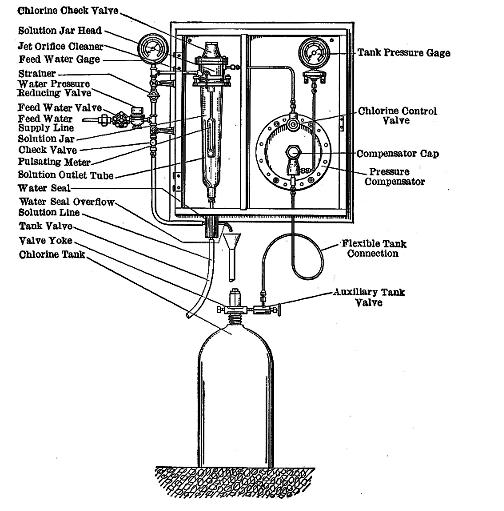
|
Disinfection Byproduct
Disinfection by-products (DBPs) result from chemical reactions between organic and inorganic matter in water with chemical treatment agents during the water disinfection process. Chlorination disinfection byproducts Chlorinated disinfection agents such as chlorine and monochloramine are strong oxidizing agents introduced into water in order to destroy pathogenic microbes, to oxidize taste/odor-forming compounds, and to form a disinfectant residual so water can reach the consumer tap safe from microbial contamination. These disinfectants may react with naturally present fulvic and humic acids, amino acids, and other natural organic matter, as well as iodide and bromide ions, to produce a range of DBPs such as the trihalomethanes (THMs), haloacetic acids (HAAs), bromate, and chlorite (which are regulated in the US), and so-called "emerging" DBPs such as halonitromethanes, haloacetonitriles, haloamides, halofuranones, iodo-acids such as iodoacetic acid, iodo-THMs ( iodotrihalo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Haloacetic Acids
Haloacetic acids are carboxylic acids in which a halogen atom takes the place of a hydrogen atom in acetic acid. Thus, in a monohaloacetic acid, a single halogen would replace a hydrogen atom. For example, chloroacetic acid would have the structural formula CH2ClCO2H. In the same manner, in dichloroacetic acid two chlorine atoms would take the place of two hydrogen atoms (CHCl2CO2H). The inductive effect caused by the electronegative halogens often result in the higher acidity of these compounds by stabilising the negative charge of the conjugate base. Contaminants in drinking water Haloacetic acids (HAAs) are a common undesirable by-product of drinking water chlorination. Exposure to such disinfection by-products in drinking water, at high levels over many years, has been associated with a number of health outcomes by epidemiological studies. In water, HAAs are stable, with the five most common being: * monochloroacetic acid (MCA) ClCH2COOH * dichloroacetic acid (DCA) Cl2CHCO ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the Atomic nucleus, nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive Chemical element, elements where both electronic and nuclear changes can occur. The substance (or substances) initially involved in a chemical reaction are called reagent, reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more Product (chemistry), products, which usually have properties different from the reactants. Reactions often consist of a sequence o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Furanone
2-Furanone is a heterocyclic organic compound. It is also known as γ-crotonolactone (GCL), as it is formally the lactone derived from γ-hydroxyisocrotonic acid. The chemical is colloquially called "butenolide", and is the parent structure for the butenolide class of compounds. It is a colourless liquid. Synthesis and reactions 2-Furanone is prepared by oxidation of furfural: : It exists in equilibrium with the tautomer 2-hydroxy furan, which serves as an intermediate in the interconversion between the β- and α-furanones. The β form is the more stable. The interconversion is catalyzed by base. 2-Furanones can be converted to furans by a two-step process of reduction followed by dehydration. See also * :Furanones, various substituted structural analogs * Pyrone Pyrones or pyranones are a class of heterocyclic chemical compounds. They contain an unsaturated six-membered ring containing one oxygen atom and a ketone functional group. There are two isomers denoted as 2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acute Toxicity
Acute toxicity describes the adverse effects of a substance that result either from a single exposure or from multiple exposures in a short period of time (usually less than 24 hours). To be described as ''acute'' toxicity, the adverse effects should occur within 14 days of the administration of the substance. Acute toxicity is distinguished from chronic toxicity, which describes the adverse health effects from repeated exposures, often at lower levels, to a substance over a longer time period (months or years). It is widely considered unethical to use humans as test subjects for acute (or chronic) toxicity research. However, some information can be gained from investigating accidental human exposures (e.g., factory accidents). Otherwise, most acute toxicity data comes from animal testing or, more recently, ''in vitro'' testing methods and inference from data on similar substances. Measures of acute toxicity Regulatory values Limits for short-term exposure, such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Developmental Toxicity
Development of the human body is the process of growth to maturity. The process begins with fertilization, where an egg released from the ovary of a female is penetrated by a sperm cell from a male. The resulting zygote develops through mitosis and cell differentiation, and the resulting embryo then implants in the uterus, where the embryo continues development through a fetal stage until birth. Further growth and development continues after birth, and includes both physical and psychological development, influenced by genetic, hormonal, environmental and other factors. This continues throughout life: through childhood and adolescence into adulthood. Before birth Development before birth, or prenatal development () is the process in which a zygote, and later an embryo and then a fetus develops during gestation. Prenatal development starts with fertilization and the formation of the zygote, the first stage in embryonic development which continues in fetal develo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine Dioxide
Chlorine dioxide is a chemical compound with the formula ClO2 that exists as yellowish-green gas above 11 °C, a reddish-brown liquid between 11 °C and −59 °C, and as bright orange crystals below −59 °C. It is usually handled as an aqueous solution. It is also commonly used as a bleach. More recent developments have extended its applications in food processing and as a disinfectant. Structure and bonding The molecule ClO2 has an odd number of valence electrons, and therefore, it is a paramagnetic radical. It is an unusual "example of an odd-electron molecule which is stable towards dimerization" ( nitric oxide being another example). In 1933, Lawrence O. Brockway, a graduate student of Linus Pauling, proposed a structure that involved a three-electron bond and two single bonds. However, Pauling in his ''General Chemistry'' shows a double bond to one oxygen and a single bond plus a three-electron bond to the other. The valence bond structur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloramination
Chloramination is the treatment of drinking water with a chloramine disinfectant. Both chlorine and small amounts of ammonia are added to the water one at a time which react together to form chloramine (also called combined chlorine), a long lasting disinfectant. Chloramine disinfection is used in both small and large water treatment plants. Use In the United States, the maintenance of what is called a "residual" of disinfectant that stays in the water distribution system while it is delivered to people's homes is required by the Environmental Protection Agency (EPA). The EPA regulations give two choices for disinfectant residual — chlorine or chloramine. Many major water agencies are changing to chloramine to better meet current and anticipated federal drinking water regulations and to protect the public health. Chlorine versus chloramine There are many similarities between chlorine and chloramine. Both provide effective residual disinfection with minimal risk to public ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water Chlorination
Water chlorination is the process of adding chlorine or chlorine compounds such as sodium hypochlorite to water. This method is used to kill bacteria, viruses and other microbes in water. In particular, chlorination is used to prevent the spread of waterborne diseases such as cholera, dysentery, and typhoid. History In a paper published in 1894, it was formally proposed to add chlorine to water to render it "germ-free". Two other authorities endorsed this proposal and published it in many other papers in 1895. Early attempts at implementing water chlorination at a water treatment plant were made in 1893 in Hamburg, Germany. In 1897 the town of Maidstone, England was the first to have its entire water supply treated with chlorine. Permanent water chlorination began in 1905, when a faulty slow sand filter and a contaminated water supply caused a serious typhoid fever epidemic in Lincoln, England. Alexander Cruickshank Houston used chlorination of the water to stop the epidemic. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lower atmosphere to (dioxygen). Ozone is formed from dioxygen by the action of ultraviolet (UV) light and electrical discharges within the Earth's atmosphere. It is present in very low concentrations throughout the latter, with its highest concentration high in the ozone layer of the stratosphere, which absorbs most of the Sun's ultraviolet (UV) radiation. Ozone's odour is reminiscent of chlorine, and detectable by many people at concentrations of as little as in air. Ozone's O3 structure was determined in 1865. The molecule was later proven to have a bent structure and to be weakly diamagnetic. In standard conditions, ozone is a pale blue gas that condenses at cryogenic temperatures to a dark blue liquid and finally a violet-black soli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloroform
Chloroform, or trichloromethane, is an organic compound with chemical formula, formula Carbon, CHydrogen, HChlorine, Cl3 and a common organic solvent. It is a colorless, strong-smelling, dense liquid produced on a large scale as a precursor to PTFE. It is also a precursor to various refrigerants. It is trihalomethane. It is a powerful anesthetic, euphoriant, anxiolytic, and sedative when inhaled or ingested. Structure The molecule adopts a tetrahedral molecular geometry with C3v symmetry group, symmetry. Natural occurrence The total global flux of chloroform through the environment is approximately tonnes per year, and about 90% of emissions are natural in origin. Many kinds of seaweed produce chloroform, and fungi are believed to produce chloroform in soil. Abiotic processes are also believed to contribute to natural chloroform productions in soils although the mechanism is still unclear. Chloroform volatilizes readily from soil and surface water and undergoes degradation in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Genotoxicity
Genotoxicity is the property of chemical agents that damage the genetic information within a cell causing mutations, which may lead to cancer. While genotoxicity is often confused with mutagenicity, all mutagens are genotoxic, but some genotoxic substances are not mutagenic. The alteration can have direct or indirect effects on the DNA: the induction of mutations, mistimed event activation, and direct DNA damage leading to mutations. The permanent, heritable changes can affect either somatic cells of the organism or germ cells to be passed on to future generations. Cells prevent expression of the genotoxic mutation by either DNA repair or apoptosis; however, the damage may not always be fixed leading to mutagenesis. To assay for genotoxic molecules, researchers assay for DNA damage in cells exposed to the toxic substrates. This DNA damage can be in the form of single- and double-strand breaks, loss of excision repair, cross-linking, alkali-labile sites, point mutations, and structura ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |