|
Methacrylic Acid
Methacrylic acid, abbreviated MAA, is an organic compound. This colorless, viscous liquid is a carboxylic acid with an acrid unpleasant odor. It is soluble in warm water and miscible with most organic solvents. Methacrylic acid is produced industrially on a large scale as a precursor to its esters, especially methyl methacrylate (MMA), and to poly(methyl methacrylate) (PMMA). MAA occurs naturally in small amounts in the oil of Roman chamomile. Production In the most common route, methacrylic acid is prepared from acetone cyanohydrin, which is converted to methacrylamide sulfate using sulfuric acid. This derivative in turn is hydrolyzed to methacrylic acid, or esterified to methyl methacrylate in one step. Another route to methacrylic acid starts with isobutylene, which obtainable by dehydration of ''tert''-butanol. Isobutylene is oxidized sequentially to methacrolein and then methacrylic acid. Methacrolein for this purpose can also be obtained from formaldehyde and ethylene. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
International Union Of Pure And Applied Chemistry
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is a member of the International Science Council (ISC). IUPAC is registered in Zürich, Switzerland, and the administrative office, known as the "IUPAC Secretariat", is in Research Triangle Park, North Carolina, United States. This administrative office is headed by IUPAC's executive director, currently Lynn Soby. IUPAC was established in 1919 as the successor of the International Congress of Applied Chemistry for the advancement of chemistry. Its members, the National Adhering Organizations, can be national chemistry societies, national academies of sciences, or other bodies representing chemists. There are fifty-four National Adhering Organizations and three Associate National Adhering Organizations. IUPAC's Inter-divisional Committee on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Decarboxylation
Decarboxylation is a chemical reaction that removes a carboxyl group and releases carbon dioxide (CO2). Usually, decarboxylation refers to a reaction of carboxylic acids, removing a carbon atom from a carbon chain. The reverse process, which is the first chemical step in photosynthesis, is called carboxylation, the addition of CO2 to a compound. Enzymes that catalyze decarboxylations are called decarboxylases or, the more formal term, carboxy-lyases (Enzyme Commission number, EC number 4.1.1). In organic chemistry The term "decarboxylation" usually means replacement of a carboxyl group () with a hydrogen atom: :RCO2H -> RH + CO2 Decarboxylation is one of the oldest known organic reactions. It is one of the processes assumed to accompany pyrolysis and destructive distillation. Metal salts, especially copper compounds, facilitate the reaction via the intermediacy of metal carboxylate complexes. Decarboxylation of aryl carboxylates can generate the equivalent of the correspond ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monomers
In chemistry, a monomer ( ; ''mono-'', "one" + '' -mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization. Classification Monomers can be classified in many ways. They can be subdivided into two broad classes, depending on the kind of the polymer that they form. Monomers that participate in condensation polymerization have a different stoichiometry than monomers that participate in addition polymerization: : Other classifications include: *natural vs synthetic monomers, e.g. glycine vs caprolactam, respectively *polar vs nonpolar monomers, e.g. vinyl acetate vs ethylene, respectively *cyclic vs linear, e.g. ethylene oxide vs ethylene glycol, respectively The polymerization of one kind of monomer gives a homopolymer. Many polymers are copolymers, meaning that they are derived from two different monomers. In the case of condensation polymerizations, the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part") is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals. The term "polymer" derives from the Greek word πολύς (''polus'', meaning "many, much") and μέρος (''meros'' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isobutyric Acid
Isobutyric acid, also known as 2-methylpropanoic acid or isobutanoic acid, is a carboxylic acid with structural formula ( CH3)2CH COOH. It is an isomer of ''n''- butyric acid. It is classified as a short-chain fatty acid. Deprotonation or esterification gives derivatives called isobutyrates. Isobutyric acid is a colorless liquid with a somewhat unpleasant odor. It is soluble in water and organic solvents. It is found naturally in carobs (''Ceratonia siliqua''), in vanilla, and in the root of ''Arnica dulcis'', and as an ethyl ester in croton oil. Production Isobutyric acid is manufactured by the oxidation of isobutyraldehyde, which is a byproduct of the hydroformylation of propylene. It can also be prepared by the high pressure hydrocarboxylation ( Koch reaction) from propylene: :CH3CH=CH2 + CO + H2O → (CH3)2CHCO2H Isobutyric acid can also be manufactured commercially using engineered bacteria with a sugar feedstock. Laboratory methods Many routes are known includ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Amalgam
Sodium amalgam, commonly denoted Na(Hg), is an alloy of mercury and sodium. The term amalgam is used for alloys, intermetallic compounds, and solutions (both solid solutions and liquid solutions) involving mercury as a major component. Sodium amalgams are often used in reactions as strong reducing agents with better handling properties compared to solid sodium. They are less dangerously reactive toward water and in fact are often used as an aqueous suspension. Sodium amalgam was used as a reagent as early as 1862. A synthesis method was described by J. Alfred Wanklyn in 1866. Structure and compositions No particular formula is assigned to "sodium amalgam". Na5Hg8 and Na3Hg are well defined compounds. In sodium amalgams, the Hg-Hg distances are expanded to around 5 Å vs. about 3 Å for mercury itself. Usually amalgams are classified on the weight percent of sodium. Amalgams with 2% Na are solids at room temperature, whereas some more dilute amalgams remain liquid. Preparation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
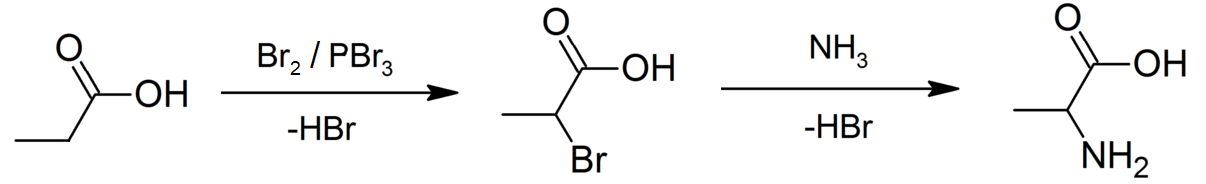
Propanoic Acid
Propionic acid (, from the Greek words πρῶτος : ''prōtos'', meaning "first", and πίων : ''píōn'', meaning "fat"; also known as propanoic acid) is a naturally occurring carboxylic acid with chemical formula CH3CH2CO2H. It is a liquid with a pungent and unpleasant smell somewhat resembling body odor. The anion CH3CH2CO2− as well as the salts and esters of propionic acid are known as propionates or propanoates. History Propionic acid was first described in 1844 by Johann Gottlieb, who found it among the degradation products of sugar. Over the next few years, other chemists produced propionic acid by different means, none of them realizing they were producing the same substance. In 1847, French chemist Jean-Baptiste Dumas established all the acids to be the same compound, which he called propionic acid, from the Greek words πρῶτος (prōtos), meaning ''first'', and πίων (piōn), meaning ''fat'', because it is the smallest H(CH2)''n''COOH acid that exhibits ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Artificial Nails
Artificial nails, also known as fake nails, false nails, acrylic nails, nail extensions or nail enhancements, are extensions placed over fingernails as fashion accessories. Many artificial nail designs attempt to mimic the appearance of real fingernails as closely as possible, while others may deliberately stray in favor of an artistic look. Artificial nails require regular upkeep; it is recommended that they are attended to, on average, every two weeks, however they may last over one month. Nonetheless, their versatility in terms of shape, size, design and comparatively high durability are some advantages they hold over other types of manicures. Types Artificial nails are an extension, not a replacement, of natural nails. There are two main approaches to creating artificial nails—''tips'' and ''forms'': * tips are lightweight "nail"-shaped plastic plates glued on the end of the natural nail; * forms are shaped sheets with a sticky edge that is effectively attached to the t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethyl Methacrylate
Ethyl methacrylate is the organic compound with the formula C2H5O2CC(CH3)=CH2. A colorless liquid, it is a common monomer for the preparation of acrylate polymers.. It is typically polymerized under free-radical conditions. Ethyl methacrylate was first obtained by treating 2-hydroxyisobutyric acid with phosphorus pentachloride in an apparent dehydration reaction.{{cite journal, journal=Justus Liebigs Annalen der Chemie, volume= 136, page= 12, title=Untersuchungen über Säuren aus der Acrylsäure-Reihe; 1) Umwandlung der Säuren aus der Milchsäure-Reihe in die der Acrylsäure-Reihe, author=E. Frankland, B. F. Duppa, year=1865, doi=10.1002/jlac.18651360102 Environmental issues and health hazards The acute toxicity of the related butyl methacrylate is the LD50 is 20 g/kg (oral, rat). Acrylate esters irritate the eyes and can cause blindness. See also * Methyl methacrylate * Butyl methacrylate Butyl methacrylate is the organic compound with the formula C4H9O2CC(CH3)=CH2. A c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkali
In chemistry, an alkali (; from ar, القلوي, al-qaly, lit=ashes of the saltwort) is a basic, ionic salt of an alkali metal or an alkaline earth metal. An alkali can also be defined as a base that dissolves in water. A solution of a soluble base has a pH greater than 7.0. The adjective alkaline, and less often, alkalescent, is commonly used in English as a synonym for basic, especially for bases soluble in water. This broad use of the term is likely to have come about because alkalis were the first bases known to obey the Arrhenius definition of a base, and they are still among the most common bases. Etymology The word "alkali" is derived from Arabic ''al qalīy'' (or ''alkali''), meaning ''the calcined ashes'' (see calcination), referring to the original source of alkaline substances. A water-extract of burned plant ashes, called potash and composed mostly of potassium carbonate, was mildly basic. After heating this substance with calcium hydroxide (''slaked lime ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mesaconic Acid
Mesaconic acid is one of several isomeric carboxylic acids obtained from citric acid. It is a colorless solid. Synthesis and reactions It is prepared from citric acid, which is first converted to itaconic anhydride by dehydration and decarboxylation. Itaconic acid anhydride is isomerized to citraconic anhydride, which is hydrolyzed and the resulting acid further isomerized under acid-catalysis to give mesaconic acid. Hydration of mesaconic acid, a conversion catalyzed by mesaconyl-C4-CoA hydratase, gives citramalic acid. History This acid was studied for the first time by Jacobus H. van 't Hoff in 1874. It was later shown to be produced by ''Clostridium tetanomorphum''. Further studies showed that this organic compound is involved in the biosynthesis of vitamin B12. It is a competitive inhibitor of fumarate reduction. The compound has been considered as a renewable precursor to the commodity chemical methacrylic acid Methacrylic acid, abbreviated MAA, is an organic compoun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |