|
Phenanthrenequinone
Phenanthrenedione is a quinone derivative of a polycyclic aromatic hydrocarbon. It is an orange, water-insoluble solid. Laboratory synthesis and use It has been prepared by oxidation of phenanthrene with chromic acid. It is used as a artificial mediator for electron acceptor/donor in Mo/W containing formate dehydrogenase reduction of Carbon dioxide to formate and vice versa. It is a better electron acceptor than the natural Nicodinamide adenosine dinucleotide(NAD+). Safety It is a cytotoxic. References Quinones Phenanthrenes {{organic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polycyclic Aromatic Hydrocarbon
A polycyclic aromatic hydrocarbon (PAH) is a class of organic compounds that is composed of multiple aromatic rings. The simplest representative is naphthalene, having two aromatic rings and the three-ring compounds anthracene and phenanthrene. PAHs are uncharged, non-polar and planar. Many are colorless. Many of them are found in coal and in oil deposits, and are also produced by the combustion of organic matter—for example, in engines and incinerators or when biomass burns in forest fires. Polycyclic aromatic hydrocarbons are discussed as possible starting materials for abiotic syntheses of materials required by the earliest forms of life. Nomenclature and structure The terms polyaromatic hydrocarbon or polynuclear aromatic hydrocarbon are also used for this concept. By definition, polycyclic aromatic hydrocarbons have multiple rings, precluding benzene from being considered a PAH. Some sources, such as the US EPA and CDC, consider naphthalene to be the simplest PA ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
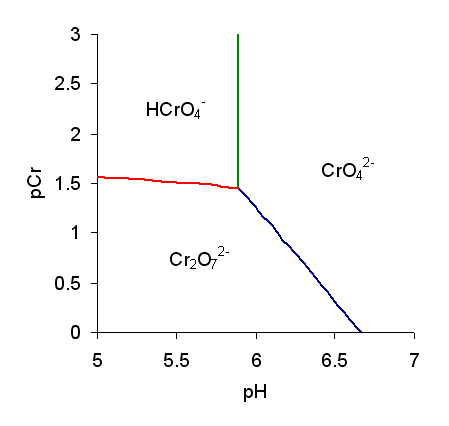
Chromic Acid
The term chromic acid is usually used for a mixture made by adding concentrated sulfuric acid to a dichromate, which may contain a variety of compounds, including solid chromium trioxide. This kind of chromic acid may be used as a cleaning mixture for glass. Chromic acid may also refer to the molecular species, H2CrO4 of which the trioxide is the anhydride. Chromic acid features chromium in an oxidation state of +6 (or VI). It is a strong and corrosive oxidising agent. Molecular chromic acid Molecular chromic acid, H2CrO4, has much in common with sulfuric acid, H2SO4. Only sulfuric acid can be classified as part of the 7 strong acids list. Due to the laws pertinent to the concept of "first order ionization energy", the first proton is lost most easily. It behaves extremely similar to sulfuric acid deprotonation. Since the process of polyvalent acid-base titrations have more than one proton (especially when the acid is starting substance and the base is the titrant), pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quinones
The quinones are a class of organic compounds that are formally "derived from aromatic compounds benzene.html" ;"title="uch as benzene">uch as benzene or naphthalene] by conversion of an even number of –CH= groups into –C(=O)– groups with any necessary rearrangement of double bonds, resulting in "a fully Conjugated system, conjugated cyclic diketone, dione structure". The archetypical member of the class is 1,4-benzoquinone or cyclohexadienedione, often called simply "quinone" (thus the name of the class). Other important examples are 1,2-benzoquinone (''ortho''-quinone), 1,4-naphthoquinone and 9,10-anthraquinone. The name is derived from that of quinic acid (with the suffix "-one" indicating a ketone), since it is one of the compounds obtained upon oxidation of quinic acid. Quinic acid, like quinine is obtained from cinchona bark, called quinaquina in the indigenous languages of Peruvian tribes. Properties Quinones are oxidized derivatives of aromatic compounds and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |