|
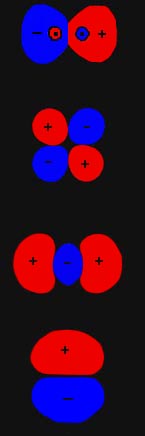
Homonuclear Triatomic Molecules
In chemistry, homonuclear molecules, or elemental molecules, or homonuclear species, are molecules composed of only one element. Homonuclear molecules may consist of various numbers of atoms. The size of the molecule an element can form depends on the element's properties, and some elements form molecules of more than one size. The most familiar homonuclear molecules are diatomic molecules, which consist of two atoms, although not all diatomic molecules are homonuclear. Homonuclear diatomic molecules include hydrogen (), oxygen (), nitrogen () and all of the halogens. Ozone () is a common triatomic homonuclear molecule. Homonuclear tetratomic molecules include arsenic () and phosphorus (). Allotropes are different chemical forms of the same element (not containing any other element). In that sense, allotropes are all homonuclear. Many elements have multiple allotropic forms. In addition to the most common form of gaseous oxygen, , and ozone, there are other allotropes of oxygen. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during chemical reaction, reactions with other chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the prop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diatomic Element
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear. Otherwise, if a diatomic molecule consists of two different atoms, such as carbon monoxide () or nitric oxide (), the molecule is said to be heteronuclear. The bond in a homonuclear diatomic molecule is non-polar. The only chemical elements that form stable homonuclear diatomic molecules at standard temperature and pressure (STP) (or at typical laboratory conditions of 1 bar and 25 °C) are the gases hydrogen (), nitrogen (), oxygen (), fluorine (), and chlorine (), and the liquid bromine (). The noble gases (helium, neon, argon, krypton, xenon, and radon) are also gases at STP, but they are monatomic. The homonuclear diatomic gases and noble gases together are called "elemental gases" or "molecular gases", to distinguish them from ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
:Category:Homonuclear Diatomic Molecules
Homonuclear diatomic molecules are molecule A molecule is a group of two or more atoms that are held together by Force, attractive forces known as chemical bonds; depending on context, the term may or may not include ions that satisfy this criterion. In quantum physics, organic chemi ...s composed of two atoms that are both the same. diatomic molecules Homonuclear molecules ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heteronuclear Molecule
A heteronuclear molecule is a molecule composed of atoms of more than one chemical element. For example, a molecule of water (H2O) is heteronuclear because it has atoms of two different elements, hydrogen (H) and oxygen (O). Similarly, a heteronuclear ion is an ion that contains atoms of more than one chemical element. For example, the carbonate ion () is heteronuclear because it has atoms of carbon (C) and oxygen (O). The lightest heteronuclear ion is the helium hydride ion (HeH+). This is in contrast to a homonuclear ion, which contains all the same kind of atom, such as the dihydrogen cation, or atomic ions that only contain one atom such as the hydrogen anion (H−). References See also *Homonuclear molecule *Chemical compound A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one elemen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Graphite
Graphite () is a Crystallinity, crystalline allotrope (form) of the element carbon. It consists of many stacked Layered materials, layers of graphene, typically in excess of hundreds of layers. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on a large scale (1.3million metric tons per year in 2022) for uses in many critical industries including refractories (50%), lithium-ion batteries (18%), foundries (10%), and lubricants (5%), among others (17%). Graphite converts to diamond under extremely high pressure and temperature. Graphite's low cost, thermal and chemical inertness and characteristic conductivity of heat and electricity finds numerous applications in high energy and high temperature processes. Types and varieties Graphite can occur naturally or be produced synthetically. Natural graphite is obtained from naturally occurring geologic deposits and synthetic graphite is produced t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Diamond is tasteless, odourless, strong, brittle solid, colourless in pure form, a poor conductor of electricity, and insoluble in water. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of carbon at Standard temperature and pressure, room temperature and pressure, but diamond is metastable and converts to it at a negligible rate under those conditions. Diamond has the highest Scratch hardness, hardness and thermal conductivity of any natural material, properties that are used in major industrial applications such as cutting and polishing tools. Because the arrangement of atoms in diamond is extremely rigid, few types of impurity can contaminate it (two exceptions are boron and nitrogen). Small numbers of lattice defect, defects or impurities (about one per million of lattice atoms) can color ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon
Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. It belongs to group 14 of the periodic table. Carbon makes up about 0.025 percent of Earth's crust. Three Isotopes of carbon, isotopes occur naturally, carbon-12, C and carbon-13, C being stable, while carbon-14, C is a radionuclide, decaying with a half-life of 5,700 years. Carbon is one of the timeline of chemical element discoveries#Pre-modern and early modern discoveries, few elements known since antiquity. Carbon is the 15th abundance of elements in Earth's crust, most abundant element in the Earth's crust, and the abundance of the chemical elements, fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. Carbon's abundance, its unique diversity of organic compounds, and its unusual abi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Octasulfur
Octasulfur is an inorganic substance with the chemical formula . It is an odourless and tasteless yellow solid, and is a major industrial chemical. It is the most common allotrope of sulfur Sulfur ( American spelling and the preferred IUPAC name) or sulphur ( Commonwealth spelling) is a chemical element; it has symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms ... and occurs widely in nature.Steudel, R., "Homocyclic Sulfur Molecules", Topics Curr. Chem. 1982, 102, 149. Nomenclature The name octasulfur is the most commonly used for this chemical. It is systematically named ''cyclo''-octasulfur (which is the preferred IUPAC name) and cyclooctasulfane. It is also the final member of the thiocane heterocylic series, where every carbon atom is substituted with a sulfur atom, thus this sulfur allotrope is systematically named octathiocane as well. Structure The chemical consists of rings of 8 sulfur ato ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexasulfur
Hexasulfur is an inorganic chemical with the chemical formula . This allotrope was first prepared by M. R. Engel in 1891 by treating thiosulfate with HCl. ''Cyclo''- is orange-red and forms a rhombohedral crystal. It is called ρ-sulfur, ε-sulfur, Engel's sulfur and Aten's sulfur. Another method of preparation involves the reaction of a polysulfane with sulfur monochloride: : (dilute solution in diethyl ether) Nomenclature The name hexasulfur is the most commonly used and preferred IUPAC name and is constructed according to the compositional nomenclature, and cyclohexasulfane. It is also the final member of the thiane heterocyclic series, where every carbon atom is substituted with a sulfur atom, thus the systematic name hexathiane, a valid IUPAC name, is constructed according to the substitutive nomenclature. Another valid IUPAC systematic name ''cyclo''-hexasulfur is constructed according to the additive nomenclature. Structure This chemical consists of rings of 6 sulfu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trisulfur
The molecule, known as trisulfur, sulfur trimer, thiozone, or triatomic sulfur, is a cherry-red allotrope of sulfur. It comprises about 10% of vaporised sulfur at and . It has been observed at cryogenic temperatures as a solid. Under ordinary conditions it converts to cyclooctasulfur. : Structure and bonding In terms of structure and bonding and ozone () are similar. Both adopt bent structures and are diamagnetic. Although represented with S=S double bonds, the bonding situation is more complex. The S–S distances are equivalent and are , and with an angle at the central atom of . However, cyclic , where the sulfur atoms are arranged in an equilateral triangle with three single bonds (similar to cyclic ozone and cyclopropane), is calculated to be lower in energy than the bent structure experimentally observed. A similar structure has been predicted for ozone, but has not been observed. The name thiozone was invented by Hugo Erdmann in 1908 who hypothesized that compr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Disulfur
Disulfur is the diatomic molecule with the formula S2. It is analogous to the dioxygen molecule but rarely occurs at room temperature. This violet gas is the dominant species in hot sulfur vapors. S2 is one of the minor components of the atmosphere of Io, which is predominantly composed of SO2. The instability of S2 is usually described in the context of the double bond rule. Synthesis This violet gas is generated by heating sulfur and is the predominant species above 720 °C, comprising 80% of all vapor species at 530°C and 100 mm Hg, and 99% of the vapor at low pressure (1 mm Hg) at 730 °C. Disulfur can be produced when an atmosphere of COS is irradiated with UV light using a mercury photosensitizer or when CS2, H2S2, S2Cl2 or C2H4S, PSF3 or COS are irradiated. Natural occurrence Gaseous disulfur has been detected emanating from the surface of Jupiter's moon Io, from the vicinity of Pele volcano. Properties The ground state of S2 is a triplet: a dir ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |