|
Disilene
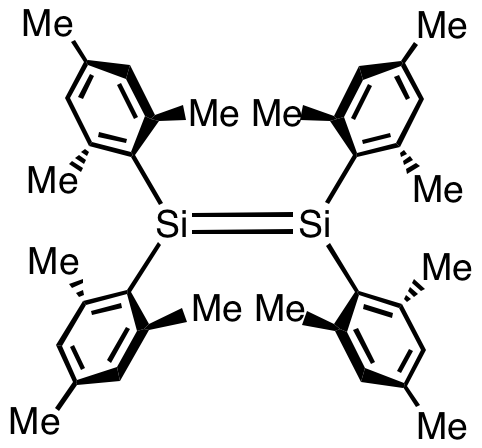
Disilene is an inorganic compound with the chemical formula . The name ''disilene'', referring to the structure of a particular prototropic tautomer of the molecule. It is the simplest silenes, silene. Properties and bonding Disilene is a molecule with one Si=Si bond and four equivalent Si-H bonds. Its structure is like that of ethylene if ethylene's carbon atoms were replaced by silicon. However, unlike ethylene, disilene is kinetically unstable with respect to tautomerisation. Disilene has two other tautomers, that are very close in energy: (μ2-''H'')disilene, and disilanylidene. Organodisilenes Disilenes bearing sterically bulky substituents have been well characterized although they remain mainly of academic interest. The first stabilised disilene was tetramesityldisilene, (C6Me3H2)4Si2. The Si=Si distance in this molecule is 2.15 Å, about 10% shorter than a typical Si–Si single bond. The Si2C4 core is roughly planar. Such species are typically prepared by reduction of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silenes
In inorganic chemistry, silenes, or disilalkenes,Philip P. Power "pi-Bonding and the Lone Pair Effect in Multiple Bonds between Heavier Main Group Elements" Chemical Reviews, 1999, 99, 3462. are silicon compounds that contain double bonds, where the oxidation state of Si is +2. The parent molecule is disilene, Si2H4. Structure The first transient disilene was reported in 1972 by D. N. Roark and Garry J. D. Peddle. Simple disilenes easily polymerize. To suppress this tendency, bulky substituents are used. Indeed the first isolable disilene, tetramesityldisilene, was described in 1981 by West, Fink, and Michl. It was prepared by UV-photolysis of the related cyclic trisilane: : 2 i(mesityl)2sub>3 → 3 (mesityl)2Si=Si(mesityl)2 Structure of tetramesityldisilene Tetramesityldisilene is a yellow-orange solid. The Si=Si double bond lengths of disilenes vary between 2.14 and 2.29 Å and are nearly 5 to 10% shorter than the Si-Si single bond lengths of corresponding disilane ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Disilyne
Disilyne is a low valent silicon compound with the chemical formula Si2R2 where oxidation state of Si is +1. Several isomers are possible, but none are sufficiently stable to be of practical value. Substituent, Substituted disilynes contain a formal silicon–silicon triple bond and as such are sometimes written R2Si2 (where R is a substituent group). They are the silicon analogues of alkynes. The term ''silyne'' has two diverse meanings. Some chemists use it to refer to compounds containing a silicon–silicon triple bond,Egon Wiberg, Arnold Frederick Holleman (2001) ''Inorganic Chemistry'', Elsevier by analogy to the carbon–carbon triple bond in alkynes, whereas others use the term to refer to compounds containing a silicon–carbon triple bond by analogy to silene, which often refers to compounds containing silicon–carbon double bonds. The term polysilyne can refer to the layer polymer (SiH)n or substituted derivatives. Substituted disilynes The first substituted disi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bond Length
In molecular geometry, bond length or bond distance is defined as the average distance between Atomic nucleus, nuclei of two chemical bond, bonded atoms in a molecule. It is a Transferability (chemistry), transferable property of a bond between atoms of fixed types, relatively independent of the rest of the molecule. Explanation Bond length is related to bond order: when more electrons participate in bond formation the bond is shorter. Bond length is also inversely related to bond strength and the bond dissociation energy: all other factors being equal, a stronger bond will be shorter. In a bond between two identical atoms, half the bond distance is equal to the covalent radius. Bond lengths are measured in the solid phase by means of X-ray diffraction, or approximated in the gas phase by microwave spectroscopy. A bond between a given pair of atoms may vary between different molecules. For example, the carbon to hydrogen bonds in methane are different from those in methyl chlori ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Disilane
Disilane is a chemical compound with general chemical formula Si2R6 that was first identified in 1902 by Henri Moissan and Samuel Smiles (1877–1953) where R = H. Moissan and Smiles reported disilane as being among the products formed by the action of dilute acids on metal silicides. Although these reactions had been previously investigated by Friedrich Woehler and Heinrich Buff between 1857 and 1858, Moissan and Smiles were the first to explicitly identify disilane. They referred to disilane as ''silicoethane''. Higher members of the homologous series formed in these reactions were subsequently identified by Carl Somiesky (sometimes spelled "Karl Somieski") and Alfred Stock. At standard temperature and pressure, disilane is a colourless, acrid gas. Disilane and ethane have similar structures, although disilane is much more reactive. Other compounds of the general formula (X = hydrogen, halogen, alkyl, aryl, and mixtures of these groups) are called disilanes. Disilane is a group ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Strain Energy
In physics, the elastic potential energy gained by a wire during elongation with a tensile (stretching) or compressive (contractile) force is called strain energy. For linearly elastic materials, strain energy is: : U = \frac 1 2 V \sigma \varepsilon = \frac 1 2 V E \varepsilon^2 = \frac 1 2 \frac V E \sigma^2 where is stress, is strain, is volume, and is Young's modulus: : E = \frac \sigma \varepsilon Molecular strain In a molecule, strain energy is released when the constituent atoms are allowed to rearrange themselves in a chemical reaction.''March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure,'' Michael B. Smith & Jerry March, Wiley-Interscience, 5th edition, 2001, The external work done on an elastic member in causing it to distort from its unstressed state is transformed into strain energy which is a form of potential energy. The strain energy in the form of elastic deformation is mostly recoverable in the form of mechanical work. For exa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Xylene
In organic chemistry, xylene or xylol (; IUPAC name: dimethylbenzene) are any of three organic compounds with the formula . They are derived from the substitution of two hydrogen atoms with methyl groups in a benzene ring; which hydrogens are substituted determines which of three structural isomers results. It is a colorless, flammable, slightly greasy liquid of great industrial value. The mixture is referred to as both xylene and, more precisely, xylenes. Mixed xylenes refers to a mixture of the xylenes plus ethylbenzene. The four compounds have identical molecular formulas . Typically the four compounds are produced together by various catalytic reforming and pyrolysis methods. Occurrence and production Xylenes are an important petrochemical produced by catalytic reforming and also by coal carbonisation in the manufacture of coke fuel. They also occur in crude oil in concentrations of about 0.5–1%, depending on the source. Small quantities occur in gasoline and aircra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isomerization
In chemistry, isomerization or isomerisation is the process in which a molecule, polyatomic ion or molecular fragment is transformed into an isomer with a different chemical structure. Enolization is an example of isomerization, as is tautomerization. When the activation energy for the isomerization reaction is sufficiently small, both isomers can often be observed and the equilibrium ratio will shift in a temperature-dependent equilibrium with each other. Many values of the standard free energy difference, \Delta G^\circ, have been calculated, with good agreement between observed and calculated data. Examples and applications Alkanes Skeletal isomerization occurs in the cracking process, used in the petrochemical industry to convert straight chain alkanes to isoparaffins as exemplified in the conversion of normal octane to 2,5-dimethylhexane (an "isoparaffin"): : Fuels containing branched hydrocarbons are favored for internal combustion engines for their higher octan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
X-ray Diffraction
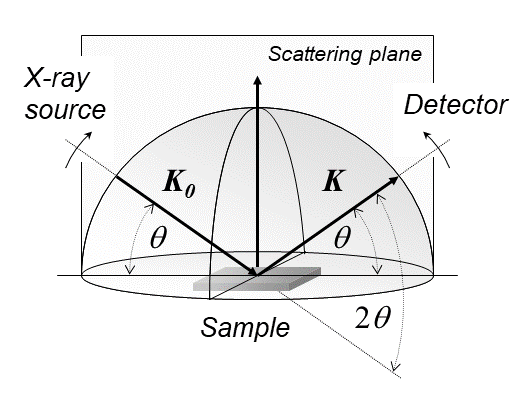
X-ray diffraction is a generic term for phenomena associated with changes in the direction of X-ray beams due to interactions with the electrons around atoms. It occurs due to elastic scattering, when there is no change in the energy of the waves. The resulting map of the directions of the X-rays far from the sample is called a diffraction pattern. It is different from X-ray crystallography which exploits X-ray diffraction to determine the arrangement of atoms in materials, and also has other components such as ways to map from experimental diffraction measurements to the positions of atoms. This article provides an overview of X-ray diffraction, starting with the early #History, history of x-rays and the discovery that they have the right spacings to be diffracted by crystals. In many cases these diffraction patterns can be #Introduction to x-ray diffraction theory, Interpreted using a single scattering or kinematical theory with conservation of energy (#Ewald's sphere, wave vecto ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Picometer
The picometre (international spelling as used by the International Bureau of Weights and Measures; SI symbol: pm) or picometer ( American spelling) is a unit of length in the International System of Units (SI), equal to , or one trillionth of a metre, which is the SI base unit of length. The picometre is one thousand femtometres, one thousandth of a nanometre ( nm), one millionth of a micrometre (also known as a micron), one billionth of a millimetre, and one trillionth of a metre. The symbol μμ was once used for it. It is also one hundredth of an ångström, an internationally known (but non-SI) unit of length. Use The picometre's length is of an order so small that its application is almost entirely confined to particle physics, quantum physics, chemistry, and acoustics. Atoms are between 62 and 520 pm in diameter, and the typical length of a carbon–carbon single bond is 154 pm. Smaller units still may be used to describe smaller particles (some of which ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inorganic Compound
An inorganic compound is typically a chemical compound that lacks carbon–hydrogen bondsthat is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as ''inorganic chemistry''. Inorganic compounds comprise most of the Earth's crust, although the compositions of the deep Mantle (geology), mantle remain active areas of investigation. All allotropes (structurally different pure forms of an element) and some simple carbon compounds are often considered inorganic. Examples include the allotropes of carbon (graphite, diamond, buckminsterfullerene, graphene, etc.), carbon monoxide , carbon dioxide , carbides, and salt (chemistry), salts of inorganic anions such as carbonates, cyanides, cyanates, thiocyanates, isothiocyanates, etc. Many of these are normal parts of mostly organic systems, including organisms; describing a chemical as inorganic does not necessarily mean that it cannot occur within life, living things. History ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Graphite
In the area of solid state chemistry, graphite intercalation compounds are a family of materials prepared from graphite. In particular, the sheets of carbon that comprise graphite can be pried apart by the insertion ( intercalation) of ions. The graphite is viewed as a host and the inserted ions as guests. The materials have the formula where ''n'' ≥ 6. The insertion of the guests increases the distance between the carbon sheets. Common guests are reducing agents such as alkali metals. Strong oxidants also intercalate into graphite. Intercalation involves electron transfer into or out of the carbon sheets. So, in some sense, graphite intercalation compounds are salts. Intercalation is often reversible: the inserted ions can be removed and the sheets of carbon collapse to a graphite-like structure. The properties of graphite intercalation compounds differ from those of the parent graphite. Preparation and structure These materials are prepared by treating graphite with ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |