|
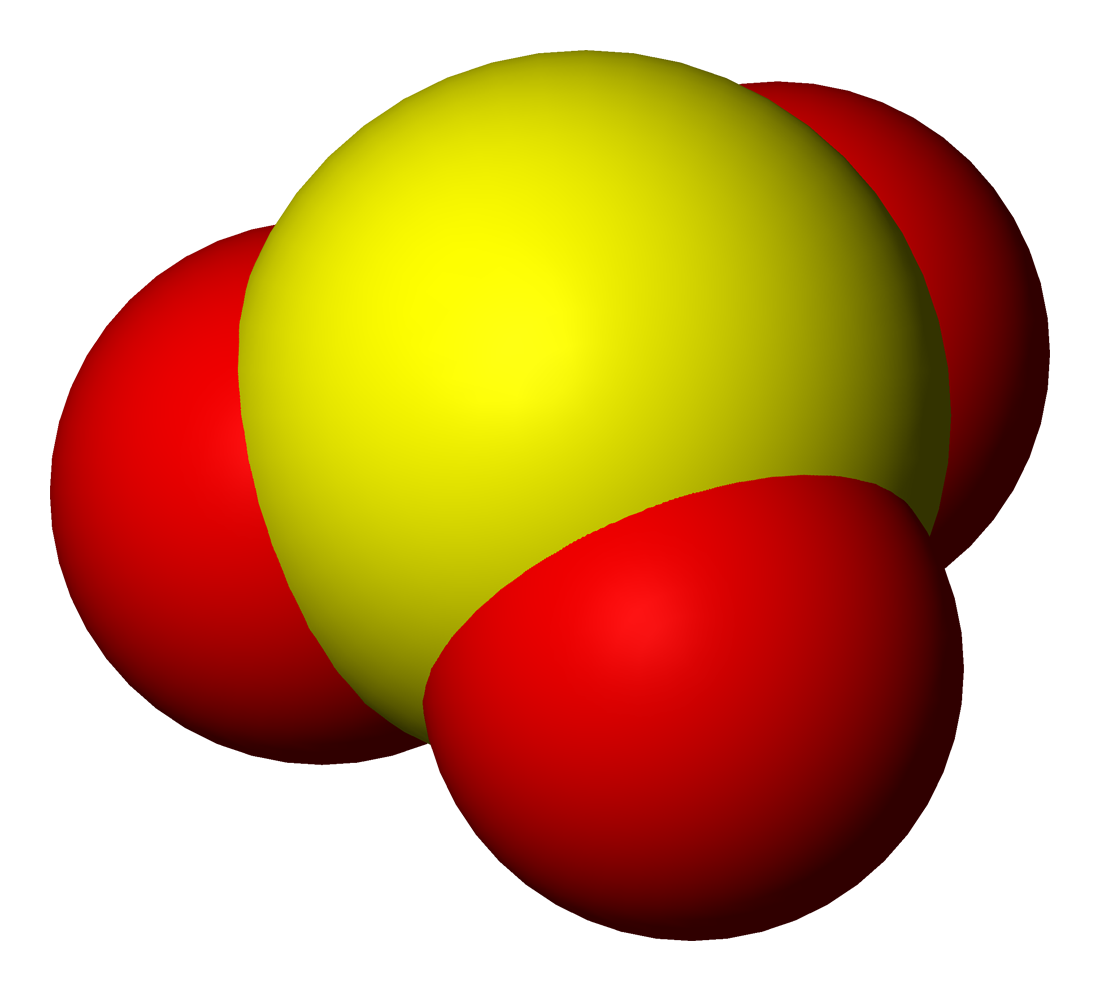
Sulfite
Sulfites or sulphites are compounds that contain the sulfite ion (systematic name: sulfate(IV) ion), . The sulfite ion is the conjugate base of bisulfite. Although its acid (sulfurous acid) is elusive, its salts are widely used. Sulfites are substances that naturally occur in some foods and the human body. They are also used as regulated food additives. When in food or drink, sulfites are often lumped together with sulfur dioxide.SeREGULATION (EU) No 1169/2011 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL/ref> Structure The structure of the sulfite anion can be described with three equivalent resonance structures. In each resonance structure, the sulfur atom is double-bonded to one oxygen atom with a formal charge of zero (neutral), and sulfur is singly bonded to the other two oxygen atoms, which each carry a formal charge of −1, together accounting for the −2 charge on the anion. There is also a non-bonded lone pair on the sulfur, so the structure predicted by VSEPR ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Bisulfite
Sodium bisulfite (or sodium bisulphite, sodium hydrogen sulfite) is a chemical mixture with the approximate chemical formula NaHSO3. Sodium bisulfite is not a real compound, but a mixture of salts that dissolve in water to give solutions composed of sodium and bisulfite ions. It appears in form of white or yellowish-white crystals with an odor of sulfur dioxide. Sodium bisulfite is used in a variety industries such as a food additive with E number E222 in the food industry. It is a reducing agent in the cosmetic and in the bleaching applications. Synthesis Sodium bisulfite solutions can be prepared by treating a solution of suitable base, such as sodium hydroxide or sodium bicarbonate with sulfur dioxide. :SO2 + NaOH → NaHSO3 :SO2 + NaHCO3 → NaHSO3 + CO2 Attempts to crystallize the product yield sodium metabisulfite (also called sodium disulfite), Na2S2O5. Upon dissolution of the metabisulfite in water, bisulfite is regenerated: :Na2S2O5 + H2O → 2 Na+ + 2 HSO3� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bisulfite
The bisulfite ion (IUPAC-recommended nomenclature: hydrogensulfite) is the ion . Salts containing the ion are also known as "sulfite lyes". Sodium bisulfite is used interchangeably with sodium metabisulfite (Na2S2O5). Sodium metabisulfite dissolves in water to give a solution of Na+. :Na2S2O5 + H2O → 2Na SO3 Structure The bisulfite anion exists in solution as a mixture of two tautomers. One tautomer has the proton attached to one of the three oxygen atoms. In the second tautomer the proton resides on sulfur. The S-protonated tautomer has ''C''3v symmetry. The O-protonated tautomer has only ''C''s symmetry. Reactions Tautomerization There exist two tautomers of bisulfite. They interconvert readily but can be characterized individually by various spectroscopic methods. They have been observed by 17O NMR spectroscopy: :HSO3− SO2(OH)− ''K'' = 4.9 Acid-base reactions Solutions of bisulfite are typically prepared by treatment of sulfur dioxide with aqueous bas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Metabisulfite
Sodium metabisulfite or sodium pyrosulfite (IUPAC spelling; Br. E. sodium metabisulphite or sodium pyrosulphite) is an inorganic compound of chemical formula Na2S2O5. The substance is sometimes referred to as disodium metabisulfite. It is used as a disinfectant, antioxidant, and preservative agent. When dissolved in water it forms sodium bisulfite. Preparation Sodium metabisulfite can be prepared by treating a solution of sodium hydroxide with sulfur dioxide. When conducted in warm water, Na2SO3 initially precipitates as a yellow solid. With more SO2, the solid dissolves to give the disulfite, which crystallises upon cooling. :SO2 + 2 NaOH → Na2SO3 + H2O :SO2 + Na2SO3 → Na2S2O5 which yields a residue of colourless solid Na2S2O5. Chemical structure The anion metabisulfite consists of an SO2 group linked to an SO3 group, with the negative charge more localised on the SO3 end. The S–S bond length is 2.22 Å, and the "thionate" and "thionite" S–O distances are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Metabisulfite
Potassium metabisulfite, K2S2O5, also known as potassium pyrosulfite, is a white crystalline powder with a pungent odour. It is mainly used as an antioxidant or chemical sterilant. As a disulfite, it is chemically very similar to sodium metabisulfite, with which it is sometimes used interchangeably. Potassium metabisulfite has a monoclinic crystal structure. Preparation and reactions Potassium metabisulfite can be prepared by treating a solution of potassium hydroxide with sulfur dioxide. :2 SO2 + 2 KOH → K2S2O5 + H2O It decomposes at 190 °C, yielding potassium sulfite and sulfur dioxide: :K2S2O5 → K2SO3 + SO2 Uses It is used as a food additive, also known as E224. It is restricted in use and may cause allergic reactions in some sensitive persons. Wine Potassium metabisulfite is a common wine or must additive, in which it forms sulfur dioxide (SO2). Sulfur dioxide is a disinfectant. It also acts as a potent antioxidant, protecting both the color and deli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Sulfite
Sodium sulfite (sodium sulphite) is the inorganic compound with the chemical formula Na2SO3. A white, water-soluble solid, it is used commercially as an antioxidant and preservative. It is also suitable for the softening of lignin in the pulping and refining processes of wood and Lignocellulosic biomass, lignocellulosic materials. A heptahydrate is also known but it is less useful because of its greater susceptibility toward oxidation by air. Preparation image:Sodium-sulfite-xtal-3x3x3-c-3D-bs-17.png, Structure of anhydrous sodium sulfite, left, thumb Sodium sulfite can be prepared by treating a solution of sodium hydroxide with sulfur dioxide. When conducted in warm water, Na2SO3 initially precipitates as a white solid. With more SO2, the solid dissolves to give the Na2S2O5, disulfite, which crystallizes upon cooling. : Sodium sulfite is made industrially by treating sulfur dioxide with a solution of sodium carbonate. The overall reaction is: : Uses Sodium sulfite is primaril ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Bisulfite
Potassium bisulfite (or potassium hydrogen sulfite) is a chemical mixture with the approximately correctly mentioned formula chemical formula KHSO3. Potassium bisulfite in fact is not an actual compound, but a mixture of salts that dissolve in water to give solutions composed of potassium ions and bisulfite ions. It is a white solid with an odor of sulfur dioxide. Attempts to crystallize potassium bisulfite yield potassium metabisulfite, K2S2O5. Potassium bisulfite is used as a sterilising agent in the production of alcoholic beverages. This additive is classified as E number E228 under the current EU-approved food additive legislation. Production It is made by the reaction of sulfur dioxide and potassium carbonate. The sulfur dioxide is passed through a solution of the potassium carbonate until no more carbon dioxide is evolved. The solution is concentrated. See also * Calcium bisulfite * Sodium bisulfite Sodium bisulfite (or sodium bisulphite, sodium hydrogen sulfite) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur Dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a colorless gas with a pungent smell that is responsible for the odor of burnt matches. It is released naturally by volcanic activity and is produced as a by-product of metals refining and the burning of Sour gas, sulfur-Sour crude oil, bearing fossil fuels. Sulfur dioxide is somewhat toxic to humans, although only when inhaled in relatively large quantities for a period of several minutes or more. It was known to medieval alchemy, alchemists as "volatile spirit of sulfur". Structure and bonding SO2 is a bent molecule with ''C''2v Point groups in three dimensions, symmetry point group. A valence bond theory approach considering just ''s'' and ''p'' orbitals would describe the bonding in terms of resonance (chemistry), resonance between two resonance structures. The sulfur–oxygen bond has a bond order of 1.5. There is support f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfurous Acid
Sulfuric(IV) acid (United Kingdom spelling: sulphuric(IV) acid), also known as sulfurous (UK: sulphurous) acid and thionic acid, is the chemical compound with the chemical formula, formula . Raman spectroscopy, Raman spectra of solutions of sulfur dioxide in water show only signals due to the molecule and the bisulfite ion, . The intensities of the signals are consistent with the following chemical equilibrium, equilibrium: 17O NMR spectroscopy provided evidence that solutions of sulfurous acid and protonated sulfites contain a mixture of isomers, which is in equilibrium: Attempts to concentrate the solutions of sulfurous acid simply reverse the equilibrium, producing sulfur dioxide and water vapor. A clathrate with the formula has been crystallised. It decomposes above 7 °C. History and production Sulfurous acid is commonly known not to exist in its free state, and owing to this, it is stated in textbooks that it cannot be isolated in the water-free form. Ho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Winemaking
Winemaking, wine-making, or vinification is the production of wine, starting with the selection of the fruit, its Ethanol fermentation, fermentation into alcohol, and the bottling of the finished liquid. The history of wine-making stretches over millennia. There is evidence that suggests that the earliest wine production took place in Georgia and Iran around 6000 to 5000 B.C. The science of wine and winemaking is known as oenology. A winemaker may also be called a #Winemakers, vintner. The growing of grapes is viticulture and there are List of grape varieties, many varieties of grapes. Winemaking can be divided into two general categories: still wine production (without carbonation) and sparkling wine production (with carbonation – natural or injected). Red wine, white wine, and rosé are the other main categories. Although most wine is made from grapes, it may also be made from other plants. (See fruit wine.) Other similar light alcoholic drinks (as opposed to beer or Liq ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Preservative
A preservative is a substance or a chemical that is added to products such as food products, beverages, pharmaceutical drugs, paints, biological samples, cosmetics, wood, and many other products to prevent decomposition by microbial growth or by undesirable chemical changes. In general, preservation is implemented in two modes, chemical and physical. Chemical preservation entails adding chemical compounds to the product. Physical preservation entails processes such as refrigeration or drying.Erich Lück and Gert-Wolfhard von Rymon Lipinski "Foods, 3. Food Additives" in ''Ullmann's Encyclopedia of Industrial Chemistry'', 2002, Wiley-VCH, Weinheim. Preservative food additives reduce the risk of foodborne infections, decrease microbial spoilage, and preserve fresh attributes and nutritional quality. Some physical techniques for food preservation include dehydration, UV-C radiation, freeze-drying, and refrigeration. Chemical preservation and physical preservation techniques ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wine
Wine is an alcoholic drink made from Fermentation in winemaking, fermented fruit. Yeast in winemaking, Yeast consumes the sugar in the fruit and converts it to ethanol and carbon dioxide, releasing heat in the process. Wine is most often made from grapes, and the term "wine" generally refers to grape wine when used without any qualification. Even so, wine can be made fruit wine, from a variety of fruit crops, including plum, cherry, pomegranate, blueberry, Ribes, currant, and Sambucus, elderberry. Different varieties of grapes and Strain (biology), strains of yeasts are major factors in different styles of wine. These differences result from the complex interactions between the Biochemistry, biochemical development of the grape, the reactions involved in fermentation, the grape's growing environment (terroir), and the wine production process. Many countries enact legal appellations intended to define styles and qualities of wine. These typically restrict the geographical origin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |