|
Dimethyldioxirane
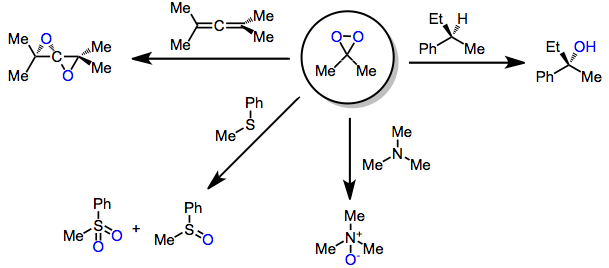
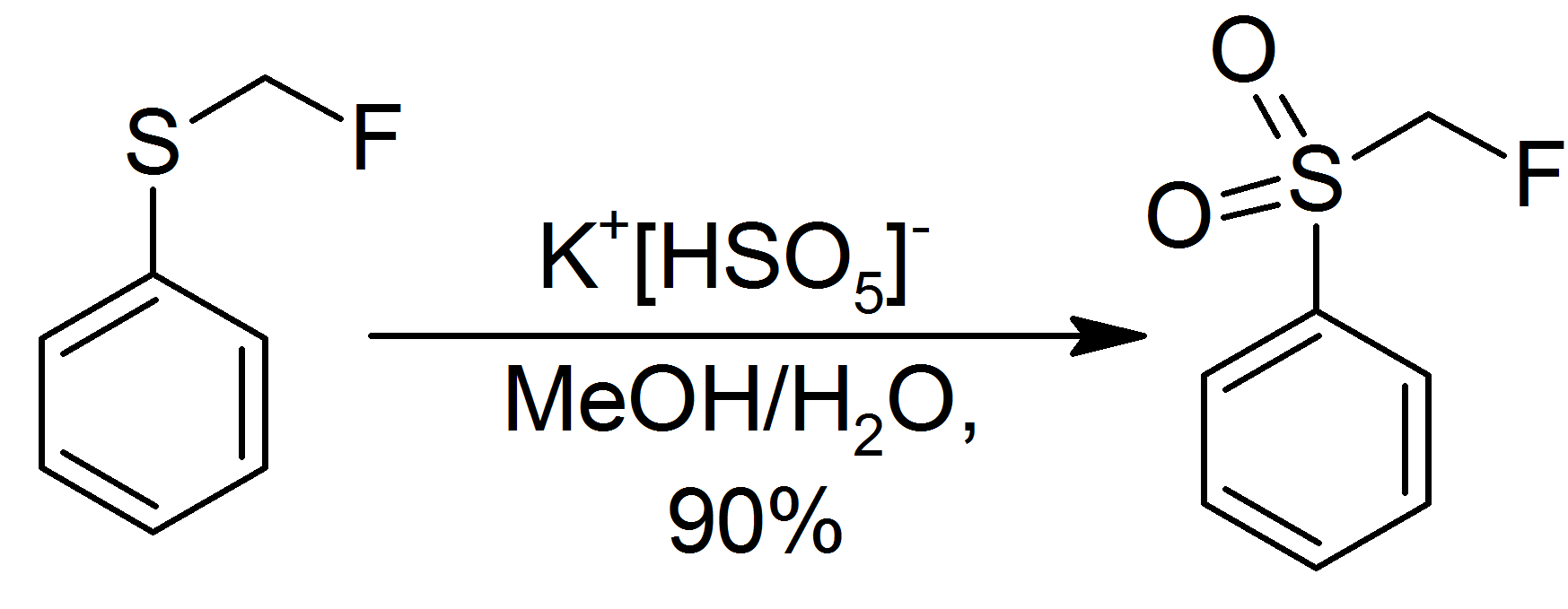
Dimethyldioxirane (DMDO) is the organic compound with the formula . It is the dioxirane derived from acetone and can be viewed as the monomer of acetone peroxide. It is a powerful selective oxidizing agent that finds some use in organic synthesis. It is known only in the form of a dilute solution, usually in acetone, and hence the properties of the pure material are largely unknown. Synthesis DMDO is not commercially available because of chemical instability. DMDO can be prepared as dilute solutions (~0.1 M) by treatment of acetone with potassium peroxymonosulfate , usually in the form of Oxone (2KHSO5·KHSO4·K2SO4). : The preparation of DMDO is rather inefficient (typical yields < 3%) and typically only yields a relatively dilute solution in acetone (only up to approximately 0.1 M). This is tolerable as preparation uses inexpensive substances: acetone, sodium bicarbonate, and oxone. Cold solutions (−10 to −20 °C) of DMDO are stable for days. Decomposition is accelerat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dioxiranes
In chemistry, dioxirane (systematically named dioxacyclopropane, also known as methylene peroxide or peroxymethane) is an organic compound with formula . The molecule consists of a ring with one Methylene group, methylene and two oxygen atoms. It is of interest as the smallest cyclic organic peroxide, but otherwise it is of little practical value. Synthesis Dioxirane is highly unstable and the majority of studies of it have been computational chemistry, computational; it has been detected during the low temperature (–196 °C) reaction of ethylene and ozone, although even at these temperatures such a mixture can be explosive. Its formation is thought to be radical in nature, preceding via a Criegee intermediate. Microwave analysis has indicated C-H, C-O and O-O bond lengths of 1.090, 1.388 and 1.516 Å respectively. The very long and weak O-O bond (cf. hydrogen peroxide O-O = 1.47 Å) is the origin of its instability. Other dioxiranes Beyond the parent dioxirane, which i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethyldioxirane Synthesis V
Dimethyldioxirane (DMDO) is the organic compound with the formula . It is the dioxirane derived from acetone and can be viewed as the monomer of acetone peroxide. It is a powerful selective oxidizing agent that finds some use in organic synthesis. It is known only in the form of a dilute solution, usually in acetone, and hence the properties of the pure material are largely unknown. Synthesis DMDO is not commercially available because of chemical instability. DMDO can be prepared as dilute solutions (~0.1 M) by treatment of acetone with potassium peroxymonosulfate , usually in the form of Oxone (2KHSO5·KHSO4·K2SO4). : The preparation of DMDO is rather inefficient (typical yields < 3%) and typically only yields a relatively dilute solution in acetone (only up to approximately 0.1 M). This is tolerable as preparation uses inexpensive substances: , |
Epoxidation With Dioxiranes
Oxidation with dioxiranes refers to the introduction of oxygen into organic substrates using dioxiranes. Dioxiranes are well known for epoxidations (synthesis of epoxides from alkenes). Dioxiranes oxidize other unsaturated functionality, heteroatoms, and alkane C-H bonds. Dioxiranes are metal-free oxidants. Epoxidations Dioxiranes are electrophilic oxidants that react more quickly with electron-rich than electron-poor double bonds; however, both classes of substrates can be epoxidized within a reasonable time frame. The mechanism of epoxidation with dioxiranes likely involves concerted oxygen transfer through a spiro transition state. As oxygen transfer occurs, the plane of the oxirane is perpendicular to and bisects the plane of the alkene pi system. The configuration of the alkene is maintained in the product, ruling out long-lived radical intermediates. In addition, the spiro transition state has been used to explain the selectivity in enantioselective epoxidations with chiral ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dioxirane Oxidations
In chemistry, dioxirane (systematically named dioxacyclopropane, also known as methylene peroxide or peroxymethane) is an organic compound with formula . The molecule consists of a ring with one methylene and two oxygen atoms. It is of interest as the smallest cyclic organic peroxide, but otherwise it is of little practical value. Synthesis Dioxirane is highly unstable and the majority of studies of it have been computational; it has been detected during the low temperature (–196 °C) reaction of ethylene and ozone, although even at these temperatures such a mixture can be explosive. Its formation is thought to be radical in nature, preceding via a Criegee intermediate. Microwave analysis has indicated C-H, C-O and O-O bond lengths of 1.090, 1.388 and 1.516 Å respectively. The very long and weak O-O bond (cf. hydrogen peroxide O-O = 1.47 Å) is the origin of its instability. Other dioxiranes Beyond the parent dioxirane, which is mainly of theoretical interest, more c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetone Peroxide
Acetone peroxide ( also called APEX and mother of Satan) is an organic peroxide and a primary explosive. It is produced by the reaction of acetone and hydrogen peroxide to yield a mixture of linear monomer and cyclic dimer, trimer, and tetramer forms. The monomer is dimethyldioxirane. The dimer is known as diacetone diperoxide (DADP). The trimer is known as triacetone triperoxide (TATP) or tri-cyclic acetone peroxide (TCAP). Acetone peroxide takes the form of a white crystalline powder with a distinctive bleach-like odor when impure, or a fruit-like smell when pure, and can explode powerfully if subjected to heat, friction, static electricity, concentrated sulfuric acid, strong UV radiation, or shock. Until about 2015, explosives detectors were not set to detect non-nitrogenous explosives, as most explosives used preceding 2015 were nitrogen-based. TATP, being nitrogen-free, has been used as the explosive of choice in several terrorist bomb attacks since 2001. History ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxone
Potassium peroxymonosulfate is widely used as an oxidizing agent, for example, in pools and spas (usually referred to as monopersulfate or "MPS"). It is the potassium salt of peroxymonosulfuric acid. Potassium peroxymonosulfate per se is rarely encountered. It is often confused with the triple salt , known as Oxone. The standard electrode potential for potassium peroxymonosulfate is +1.81 V with a half reaction generating the hydrogen sulfate (): : Oxone Potassium peroxymonosulfate per se is a relatively obscure salt, but its derivative called Oxone is of commercial value. Oxone refers to the triple salt . As such about one third by weight is potassium peroxymonosulfate. Oxone has a longer shelf life than does potassium peroxymonosulfate. A white, water-soluble solid, Oxone loses <1% of its oxidizing power per month. Oxone, which is commercially available, is produced from peroxysulfuric acid, which is generated in situ by combining |
Potassium Peroxymonosulfate
Potassium peroxymonosulfate is widely used as an oxidizing agent, for example, in pools and spas (usually referred to as monopersulfate or "MPS"). It is the potassium salt (chemistry), salt of peroxymonosulfuric acid. Potassium peroxymonosulfate per se is rarely encountered. It is often confused with the triple salt , known as Oxone. The standard electrode potential for potassium peroxymonosulfate is +1.81 V with a half reaction generating the hydrogen sulfate (): : Oxone Potassium peroxymonosulfate per se is a relatively obscure salt, but its derivative called Oxone is of commercial value. Oxone refers to the triple salt . As such about one third by weight is potassium peroxymonosulfate. Oxone has a longer shelf life than does potassium peroxymonosulfate. A white, water-soluble solid, Oxone loses <1% of its oxidizing power per month. Oxone, which is commercially available, is produced from peroxysulfuric acid, which is generated in situ by combining oleum and hydrogen perox ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxone
Potassium peroxymonosulfate is widely used as an oxidizing agent, for example, in pools and spas (usually referred to as monopersulfate or "MPS"). It is the potassium salt of peroxymonosulfuric acid. Potassium peroxymonosulfate per se is rarely encountered. It is often confused with the triple salt , known as Oxone. The standard electrode potential for potassium peroxymonosulfate is +1.81 V with a half reaction generating the hydrogen sulfate (): : Oxone Potassium peroxymonosulfate per se is a relatively obscure salt, but its derivative called Oxone is of commercial value. Oxone refers to the triple salt . As such about one third by weight is potassium peroxymonosulfate. Oxone has a longer shelf life than does potassium peroxymonosulfate. A white, water-soluble solid, Oxone loses <1% of its oxidizing power per month. Oxone, which is commercially available, is produced from peroxysulfuric acid, which is generated in situ by combining |
Nef Reaction
In organic chemistry, the Nef reaction is an organic reaction describing the acid hydrolysis of a salt of a primary or secondary nitroalkane () to an aldehyde () or a ketone () and nitrous oxide (). The reaction has been the subject of several literature reviews. The reaction was reported in 1894 by the chemist John Ulric Nef, who treated the sodium salt of nitroethane with sulfuric acid resulting in an 85–89% yield of nitrous oxide and at least 70% yield of acetaldehyde. However, the reaction was pioneered a year earlier in 1893 by Konovalov, who converted the potassium salt of 1-phenylnitroethane with sulfuric acid to acetophenone. Reaction mechanism The reaction mechanism starting from the nitronate salt as the resonance structures 1a and 1b is depicted below: The salt is protonated forming the nitronic acid 2 (in some cases these nitronates have been isolated) and once more to the iminium ion 3. This intermediate is attacked by water in a nucleophilic addition ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Synlett
''Synlett'' is an international scientific journal for accounts and rapid communications of original contributions of fundamental research in synthetic organic chemistry. The impact factor of this journal is 2.419 (2017). ''Nature'' featured a brief piece by the editor-in-chief of the journal in 2017, Benjamin List Benjamin List (; born 11 January 1968) is a German chemist who is one of the directors of the Max Planck Institute for Coal Research and professor of organic chemistry at the University of Cologne. He co-developed organocatalysis, a method of acc ..., where he discussed the journal's experience with the non-traditional peer review system. References Chemistry journals Thieme academic journals Academic journals established in 1989 {{chem-journal-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |