|
Fire Clay
Fire clay is a range of refractory clays used in the manufacture of ceramics, especially fire brick. The United States Environmental Protection Agency defines fire clay very generally as a "mineral aggregate composed of hydrous silicates of aluminium (Al2O3·2SiO2·2H2O) with or without free silica." Properties High-grade fire clays can withstand temperatures of 1,775 °C (3,227 °F), but to be referred to as a "fire clay" the material must withstand a minimum temperature of .Minerals Zone, World Mineral Exchange. Retrieved 2011-6-23. Fire clays range from '' flint clays'' to ''plastic fire clays'', but there are ''semi-flint'' and ''semi-plastic'' fire clays as well. Fire clays consist of natural [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saggar
A saggar (also misspelled as sagger or segger) is a type of kiln furniture. It is a ceramic boxlike container used in the firing of pottery to enclose or protect ware being fired inside a kiln. Traditionally, saggars were made primarily from fireclay. Saggars have been used to protect, or safeguard, ware from open flame, smoke, gases and kiln debris: the name may be a contraction of the word ''safeguard''. Their use is widespread, including in China, Korea, Japan and the United Kingdom. Saggars are still used in the production of ceramics to shield ware from the direct contact of flames and from damage by kiln debris. Modern saggars are made of alumina ceramic, cordierite ceramic, mullite ceramic silicon carbide and in special cases from zirconia. Use of Saggars in Ming porcelain Saggars were used to protect the precious imperial porcelain wares created at Jingdezhen from the flames of the kiln, and keep them from fusing to one another. The creation of saggars in the fifte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Brick
A brick is a type of block used to build walls, pavements and other elements in masonry construction. Properly, the term ''brick'' denotes a block composed of dried clay, but is now also used informally to denote other chemically cured construction blocks. Bricks can be joined using mortar, adhesives or by interlocking them. Bricks are usually produced at brickworks in numerous classes, types, materials, and sizes which vary with region and time period, and are produced in bulk quantities. ''Block'' is a similar term referring to a rectangular building unit composed of similar materials, but is usually larger than a brick. Lightweight bricks (also called lightweight blocks) are made from expanded clay aggregate. Fired bricks are one of the longest-lasting and strongest building materials, sometimes referred to as artificial stone, and have been used since circa 4000 BC. Air-dried bricks, also known as mud-bricks, have a history older than fired bricks, and have an addit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Titanium Dioxide
Titanium dioxide, also known as titanium(IV) oxide or titania , is the inorganic compound with the chemical formula . When used as a pigment, it is called titanium white, Pigment White 6 (PW6), or CI 77891. It is a white solid that is insoluble to water, although mineral forms can appear black. As a pigment, it has a wide range of applications, including paint, sunscreen, and food coloring. When used as a food coloring, it has E number E171. World production in 2014 exceeded 9 million tonnes. It has been estimated that titanium dioxide is used in two-thirds of all pigments, and pigments based on the oxide have been valued at a price of $13.2 billion. Structure In all three of its main dioxides, titanium exhibits octahedral geometry, being bonded to six oxide anions. The oxides in turn are bonded to three Ti centers. The overall crystal structure of rutile is tetragonal in symmetry whereas anatase and brookite are orthorhombic. The oxygen substructures are all slight di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Oxide
Sodium oxide is a chemical compound with the formula Na2 O. It is used in ceramics and glasses. It is a white solid but the compound is rarely encountered. Instead "sodium oxide" is used to describe components of various materials such as glasses and fertilizers which contain oxides that include sodium and other elements. Structure The structure of sodium oxide has been determined by X-ray crystallography. Most alkali metal oxides M2O (M = Li, Na, K, Rb) crystallise in the antifluorite structure. In this motif the positions of the anions and cations are reversed relative to their positions in CaF2, with sodium ions tetrahedrally coordinated to 4 oxide ions and oxide cubically coordinated to 8 sodium ions. Preparation Sodium oxide is produced by the reaction of sodium with sodium hydroxide, sodium peroxide, or sodium nitrite: : 2 NaOH + 2 Na → 2 Na2O + H2 To the extent that NaOH is contaminated with water, correspondingly greater amounts of sodium are employed. E ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Oxide
Potassium oxide ( K O) is an ionic compound of potassium and oxygen. It is a base. This pale yellow solid is the simplest oxide of potassium. It is a highly reactive compound that is rarely encountered. Some industrial materials, such as fertilizers and cements, are assayed assuming the percent composition that would be equivalent to K2O. Production Potassium oxide is produced from the reaction of oxygen and potassium; this reaction affords potassium peroxide, K2O2. Treatment of the peroxide with potassium produces the oxide: : K2O2 + 2 K -> 2 K2O Alternatively and more conveniently, K2O is synthesized by heating potassium nitrate with metallic potassium: :2KNO3 + 10K -> 6K2O + N2 (^) Other possibility is to heat potassium peroxide at 500 °C which decomposes at that temperature giving pure potassium oxide and oxygen. :2K2O2 -> 2K2O + O2 (^) Potassium hydroxide cannot be further dehydrated to the oxide but it can react with molten potassium to produce it, releasing hy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnesium Oxide
Magnesium oxide ( Mg O), or magnesia, is a white hygroscopic solid mineral that occurs naturally as periclase and is a source of magnesium (see also oxide). It has an empirical formula of MgO and consists of a lattice of Mg2+ ions and O2− ions held together by ionic bonding. Magnesium hydroxide forms in the presence of water (MgO + H2O → Mg(OH)2), but it can be reversed by heating it to remove moisture. Magnesium oxide was historically known as magnesia alba (literally, the white mineral from Magnesia), to differentiate it from '' magnesia negra'', a black mineral containing what is now known as manganese. Related oxides While "magnesium oxide" normally refers to MgO, the compound magnesium peroxide MgO2 is also known. According to evolutionary crystal structure prediction, MgO2 is thermodynamically stable at pressures above 116 GPa (gigapascals), and a semiconducting suboxide Mg3O2 is thermodynamically stable above 500 GPa. Because of its stability, MgO is used ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Oxide
Calcium oxide (CaO), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature. The broadly used term "'' lime''" connotes calcium-containing inorganic materials, in which carbonates, oxides and hydroxides of calcium, silicon, magnesium, aluminium, and iron predominate. By contrast, ''quicklime'' specifically applies to the single chemical compound calcium oxide. Calcium oxide that survives processing without reacting in building products such as cement is called free lime. Quicklime is relatively inexpensive. Both it and a chemical derivative ( calcium hydroxide, of which quicklime is the base anhydride) are important commodity chemicals. Preparation Calcium oxide is usually made by the thermal decomposition of materials, such as limestone or seashells, that contain calcium carbonate (CaCO3; mineral calcite) in a lime kiln. This is accomplished by heating the material to above ,Me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron(III) Oxide
Iron(III) oxide or ferric oxide is the inorganic compound with the formula Fe2O3. It is one of the three main oxides of iron, the other two being iron(II) oxide (FeO), which is rare; and iron(II,III) oxide (Fe3O4), which also occurs naturally as the mineral magnetite. As the mineral known as hematite, Fe2O3 is the main source of iron for the steel industry. Fe2O3 is readily attacked by acids. Iron(III) oxide is often called rust, and to some extent this label is useful, because rust shares several properties and has a similar composition; however, in chemistry, rust is considered an ill-defined material, described as Hydrous ferric oxide. Structure Fe2O3 can be obtained in various polymorphs. In the main one, α, iron adopts octahedral coordination geometry. That is, each Fe center is bound to six oxygen ligands. In the γ polymorph, some of the Fe sit on tetrahedral sites, with four oxygen ligands. Alpha phase α-Fe2O3 has the rhombohedral, corundum (α-Al2O3) structure and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Loss On Ignition
Loss on ignition (LOI) is a test used in inorganic analytical chemistry and soil science, particularly in the analysis of minerals and the chemical makeup of soil. It consists of strongly heating ( "igniting") a sample of the material at a specified temperature, allowing volatile substances to escape, until its mass ceases to change. This may be done in air, or in some other reactive or inert atmosphere. The simple test typically consists of placing a few grams of the material in a tared, pre-ignited crucible and determining its mass, placing it in a temperature-controlled furnace for a set time, cooling it in a controlled (e.g. water-free, CO2-free) atmosphere, and redetermining the mass. The process may be repeated to show that mass-change is complete. A variant of the test in which mass-change is continually monitored as the temperature is changed, is thermogravimetry. Theory The loss on ignition is reported as part of an elemental or oxide analysis of a mineral. The volat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
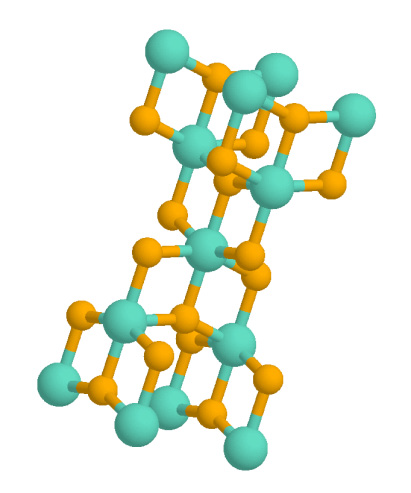
Silicon Dioxide
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , most commonly found in nature as quartz and in various living organisms. In many parts of the world, silica is the major constituent of sand. Silica is one of the most complex and most abundant families of materials, existing as a compound of several minerals and as a synthetic product. Notable examples include fused quartz, fumed silica, silica gel, opal and aerogels. It is used in structural materials, microelectronics (as an electrical insulator), and as components in the food and pharmaceutical industries. Structure In the majority of silicates, the silicon atom shows tetrahedral coordination, with four oxygen atoms surrounding a central Si atomsee 3-D Unit Cell. Thus, SiO2 forms 3-dimensional network solids in which each silicon atom is covalently bonded in a tetrahedral manner to 4 oxygen atoms. In contrast, CO2 is a linear molecule. The starkly different structures of the d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |