|
Polysulfide
Polysulfides are a class of chemical compounds derived from anionic chains of sulfur atoms. There are two main classes of polysulfides: inorganic and organic. The inorganic polysulfides have the general formula . These anions are the conjugate bases of polysulfanes . Organic polysulfides generally have the formulae , where R is an alkyl or aryl group. Polysulfide salts and complexes The alkali metal polysulfides arise by treatment of a solution of the sulfide with elemental sulfur, e.g. sodium sulfide to sodium polysulfide: In some cases, these anions have been obtained as organic salts, which are soluble in organic solvents. The energy released in the reaction of sodium and elemental sulfur is the basis of battery technology. The sodium–sulfur battery and the lithium–sulfur battery require high temperatures to maintain liquid polysulfide and -conductive membranes that are unreactive toward sodium, sulfur, and sodium sulfide. Polysulfides are ligands in coordination ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium–sulfur Battery
The lithium–sulfur battery (Li–S battery) is a type of rechargeable battery. It is notable for its high specific energy. The low atomic weight of lithium and moderate atomic weight of sulfur means that Li–S batteries are relatively light (about the density of water). They were used on the longest and highest-altitude unmanned solar-powered aeroplane flight (at the time) by Airbus Zephyr, Zephyr 6 in August 2008. Lithium–sulfur batteries may displace lithium-ion cells because of their higher energy density and reduced cost. This is due to two factors. First the use of sulfur instead of a less energy dense and more expensive substances such as cobalt and/or iron compounds found in Lithium-ion battery, lithium-ion batteries. Secondly, the use of metallic lithium instead of intercalating lithium ions allows for much higher energy density, as less substances are needed to hold "lithium" and lithium is directly oxidized. Li–S batteries offer specific energies on the order of 5 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Polysulfide
Sodium polysulfide is a general term for salts with the formula sodium, Na2Sulfur, Sx, where ''x'' = 2 to 5. The species Sx2−, called polysulfide anions, include disulfide (S22−), trisulfide (S32−), tetrasulfide (S42−), and pentasulfide (S52−). In principle, but not in practice, the chain lengths could be longer. The salts are dark red solids that dissolve in water to give highly alkaline and corrosion, corrosive solutions. In air, these salts oxidize, and they evolve hydrogen sulfide by hydrolysis. Structure The polysulfide anions form chains with S---S bond distances around 2 Å in length. The chains adopt skewed conformations. In the solid state, these salts are dense solids with strong association of the sodium cations with the anionic termini of the chains. Production and occurrence Sodium polysulfide can be produced by dissolving sulfur in a solution of sodium sulfide. Alternatively they are produced by the redox reaction of aqueous sodium hydroxide with sulfu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium–sulfur Battery
A sodium–sulfur (NaS) battery is a type of molten-salt battery, molten-salt battery (electricity), battery that uses liquid sodium and liquid sulfur electrodes. This type of battery has a similar energy density to lithium-ion battery, lithium-ion batteries, and is fabricated from inexpensive and low-toxicity materials. Due to the high operating temperature required (usually between 300 and 350 °C), as well as the highly reactive nature of sodium and sodium polysulfides, these batteries are primarily suited for grid energy storage, stationary energy storage applications, rather than for use in vehicles. Molten Na-S batteries are scalable in size: there is a 1 MW microgrid support system on Santa Catalina Island (California), Catalina Island CA (USA) and a 50 MW/300 MWh system in Fukuoka, Kyushu, (Japan). In 2024, only one company (NGK Insulators) produced molten NaS batteries on a commercial scale. BASF Stationary Energy Storage GmbH, a wholly owned subsidiary of BASF SE, a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiokol (polymer)
''Thiokol'' is a trade mark for various organic polysulfide polymers. Thiokol polymers are used as an elastomer in seals and sealants. The distinction between the polymers first commercialized by the Thiokol Chemical Company and subsequent polysulfide materials is often unclear. The name Thiokol is a portmanteau of the Greek words for sulfur () and glue () Preparation and structure A variety of thiokols are recognized. Typically they are prepared by the combination of 2-chloroethanol, formaldehyde, and sodium polysulfide (Na2S''x''). The chloroethanol is produced in situ from ethylene oxide and hydrogen chloride. The rank ''x'' of the polysulfide is an important variable. Crosslinking agents are used, such as 1,2,3-trichloropropane. An idealized polymer is represented by this formula HS(CH2CH2OCH2OCH2CH2SS)''n''CH2CH2OCH2OCH2CH2SH. Thiol-terminated resins can be cured oxidatively. History In 1838, Swiss chemists reported the preparation of hydrophobic rubbery materials by the a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur
Sulfur ( American spelling and the preferred IUPAC name) or sulphur ( Commonwealth spelling) is a chemical element; it has symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with the chemical formula S8. Elemental sulfur is a bright yellow, crystalline solid at room temperature. Sulfur is the tenth most abundant element by mass in the universe and the fifth most common on Earth. Though sometimes found in pure, native form, sulfur on Earth usually occurs as sulfide and sulfate minerals. Being abundant in native form, sulfur was known in ancient times, being mentioned for its uses in ancient India, ancient Greece, China, and ancient Egypt. Historically and in literature sulfur is also called brimstone, which means "burning stone". Almost all elemental sulfur is produced as a byproduct of removing sulfur-containing contaminants from natural gas and petroleum.. Downloahere Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Sulfide
Sodium sulfide is a chemical compound with the formula Na2 S, or more commonly its hydrate Na2S·9 H2O. Both the anhydrous and the hydrated salts are colorless solids, although technical grades of sodium sulfide are generally yellow to brick red owing to the presence of polysulfides. It is commonly supplied as a crystalline mass, in flake form, or as a fused solid. They are water-soluble, giving strongly alkaline solutions. When exposed to moisture, Na2S immediately hydrates to give sodium hydrosulfide. Sodium sulfide has an unpleasant rotten egg smell due to the hydrolysis to hydrogen sulfide in moist air. Some commercial samples are described as Na2S·''x''H2O, where a weight percentage of Na2S is specified. Commonly available grades have around 60% Na2S by weight, which means that ''x'' is around 3. These grades of sodium sulfide are often marketed as 'sodium sulfide flakes'. These samples consist of NaSH, NaOH, and water. Structure The structures of sodium sulfides have bee ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polysulfane
A polysulfane is a chemical compound of formula , where ''n'' > 1 (although disulfane () is sometimes excluded). Compounds containing 2 – 8 sulfur atoms have been isolated, longer chain compounds have been detected, but only in solution.R. Steudel "Inorganic Polysulfanes H2S''n'' with ''n'' > 1" in Elemental Sulfur and Sulfur-Rich Compounds II (Topics in Current Chemistry) 2003, Volume 231, pp 99–125. is colourless, higher members are yellow with the colour increasing with the sulfur content. In the chemical literature the term polysulfanes is sometimes used for compounds containing , e.g. organic polysulfanes . Structures Polysulfanes consist of unbranched chains of sulfur atoms terminated with hydrogen atoms. The branched isomer of tetrasulfane , in which the fourth sulfur is bonded to the central sulfur, would be described as trithiosulfurous acid, . Computations suggests that it is less stable than the linear isomer . The S-S-S angles approach 90° in trisulfane and hig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfide
Sulfide (also sulphide in British English) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to large families of inorganic and organic compounds, e.g. lead sulfide and dimethyl sulfide. Hydrogen sulfide (H2S) and bisulfide (HS−) are the conjugate acids of sulfide. Chemical properties The sulfide ion does not exist in aqueous alkaline solutions of Na2S. Instead sulfide converts to hydrosulfide: :S2− + H2O → SH− + OH− Upon treatment with an acid, sulfide salts convert to hydrogen sulfide: :S2− + H+ → SH− :SH− + H+ → H2S Oxidation of sulfide is a complicated process. Depending on the conditions, the oxidation can produce elemental sulfur, polysulfides, polythionates, sulfite, or sulfate. Metal sulfides react with halogens, forming sulfur and metal salts. :8 MgS + 8 I2 → S8 + 8 MgI2 Metal de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Curing (chemistry)
Curing is a chemical process employed in polymer chemistry and process engineering that produces the toughening or hardening of a polymer material by cross-linking of polymer chains. Even if it is strongly associated with the production of thermosetting polymers, the term "curing" can be used for all the processes where a solid product is obtained from a liquid solution, such as with PVC plastisols. Curing process During the curing process, single monomers and oligomers, mixed with or without a curing agent, react to form a tridimensional polymeric network. In the very first part of the reaction Branching (polymer chemistry), branches of molecules with various architectures are formed, and their molecular weight increases in time with the extent of the reaction until the network size is equal to the size of the system. The system has lost its solubility and its viscosity tends to infinite. The remaining molecules start to coexist with the macroscopic network until they react wit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cross-link
In chemistry and biology, a cross-link is a bond or a short sequence of bonds that links one polymer chain to another. These links may take the form of covalent bonds or ionic bonds and the polymers can be either synthetic polymers or natural polymers (such as proteins). In polymer chemistry "cross-linking" usually refers to the use of cross-links to promote a change in the polymers' physical properties. When "crosslinking" is used in the biological field, it refers to the use of a probe to link proteins together to check for protein–protein interactions, as well as other creative cross-linking methodologies. Although the term is used to refer to the "linking of polymer chains" for both sciences, the extent of crosslinking and specificities of the crosslinking agents vary greatly. Synthetic polymers : 260px, left, Chemical reactions associated with crosslinking of drying oils, the process that produces curing'' refers to the crosslinking of thermosetting">linoleum. Cros ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rubber
Rubber, also called India rubber, latex, Amazonian rubber, ''caucho'', or ''caoutchouc'', as initially produced, consists of polymers of the organic compound isoprene, with minor impurities of other organic compounds. Types of polyisoprene that are used as natural rubbers are classified as elastomers. Currently, rubber is harvested mainly in the form of the latex from the Hevea brasiliensis, Pará rubber tree (''Hevea brasiliensis'') or others. The latex is a sticky, milky and white colloid drawn off by making incisions in the bark and collecting the fluid in vessels in a process called "tapping". Manufacturers refine this latex into the rubber that is ready for commercial processing. Natural rubber is used extensively in many applications and products, either alone or in combination with other materials. In most of its useful forms, it has a large stretch ratio and high resilience and also is buoyant and water-proof. Industrial demand for rubber-like materials began to out ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
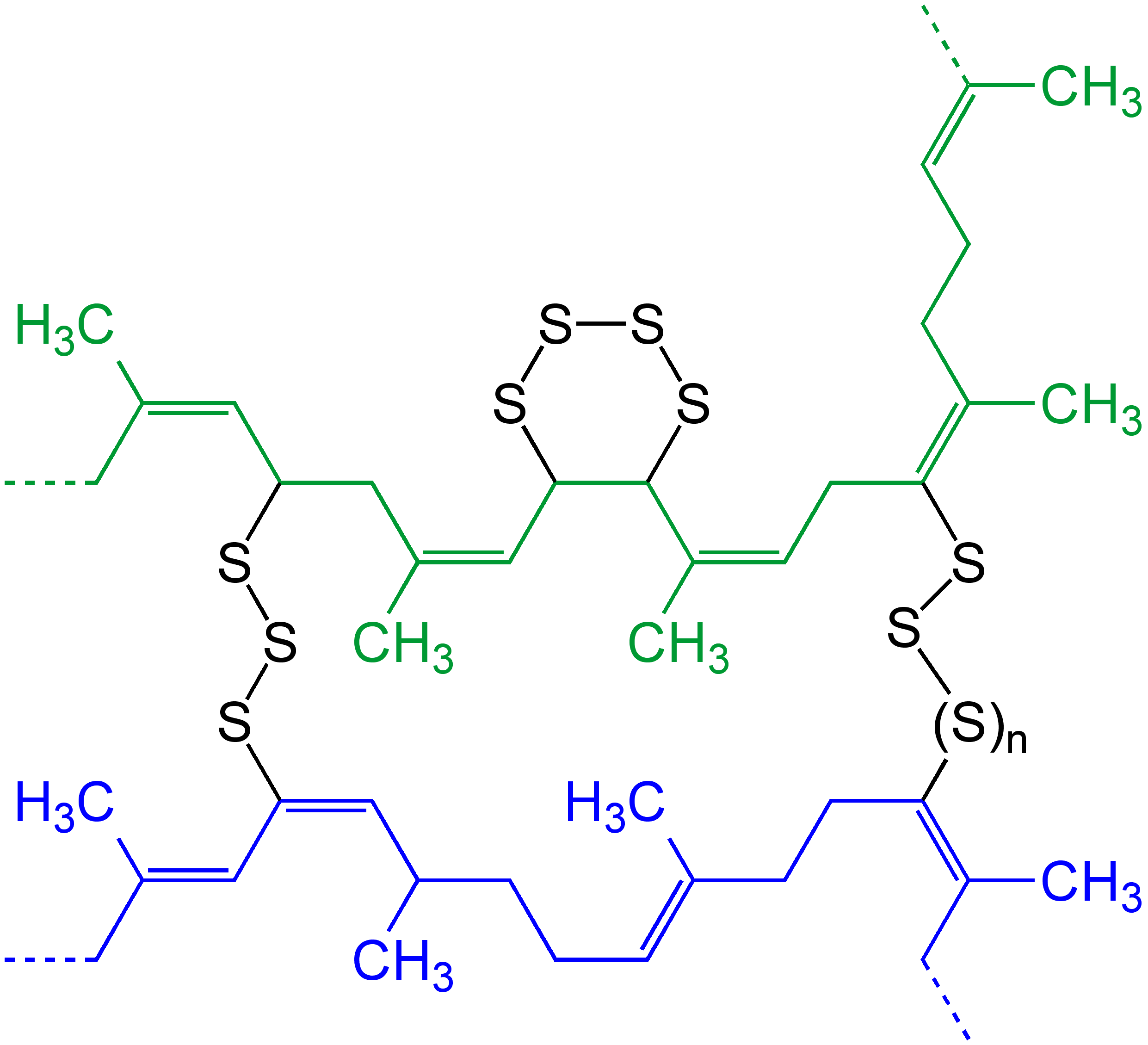
Vulcanization
Vulcanization (British English: vulcanisation) is a range of processes for hardening rubbers. The term originally referred exclusively to the treatment of natural rubber with sulfur, which remains the most common practice. It has also grown to include the hardening of other (synthetic) rubbers via various means. Examples include silicone rubber via room temperature vulcanizing and chloroprene rubber (neoprene) using metal oxides. Vulcanization can be defined as the curing of elastomers, with the terms 'vulcanization' and 'curing' sometimes used interchangeably in this context. It works by forming cross-links between sections of the polymer chain which results in increased rigidity and durability, as well as other changes in the mechanical and electrical properties of the material. Vulcanization, in common with the curing of other thermosetting polymers, is generally irreversible. The word was suggested by William Brockedon (a friend of Thomas Hancock who attained the B ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |