning "blood". Function Hemoproteins have diverse biological fun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetraphenylporphyrin
Tetraphenylporphyrin, abbreviated TPP or H2TPP, is a synthetic heterocyclic compound that resembles naturally occurring porphyrins. Porphyrins are dyes and cofactors found in hemoglobin and cytochromes and are related to chlorophyll and vitamin B12. The study of naturally occurring porphyrins is complicated by their low symmetry and the presence of polar substituents. Tetraphenylporphyrin is hydrophobic, symmetrically substituted, and easily synthesized. The compound is a dark purple solid that dissolves in nonpolar organic solvents such as chloroform and benzene. Synthesis and structure Tetraphenylporphyrin was first synthesized in 1935 by Rothemund, who caused benzaldehyde and pyrrole to react in a sealed bomb at 150 °C for 24 h. Adler and Longo modified the Rothemund method by allowing benzaldehyde and pyrrole to react for 30 min in refluxing propionic acid (141 °C) open to the air: :8 C4H4NH + 8 C6H5CHO + 3 O2 → 2 (C6H5C)4(C4H2N)2(C4H2NH)2 + 14 H2O De ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Porphyrin
Porphyrins ( ) are a group of heterocyclic macrocycle organic compounds, composed of four modified pyrrole subunits interconnected at their α carbon atoms via methine bridges (=CH−). The parent of porphyrin is porphine, a rare chemical compound of exclusively theoretical interest. Substituted porphines are called porphyrins. With a total of 26 π-electrons, of which 18 π-electrons form a planar, continuous cycle, the porphyrin ring structure is often described as aromatic. One result of the large conjugated system is that porphyrins typically absorb strongly in the visible region of the electromagnetic spectrum, i.e. they are deeply colored. The name "porphyrin" derives from the Greek word πορφύρα (''porphyra''), meaning ''purple''. Complexes of porphyrins Concomitant with the displacement of two N-''H'' protons, porphyrins bind metal ions in the N4 "pocket". The metal ion usually has a charge of 2+ or 3+. A schematic equation for these syntheses is shown: :H2porp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrrole
Pyrrole is a heterocyclic aromatic organic compound, a five-membered ring with the formula C4 H4 NH. It is a colorless volatile liquid that darkens readily upon exposure to air. Substituted derivatives are also called pyrroles, e.g., ''N''-methylpyrrole, C4H4NCH3. Porphobilinogen, a trisubstituted pyrrole, is the biosynthetic precursor to many natural products such as heme. Pyrroles are components of more complex macrocycles, including the porphyrinogens and products derived therefrom, including porphyrins of heme, the chlorins, bacteriochlorins, and chlorophylls. Properties Pyrrole is a colorless volatile liquid that darkens readily upon exposure to air, and is usually purified by distillation immediately before use. Pyrrole has a nutty odor. Pyrrole is a 5-membered aromatic heterocycle, like furan and thiophene. Unlike furan and thiophene, it has a dipole in which the positive end lies on the side of the heteroatom, with a dipole moment of 1.58 D. In CDCl3, it ha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |