|
Sulfide Stress Cracking
Sulfide stress cracking (SSC) is a form of hydrogen embrittlement which is a cathodic cracking mechanism. It should not be confused with the term stress corrosion cracking which is an anodic cracking mechanism. Susceptible alloys, especially steels, react with hydrogen sulfide (), forming metal sulfides (MeS) and atomic hydrogen (H•) as corrosion byproducts. Atomic hydrogen either combines to form H2 at the metal surface or diffuses into the metal matrix. Since sulfur is a hydrogen recombination poison, the amount of atomic hydrogen which recombines to form H2 on the surface is greatly reduced, thereby increasing the amount of diffusion of atomic hydrogen into the metal matrix. This aspect is what makes wet H2S environments so severe.Foroulis Z.A ed., High-Temperature Metallic Corrosion by Sulfur and Its Compounds, The Electrochemical Society, Princeton, NJ. (1970) Conference proceedings Since SSC is a form of hydrogen embrittlement, it is most susceptibile to cracking at or s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Embrittlement
Hydrogen embrittlement (HE), also known as hydrogen-assisted cracking or hydrogen-induced cracking (HIC), is a reduction in the ductility of a metal due to absorbed hydrogen. Hydrogen atoms are small and can permeate solid metals. Once absorbed, hydrogen lowers the stress required for cracks in the metal to initiate and propagate, resulting in embrittlement. Hydrogen embrittlement occurs most notably in steels, as well as in iron, nickel, titanium, cobalt, and their alloys. Copper, aluminium, and stainless steels are less susceptible to hydrogen embrittlement. The essential facts about the nature of hydrogen embrittlement have been known since the 19th century. Hydrogen embrittlement is maximised at around room temperature in steels, and most metals are relatively immune to hydrogen embrittlement at temperatures above 150 °C. Hydrogen embrittlement requires the presence of both atomic ("diffusible") hydrogen and a mechanical stress to induce crack growth, although that str ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crude Oil
Petroleum, also known as crude oil, or simply oil, is a naturally occurring yellowish-black liquid mixture of mainly hydrocarbons, and is found in geological formations. The name ''petroleum'' covers both naturally occurring unprocessed crude oil and petroleum products that consist of refined crude oil. A fossil fuel, petroleum is formed when large quantities of dead organisms, mostly zooplankton and algae, are buried underneath sedimentary rock and subjected to both prolonged heat and pressure. Petroleum is primarily recovered by oil drilling. Drilling is carried out after studies of structural geology, sedimentary basin analysis, and reservoir characterisation. Recent developments in technologies have also led to exploitation of other Unconventional (oil & gas) reservoir, unconventional reserves such as oil sands and oil shale. Once extracted, oil is refined and separated, most easily by Continuous distillation#Continuous distillation of crude oil, distillation, into innume ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfidation
Sulfidation (British spelling also sulphidation) is a process of installing sulfide ions in a material or molecule. The process is widely used to convert oxides to sulfides but is also related to corrosion and surface modification. Inorganic, materials, and organic chemistry Sulfidation is relevant to the formation of sulfide minerals. A large scale application of sulfidation is the conversion of molybdenum oxides to the corresponding sulfides. This conversion is a step in the preparation of catalysts for hydrodesulfurization wherein alumina impregnated with molybdate salts are converted to molybdenum disulfide by the action of hydrogen sulfide. In organosulfur chemistry, sulfiding is often called thiation. The preparation of thioamides from amides involves thiation. A typical reagent is phosphorus pentasulfide (P4S10). The idealized equation for this conversion is: :RC(O)NH2 + 1/4 P4S10 → RC(S)NH2 + 1/4 P4S6O4 This conversion where an oxygen atom in the amide f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pitting Corrosion
Pitting corrosion, or pitting, is a form of extremely localized corrosion that leads to the random creation of small holes in metal. The driving power for pitting corrosion is the depassivation of a small area, which becomes anodic (oxidation reaction) while an unknown but potentially vast area becomes cathodic (reduction reaction), leading to very localized galvanic corrosion. The corrosion penetrates the mass of the metal, with a limited diffusion of ions. Another term arises, pitting factor, which is defined as the ratio of the depth of the deepest pit (resulting due to corrosion) to the average penetration, which can be calculated based on the weight loss. Development and kinetics of pitting According to Frankel (1998) who performed a review on pitting corrosion, it develops in three successive steps: (or nucleation) by breakdown of the passive film protecting the metal surface from oxidation, (2) growth of metastable pits (growing up to the micron scale and then repassiva ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crevice Corrosion
Crevice corrosion refers to corrosion occurring in occluded spaces such as interstices in which a stagnant solution is trapped and not renewed. These spaces are generally called crevices. Examples of crevices are gaps and contact areas between parts, under gaskets or seals, inside cracks and seams, spaces filled with deposits and under sludge piles. Mechanism The corrosion resistance of a stainless steel is dependent on the presence of an ultra-thin protective oxide film (passive film) on its surface, but it is possible under certain conditions for this oxide film to break down, for example in halide solutions or reducing acids. Areas where the oxide film can break down can also sometimes be the result of the way components are designed, for example under gaskets, in sharp re-entrant corners or associated with incomplete weld penetration or overlapping surfaces. These can all form crevices which can promote corrosion. To function as a corrosion site, a crevice has to be of suffi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrosion Engineering
Corrosion engineering is an engineering specialty that applies scientific, technical, engineering skills, and knowledge of natural laws and physical resources to design and implement materials, structures, devices, systems, and procedures to manage corrosion. From a holistic perspective, corrosion is the phenomenon of metals returning to the state they are found in nature. The driving force that causes metals to corrode is a consequence of their temporary existence in metallic form. To produce metals starting from naturally occurring minerals and ores, it is necessary to provide a certain amount of energy, e.g. Iron ore in a blast furnace. It is therefore thermodynamically inevitable that these metals when exposed to various environments would revert to their state found in nature. Corrosion and corrosion engineering thus involves a study of chemical kinetics, thermodynamics, electrochemistry and materials science. General background Generally related to metallurgy or material ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Earth makes it an economically attractive fuel, although capturing and storing it poses technical challenges due to its gaseous state under normal conditions for temperature and pressure. Naturally occurring methane is found both below ground and under the seafloor and is formed by both geological and biological processes. The largest reservoir of methane is under the seafloor in the form of methane clathrates. When methane reaches the surface and the atmosphere, it is known as atmospheric methane. The Earth's atmospheric methane concentration has increased by about 150% since 1750, and it accounts for 20% of the total radiative forcing from all of the long-lived and globally mixed greenhouse gases. It has also been detected on other plane ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules. Description The combining capacity, or affinity of an ...—its atom making four electrons available to form covalent bond, covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes up only about 0.025 percent of Earth's crust. Three Isotopes of carbon, isotopes occur naturally, Carbon-12, C and Carbon-13, C being stable, while Carbon-14, C is a radionuclide, decaying with a half-life of about 5,730 years. Carbon is one of the Timeline of chemical element discoveries#Ancient discoveries, few elements known since antiquity. Carbon is the 15th Abundance of elements in Earth's crust, most abundant element in the Earth's crust, and the Abundance of the c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
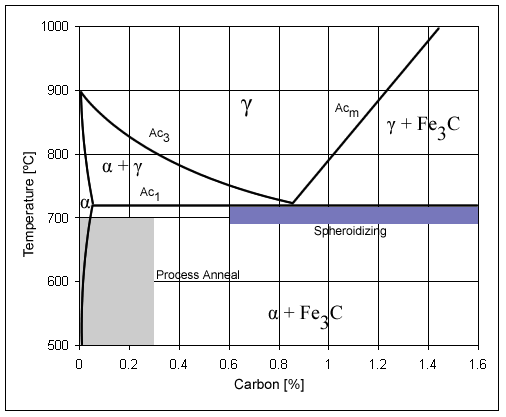
Carbon Steel
Carbon steel is a steel with carbon content from about 0.05 up to 2.1 percent by weight. The definition of carbon steel from the American Iron and Steel Institute (AISI) states: * no minimum content is specified or required for chromium, cobalt, molybdenum, nickel, niobium, titanium, tungsten, vanadium, zirconium, or any other element to be added to obtain a desired alloying effect; * the specified minimum for copper does not exceed 0.40%; * or the maximum content specified for any of the following elements does not exceed the percentages noted: manganese 1.65%; silicon 0.60%; copper 0.60%. The term ''carbon steel'' may also be used in reference to steel which is not stainless steel; in this use carbon steel may include alloy steels. High carbon steel has many different uses such as milling machines, cutting tools (such as chisels) and high strength wires. These applications require a much finer microstructure, which improves the toughness. Carbon steel is a popular metal choic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
High Temperature Hydrogen Attack
High temperature hydrogen attack (HTHA), also called hot hydrogen attack or methane reaction, is a problem which concerns steels operating at elevated temperatures (typically above ) in hydrogen-rich atmospheres, such as refineries, petrochemical and other chemical facilities and, possibly, high pressure steam boilers. It is not to be confused with hydrogen embrittlement. If a steel is exposed to very hot hydrogen, the high temperature enables the hydrogen molecules to dissociate and to diffuse into the alloy as individual ''diffusible'' atoms. There are two stages to the damage: # First, dissolved carbon in the steel reacts with the surface hydrogen and escapes into the gas as methane. This leads to superficial decarburization and a loss of strength in the surface. Initially, the damage is not visible. # Second, the reduction in the concentration of dissolved carbon creates a driving force which dissolves the carbides in the steel. This leads to a loss of strength deeper in the ste ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
NACE International
The Association for Materials Protection and Performance (AMPP), is a professional association focused on the protection of assets and performance of materials. AMPP was created when NACE International and SSPC the Society for Protective Coatings merged in 2021. AMPP is active in more than 130 countries and has more than 40,000 members. AMPP is headquartered in the U.S. with offices in Houston, Texas and Pittsburgh, Pennsylvania. Additional offices are located in the U.K., China, Malaysia, Brazil, and Saudi Arabia with a training center in Dubai. Publications *Materials Performance magazine *CoatingsPro magazine *CORROSION journal *technical and reference books *podcasts and webcasts Standards Both the legacy NACE and SSPC organizations were ANSI-accredited standards developers, which AMPP plans to continue. The merged standards program includes 25 standing standards committees that develop technical standards for industries including cathodic protection, coatings, defense, hig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Natural Gas
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbon dioxide, nitrogen, hydrogen sulfide, and helium are also usually present. Natural gas is colorless and odorless, so odorizers such as mercaptan (which smells like sulfur or rotten eggs) are commonly added to natural gas supplies for safety so that leaks can be readily detected. Natural gas is a fossil fuel and non-renewable resource that is formed when layers of organic matter (primarily marine microorganisms) decompose under anaerobic conditions and are subjected to intense heat and pressure underground over millions of years. The energy that the decayed organisms originally obtained from the sun via photosynthesis is stored as chemical energy within the molecules of methane and other hydrocarbons. Natural gas can be burned fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |