|
Hemoproteins
A hemeprotein (or haemprotein; also hemoprotein or haemoprotein), or heme protein, is a protein that contains a heme prosthetic group. They are a very large class of metalloproteins. The heme group confers functionality, which can include oxygen carrying, oxygen reduction, electron transfer, and other processes. Heme is bound to the protein either covalently or noncovalently or both. The heme consists of iron cation bound at the center of the conjugate base of the porphyrin, as well as other ligands attached to the "axial sites" of the iron. The porphyrin ring is a planar dianionic, tetradentate ligand. The iron is typically Fe2+ or Fe3+. One or two ligands are attached at the axial sites. The porphyrin ring has four nitrogen atoms that bind to the iron, leaving two other coordination positions of the iron available for bonding to the histidine of the protein and a divalent atom. Hemeproteins probably evolved to incorporate the iron atom contained within the protoporphyr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heme
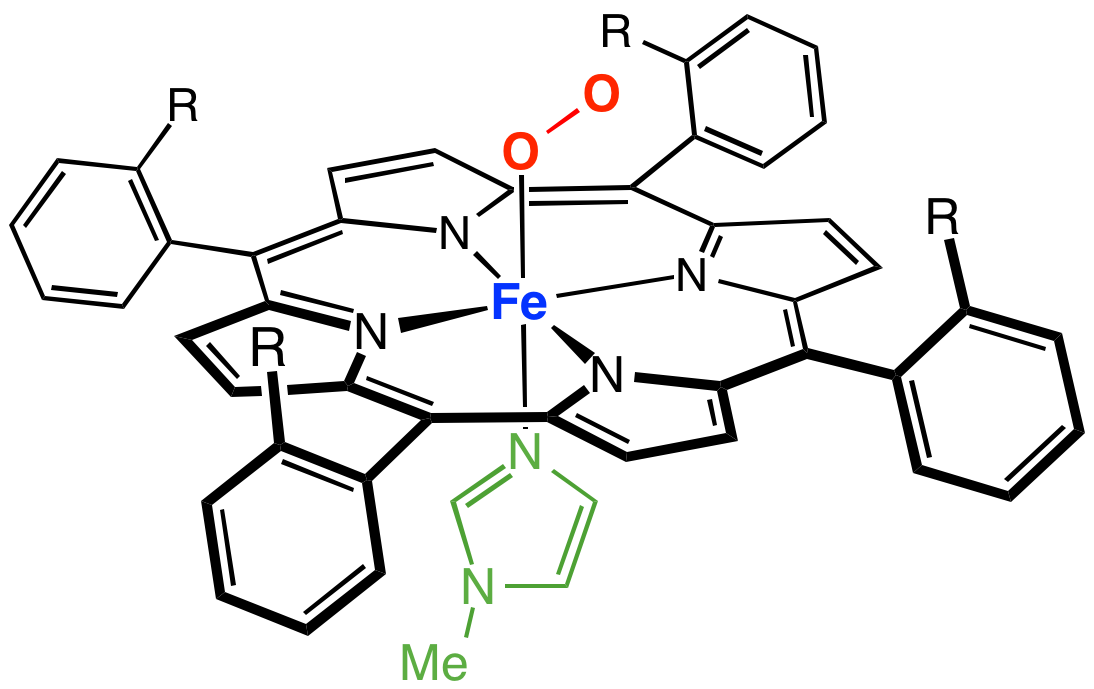
Heme (American English), or haem (Commonwealth English, both pronounced /Help:IPA/English, hi:m/ ), is a ring-shaped iron-containing molecule that commonly serves as a Ligand (biochemistry), ligand of various proteins, more notably as a Prosthetic group, component of hemoglobin, which is necessary to bind oxygen in the bloodstream. It is composed of four pyrrole rings with 2 Vinyl group, vinyl and 2 propionic acid side chains. Heme is biosynthesized in both the bone marrow and the liver. Heme plays a critical role in multiple different redox reactions in mammals, due to its ability to carry the oxygen molecule. Reactions include oxidative metabolism (cytochrome c oxidase, succinate dehydrogenase), xenobiotic detoxification via cytochrome P450 pathways (including Drug metabolism, metabolism of some drugs), gas sensing (Guanylate cyclase, guanyl cyclases, nitric oxide synthase), and microRNA processing (DGCR8). Heme is a coordination complex "consisting of an iron ion coordinated ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cytochrome C Oxidase
The enzyme cytochrome c oxidase or Complex IV (was , now reclassified as a translocasEC 7.1.1.9 is a large transmembrane protein complex found in bacteria, archaea, and the mitochondria of eukaryotes. It is the last enzyme in the Cellular respiration, respiratory electron transport chain of cell (biology), cells located in the membrane. It receives an electron from each of four cytochrome c molecules and transfers them to one oxygen molecule and four protons, producing two molecules of water. In addition to binding the four protons from the inner aqueous phase, it transports another four protons across the membrane, increasing the transmembrane difference of proton electrochemical potential, which the ATP synthase then uses to synthesize Adenosine triphosphate, ATP. Structure The complex The complex is a large integral membrane protein composed of several Cofactor (biochemistry)#Metal ions, metal prosthetic sites and 13 protein subunits in mammals. In mammals, ten subunits a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metalloprotein
Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large proportion of all proteins are part of this category. For instance, at least 1000 human proteins (out of ~20,000) contain zinc-binding protein domains although there may be up to 3000 human zinc metalloproteins. Abundance It is estimated that approximately half of all proteins contain a metal. In another estimate, about one quarter to one third of all proteins are proposed to require metals to carry out their functions. Thus, metalloproteins have many different functions in cells, such as storage and transport of proteins, enzymes and signal transduction proteins, or infectious diseases. The abundance of metal binding proteins may be inherent to the amino acids that proteins use, as even artificial proteins without evolutionary history will readily bind metals. Most metals in the human body are bound to proteins. For instance, the relatively high concentration of iron in the human body ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Peroxidase
Peroxidases or peroxide reductases ( EC numberbr>1.11.1.x are a large group of enzymes which play a role in various biological processes. They are named after the fact that they commonly break up peroxides, and should not be confused with other enzymes that ''produce'' peroxide, which are often oxidases. Functionality Peroxidases typically catalyze a reaction of the form: :ROOR' + \overset + 2H+ -> ce + R'OH Optimal substrates For many of these enzymes the optimal substrate is hydrogen peroxide, but others are more active with organic hydroperoxides such as lipid peroxides. Peroxidases can contain a heme cofactor in their active sites, or alternately redox-active cysteine or selenocysteine residues. The nature of the electron donor is very dependent on the structure of the enzyme. * For example, horseradish peroxidase can use a variety of organic compounds as electron donors and acceptors. Horseradish peroxidase has an accessible active site, and many compounds can re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalase
Catalase is a common enzyme found in nearly all living organisms exposed to oxygen (such as bacteria, plants, and animals) which catalyzes the decomposition of hydrogen peroxide to water and oxygen. It is a very important enzyme in protecting the cell from oxidative damage by reactive oxygen species (ROS). Catalase has one of the highest turnover numbers of all enzymes; one catalase molecule can convert millions of hydrogen peroxide molecules to water and oxygen each second. Catalase is a tetramer of four polypeptide chains, each over 500 amino acids long. It contains four iron-containing heme groups that allow the enzyme to react with hydrogen peroxide. The optimum pH for human catalase is approximately 7, and has a fairly broad maximum: the rate of reaction does not change appreciably between pH 6.8 and 7.5. The pH optimum for other catalases varies between 4 and 11 depending on the species. The optimum temperature also varies by species. Structure Human catalase for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cytochrome C Oxidase
The enzyme cytochrome c oxidase or Complex IV (was , now reclassified as a translocasEC 7.1.1.9 is a large transmembrane protein complex found in bacteria, archaea, and the mitochondria of eukaryotes. It is the last enzyme in the Cellular respiration, respiratory electron transport chain of cell (biology), cells located in the membrane. It receives an electron from each of four cytochrome c molecules and transfers them to one oxygen molecule and four protons, producing two molecules of water. In addition to binding the four protons from the inner aqueous phase, it transports another four protons across the membrane, increasing the transmembrane difference of proton electrochemical potential, which the ATP synthase then uses to synthesize Adenosine triphosphate, ATP. Structure The complex The complex is a large integral membrane protein composed of several Cofactor (biochemistry)#Metal ions, metal prosthetic sites and 13 protein subunits in mammals. In mammals, ten subunits a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Leghemoglobin
Leghemoglobin (also leghaemoglobin or legoglobin) is an oxygen-carrying phytoglobin found in the nitrogen-fixing root nodules of leguminous plants. It is produced by these plants in response to the roots being colonized by nitrogen-fixing bacteria, termed rhizobia, as part of the symbiotic interaction between plant and bacterium: roots not colonized by ''Rhizobium'' do not synthesise leghemoglobin. Leghemoglobin has close chemical and structural similarities to hemoglobin, and, like hemoglobin, is red in colour. It was originally thought that the heme prosthetic group for plant leghemoglobin was provided by the bacterial symbiont within symbiotic root nodules. However, subsequent work shows that the plant host strongly expresses heme biosynthesis genes within nodules, and that activation of those genes correlates with leghemoglobin gene expression in developing nodules. In plants colonised by ''Rhizobium'', such as alfalfa or soybeans, the presence of oxygen in the root nodule ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cytoglobin
Cytoglobin is the protein product of CYGB, a human and mammalian gene. Cytoglobin is a globin molecule ubiquitously expressed in all tissues and most notably utilized in marine mammals. It was discovered in 2001 in hepatic stellate cells during liver fibrosis. Thus, it was originally called "stellate cell activated protein" or STAP. It received its current name in 2002. It is thought to help in the distribution and storage of oxygen as well as protect against hypoxia by scavenging reactive oxygen species . The predicted function of cytoglobin is the facilitation of oxygen among tissues that don't express myoglobin. Function Cytoglobin is a ubiquitously expressed hexacoordinate hemoglobin that may facilitate diffusion of oxygen through tissues, scavenge nitric oxide or reactive oxygen species, or serve a protective function during oxidative stress. Structure Cytoglobin has 30-40% sequence homology with myoglobin, and has a similar oxygen binding affinity. One of the major ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neuroglobin
Neuroglobin is a member of the vertebrate globin family involved in cellular oxygen homeostasis and reactive oxygen/nitrogen scavenging. It is an intracellular hemoprotein expressed in the central and peripheral nervous system, cerebrospinal fluid, retina and endocrine tissues. Neuroglobin is a monomer that reversibly binds oxygen with an affinity higher than that of hemoglobin. It also increases oxygen availability to brain tissue and provides protection under hypoxic or ischemic conditions, potentially limiting brain damage. Neuroglobin were in the past found only in vertebrate neurons, but recently in 2013, were found in the neurons of unrelated protostomes, like photosynthetic acoel as well as radiata such as jellyfish. In addition to neurons, neuroglobin is present in astrocytes in certain pathologies of the rodent brain and in the physiological seal brain. This is thought to be due to convergent evolution. It is of ancient evolutionary origin, and is homologous to nerve glob ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Myoglobin
Myoglobin (symbol Mb or MB) is an iron- and oxygen-binding protein found in the cardiac and skeletal muscle, skeletal Muscle, muscle tissue of vertebrates in general and in almost all mammals. Myoglobin is distantly related to hemoglobin. Compared to hemoglobin, myoglobin has a higher affinity for oxygen and does not have cooperative binding with oxygen like hemoglobin does. Myoglobin consists of non-polar amino acids at the core of the globulin, where the heme group is non-covalently bounded with the surrounding polypeptide of myoglobin. In humans, myoglobin is found in the bloodstream only after Strain (injury), muscle injury. (Google books link is the 2008 edition) High concentrations of myoglobin in muscle cells allow organisms to hold their breath for a longer period of time. Diving mammals such as whales and seals have muscles with particularly high abundance of myoglobin. Myoglobin is found in Type I muscle, Type II A, and Type II B; although many older texts describe myo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cytochrome B
Cytochrome b is a protein found in the membranes of aerobic cells. In eukaryotic mitochondria (inner membrane) and in aerobic prokaryotes, cytochrome b is a component of respiratory chain complex III () — also known as the bc1 complex or ubiquinol-cytochrome c reductase. In plant chloroplasts and cyanobacteria, there is a homologous protein, cytochrome b6, a component of the plastoquinone-plastocyanin reductase (), also known as the b6f complex. These complexes are involved in electron transport, the pumping of protons to create a proton-motive force ( PMF). This proton gradient is used for the generation of ATP. These complexes play a vital role in cells. Structure and function Cytochrome b/b6 is an integral membrane protein of approximately 400 amino acid residues that probably has 8 transmembrane segments. In plants and cyanobacteria, cytochrome b6 consists of two protein subunits encoded by the petB and petD genes. Cytochrome b/b6 non-covalently binds two heme ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |