|
Dispersion Stabilized Molecules
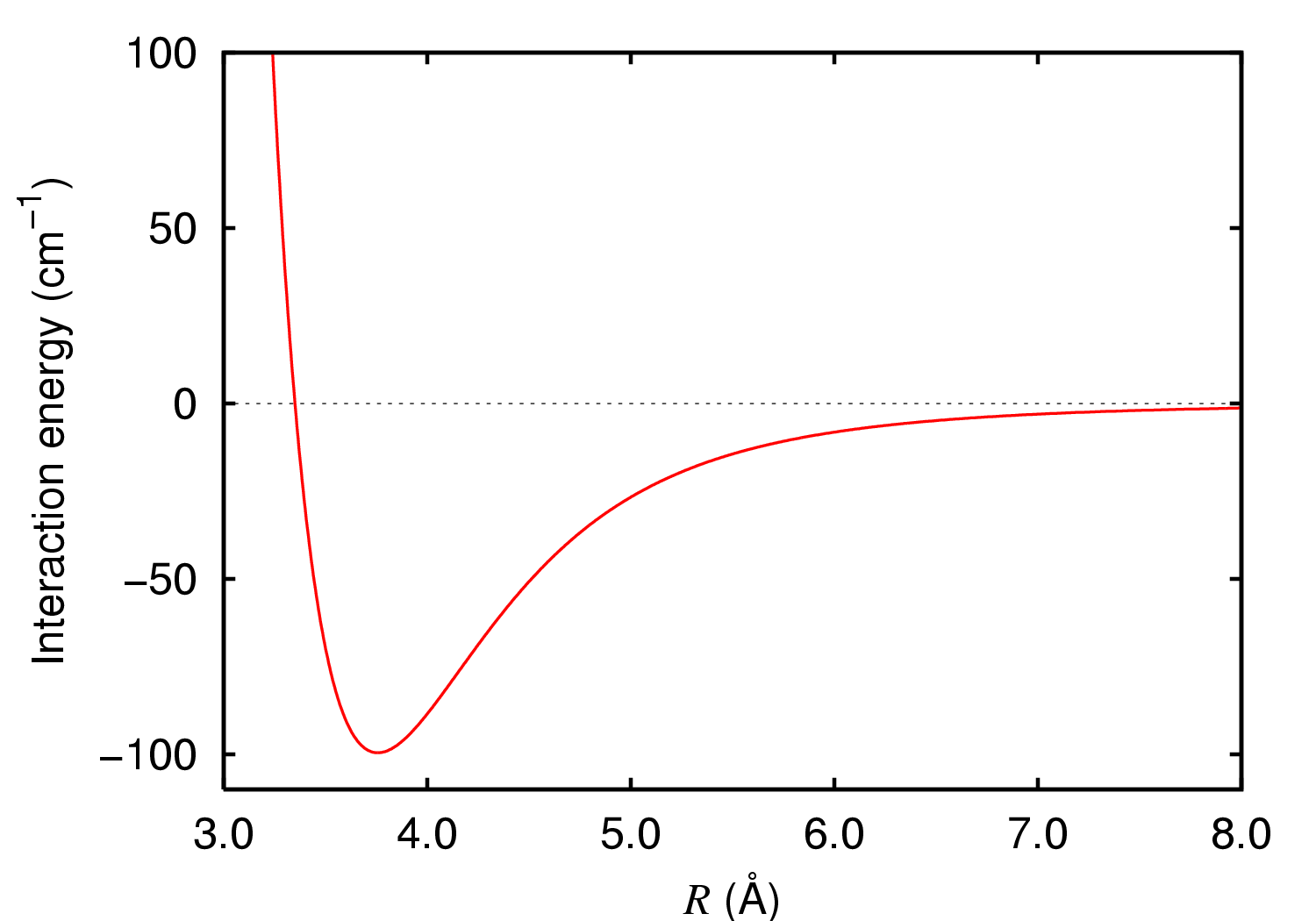
Dispersion stabilized molecules are molecules where the London dispersion force (LDF), a non-covalent attractive force between atoms and molecules, plays a significant role in promoting the molecule's stability. Distinct from Steric effects, steric hindrance, dispersion stabilization has only recently been considered in depth by organic and inorganic chemists after earlier gaining prominence in Protein folding, protein science and supramolecular chemistry. Although usually weaker than covalent bonding and other forms of non-covalent interactions like hydrogen bonding, dispersion forces are known to be a significant if not dominating stabilizing force in certain organic, inorganic, and main group molecules, stabilizing otherwise reactive moieties and exotic bonding. Stabilization through dispersion Dispersion interactions are a stabilizing force arising from quantum mechanical electron correlation. Although quantum mechanical in nature, the energy of dispersion interactions can be a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
London Dispersion Force
London dispersion forces (LDF, also known as dispersion forces, London forces, instantaneous dipole–induced dipole forces, fluctuating induced dipole bonds or loosely as van der Waals forces) are a type of intermolecular force acting between atoms and molecules that are normally electrically symmetric; that is, the electrons are symmetrically distributed with respect to the nucleus. They are part of the van der Waals forces. The LDF is named after the German physicist Fritz London. They are the weakest of the intermolecular forces. Introduction The electron distribution around an atom or molecule undergoes fluctuations in time. These fluctuations create instantaneous electric fields which are felt by other nearby atoms and molecules, which in turn adjust the spatial distribution of their own electrons. The net effect is that the fluctuations in electron positions in one atom induce a corresponding redistribution of electrons in other atoms, such that the electron motions be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gomberg's Dimer
Gomberg's dimer is the organic compound with the formula Ph2C=C6H5-CPh3, where Ph = phenyl, C6H5. It is a yellow solid that is air-stable for hours at room temperature and soluble in organic solvents. The compound achieved fame as the Dimer (chemistry), dimer of triphenylmethyl radical, which was prepared by Moses Gomberg in his quest for hexaphenylethane. Its quinoid structure has been determined by X-ray crystallography. The C-C bond that reversibly breaks is rather long at 159.7 picometers. Synthesis and reactions Gomberg's dimer can be prepared quantitatively by treating trityl bromide with powdered copper or silver: :2Ph3CBr + 2Cu → Ph2C=C6H5-CPh3 + 2CuBr Gomberg's dimer reversibly dissociates to the triphenylmethyl radical in organic solvents: : See also * Hexaphenylethane References {{reflist Aromatic hydrocarbons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
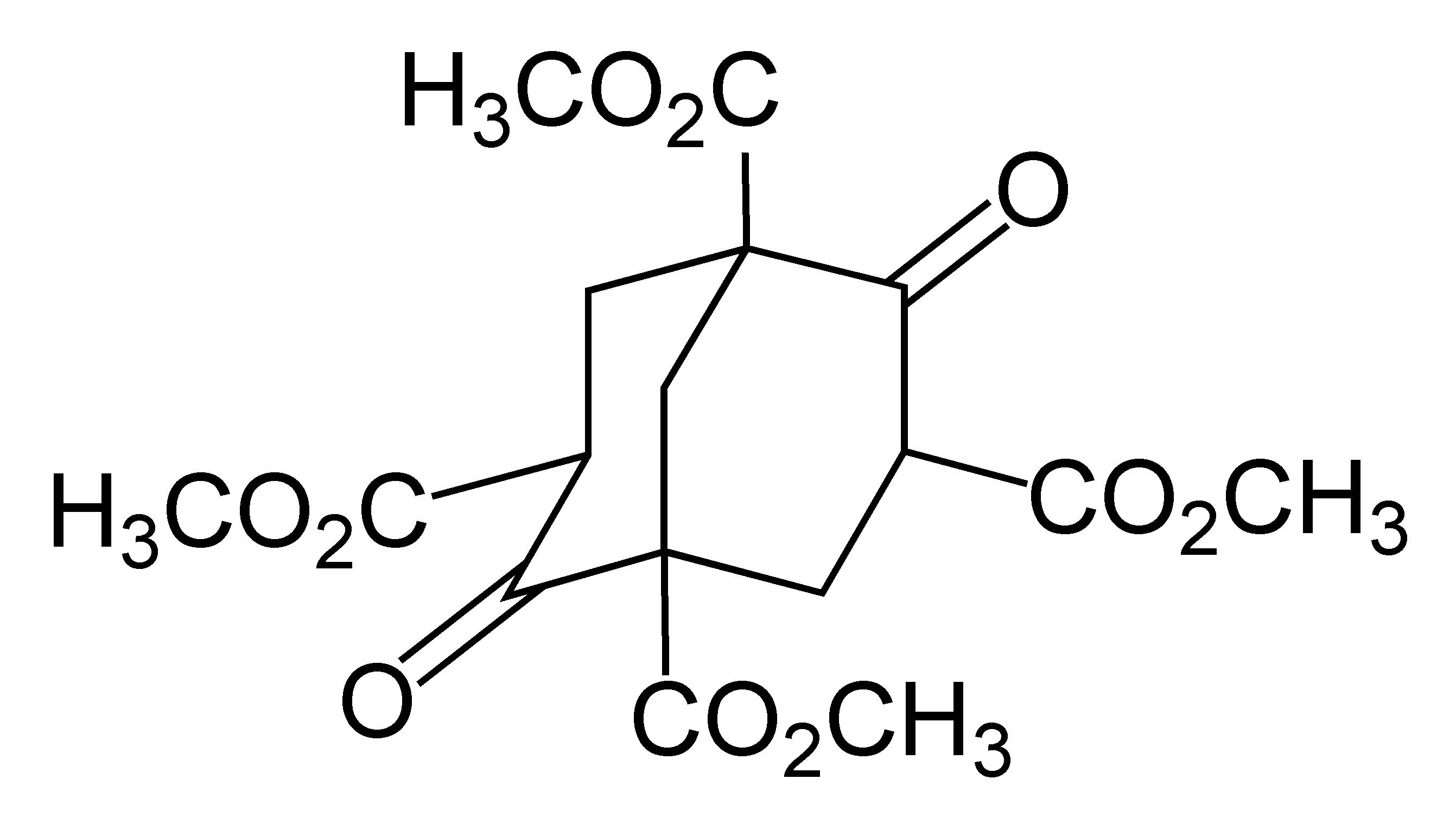
Metal Tetranorbornyl
In organometallic chemistry, metal tetranorbornyls are compounds with the formula M(nor)4 (M = a metal in a +4 oxidation state) (1-nor = 4bicyclo[2.2.1]hept-1-yl) and are one of the largest series of tetraalkyl complexes derived from identical ligands. Metal tetranorbornyls display uniform stoichiometry, Low spin, low-spin configurations, and high Stability constants of complexes, stability, which can be attributed to their +4 oxidation state metal center. The stability of metal tetranorbornyls is predominately considered to be derived from the unfavorable Beta-Hydride elimination, β-hydride elimination. Computational calculations have determined that London dispersion force, London dispersion effects significantly contribute to the stability of metal tetranorbornyls. Specifically, Fe(nor)4 has a stabilization of 45.9 kcal/mol−1. Notable metal tetranorbornyls are those synthesized with metal centers of cobalt, manganese, or iron. Preparation Traditionally, metal tetranorbor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Disproportionation
In chemistry, disproportionation, sometimes called dismutation, is a redox reaction in which one compound of intermediate oxidation state converts to two compounds, one of higher and one of lower oxidation state. The reverse of disproportionation, such as when a compound in an intermediate oxidation state is formed from precursors of lower and higher oxidation states, is called ''comproportionation'', also known as ''symproportionation''. More generally, the term can be applied to any desymmetrizing reaction where two molecules of one type react to give one each of two different types: : This expanded definition is not limited to redox reactions, but also includes some molecular autoionization reactions, such as the self-ionization of water. In contrast, some authors use the term ''redistribution'' to refer to reactions of this type (in either direction) when only ligand exchange but no redox is involved and distinguish such processes from disproportionation and comproportionati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
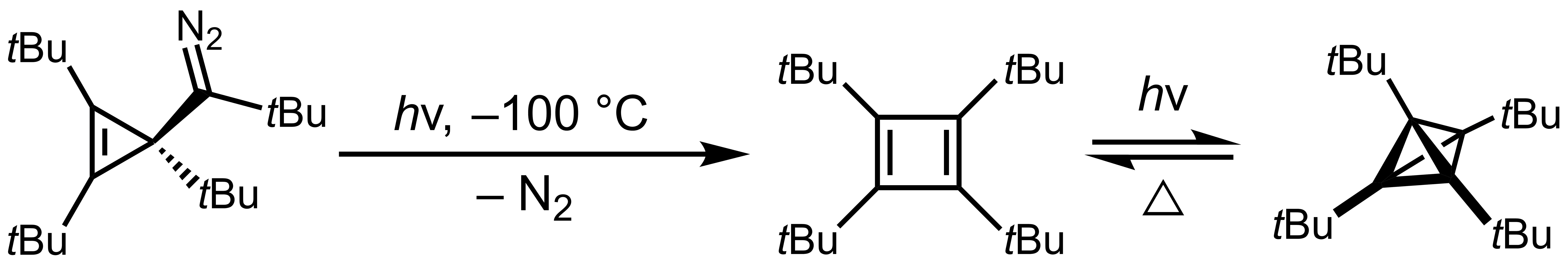
Tetrahedrane
Tetrahedrane is a hypothetical platonic hydrocarbon with chemical formula and a tetrahedral structure. The molecule would be subject to considerable angle strain and has not been synthesized . However, a number of derivatives have been prepared. In a more general sense, the term ''tetrahedranes'' is used to describe a class of molecules and ions with related structure, e.g. white phosphorus. C4 tetrahedranes Tetrahedrane () is one of the possible platonic hydrocarbons and has the IUPAC name tricyclo .1.0.02,4utane. Unsubstituted tetrahedrane remains elusive, although predicted kinetically stable. One strategy that has been explored (but thus far failed) is reaction of propene with atomic carbon. Contrariwise, several organic compounds with the tetrahedrane core are known. All have multiply bulky substituents, ''tert''-butyl (''t''-Bu) or larger. Locking a tetrahedrane molecule inside a fullerene has only been attempted ''in silico''. All known syntheses have ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron Diffraction
Neutron diffraction or elastic neutron scattering is the application of neutron scattering to the determination of the atomic and/or magnetic structure of a material. A sample to be examined is placed in a beam of Neutron temperature, thermal or cold neutron radiation, neutrons to obtain a diffraction pattern that provides information of the structure of the material. The technique is similar to X-ray diffraction but due to their different scattering properties, neutrons and X-rays provide complementary information: X-Rays are suited for superficial analysis, strong x-rays from synchrotron radiation are suited for shallow depths or thin specimens, while neutrons having high penetration depth are suited for bulk samples.Measurement of residual stress in materials using neutrons IAEA, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adamantyl
Adamantane is an organic compound with formula C10H16 or, more descriptively, (CH)4(CH2)6. Adamantane molecules can be described as the fusion of three cyclohexane rings. The molecule is both rigid and virtually stress-free. Adamantane is the most stable isomer of C10H16. The spatial arrangement of carbon atoms in the adamantane molecule is the same as in the diamond crystal. This similarity led to the name ''adamantane'', which is derived from the Greek ''adamantinos'' (relating to steel or diamond). It is a white solid with a camphor-like odor. It is the simplest diamondoid. The discovery of adamantane in petroleum in 1933 launched a new field of chemistry dedicated to the synthesis and properties of polyhedral organic compounds. Adamantane derivatives have found practical application as drugs, polymeric materials, and thermally stable lubricants. History and synthesis In 1924, H. Decker suggested the existence of adamantane, which he called decaterpene. The first attempted ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclohexyl
Cyclohexane is a cycloalkane with the molecular formula . Cyclohexane is non-polar. Cyclohexane is a colourless, flammable liquid with a distinctive detergent-like odor, reminiscent of cleaning products (in which it is sometimes used). Cyclohexane is mainly used for the industrial production of adipic acid and caprolactam, which are precursors to nylon. Cyclohexyl () is the alkyl substituent of cyclohexane and is abbreviated Cy. Production Cyclohexane is one of components of naphtha, from which it can be extracted by advanced distillation methods. Distillation is usually combined with isomerization of methylcyclopentane, a similar component extracted from naphtha by similar methods. Together, these processes cover only a minority (15-20%) of the modern industrial demand, and are complemented by synthesis. Modern industrial synthesis On an industrial scale, cyclohexane is produced by hydrogenation of benzene in the presence of a Raney nickel catalyst. Producers of c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |