|
Cis–trans Isomerism
''Cis''–''trans'' isomerism, also known as geometric isomerism, describes certain arrangements of atoms within molecules. The prefixes "''cis''" and "''trans''" are from Latin: "this side of" and "the other side of", respectively. In the context of chemistry, ''cis'' indicates that the functional groups (substituents) are on the same side of some plane, while ''trans'' conveys that they are on opposing (transverse) sides. ''Cis''–''trans'' isomers are stereoisomers, that is, pairs of molecules which have the same formula but whose functional groups are in different orientations in three-dimensional space. ''Cis'' and ''trans'' isomers occur both in organic molecules and in inorganic coordination complexes. ''Cis'' and ''trans'' descriptors are not used for cases of conformational isomerism where the two geometric forms easily interconvert, such as most open-chain single-bonded structures; instead, the terms "''syn''" and "''anti''" are used. According to IUPAC, "geome ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cis-2-pentene
CIS may refer to: Computing * Card information structure, formatting and organization data stored on a PC card * Center for Internet Security, cybersecurity benchmarks, controls, practices and tools * Center for Internet and Society (other) * Comodo Internet Security suite * CompuServe Information Service, a US commercial online service * Computer and information science, a field that emphasizes both computing and informatics * Computer information systems, technologies which process data to solve business problems * Configuration interaction singles, a quantum-chemical method for computing electronic states * Contact image sensor, a technology developed for optical flatbed scanners * Continuous ink system, for ink-jet printers Education * Calcutta International School, in India * Canadian International School (other) ** Canadian International School (Bangalore) ** Canadian International School Vietnam ** Canadian International School of Sanya, in Chin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fumaric Acid
Fumaric acid or ''trans''-butenedioic acid is an organic compound with the formula HO2CCH=CHCO2H. A white solid, fumaric acid occurs widely in nature. It has a fruit-like taste and has been used as a food additive. Its E number is E297. The salts and esters are known as fumarates. Fumarate can also refer to the ion (in solution). Fumaric acid is the ''trans'' isomer of butenedioic acid, while maleic acid is the ''cis'' isomer. Biosynthesis and occurrence It is produced in eukaryotic organisms from succinate in complex 2 of the electron transport chain via the enzyme succinate dehydrogenase. Fumaric acid is found in fumitory (''Fumaria officinalis''), bolete mushrooms (specifically ''Boletus fomentarius var. pseudo-igniarius''), lichen, and Iceland moss. Fumarate is an intermediate in the citric acid cycle used by cells to produce energy in the form of adenosine triphosphate (ATP) from food. It is formed by the oxidation of succinate by the enzyme succinate dehydr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Maleic Acid
Maleic acid or ''cis''-butenedioic acid is an organic compound that is a dicarboxylic acid, a molecule with two carboxyl groups. Its chemical formula is HO2CCH=CHCO2H. Maleic acid is the ''cis'' Cis–trans isomerism, isomer of butenedioic acid, whereas fumaric acid is the ''trans'' isomer. Maleic acid is mainly used as a precursor to fumaric acid, and relative to its parent maleic anhydride, which has many applications. Etymology 'Maleic acid' from the French altered from , malic acid, from which it was synthesised. The word 'malic' is derived from Latin , meaning 'apple'. Physical properties Maleic acid has a heat of combustion of −1,355 kJ/mol., 22.7 kJ/mol higher than that of fumaric acid. Maleic acid is more soluble in water than fumaric acid. The melting point of maleic acid (135 °C) is also much lower than that of fumaric acid (287 °C). As confirmed by X-ray crystallography, maleic acid is planar. Two normal hydrogen bonds are observed, one Intramolec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quadrupole Moment
A quadrupole or quadrapole is one of a sequence of configurations of things like electric charge or current, or gravitational mass that can exist in ideal form, but it is usually just part of a multipole expansion of a more complex structure reflecting various orders of complexity. Mathematical definition The quadrupole moment tensor ''Q'' is a rank-two tensor—3×3 matrix. There are several definitions, but it is normally stated in the traceless form (i.e. Q_ + Q_ + Q_ = 0). The quadrupole moment tensor has thus nine components, but because of transposition symmetry and zero-trace property, in this form only five of these are independent. For a discrete system of \ell point charges or masses in the case of a gravitational quadrupole, each with charge q_\ell, or mass m_\ell, and position \mathbf_\ell = \left(r_, r_, r_\right) relative to the coordinate system origin, the components of the ''Q'' matrix are defined by: Q_ = \sum_\ell q_\ell\left(3r_ r_ - \left\, \mathbf_\ell ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
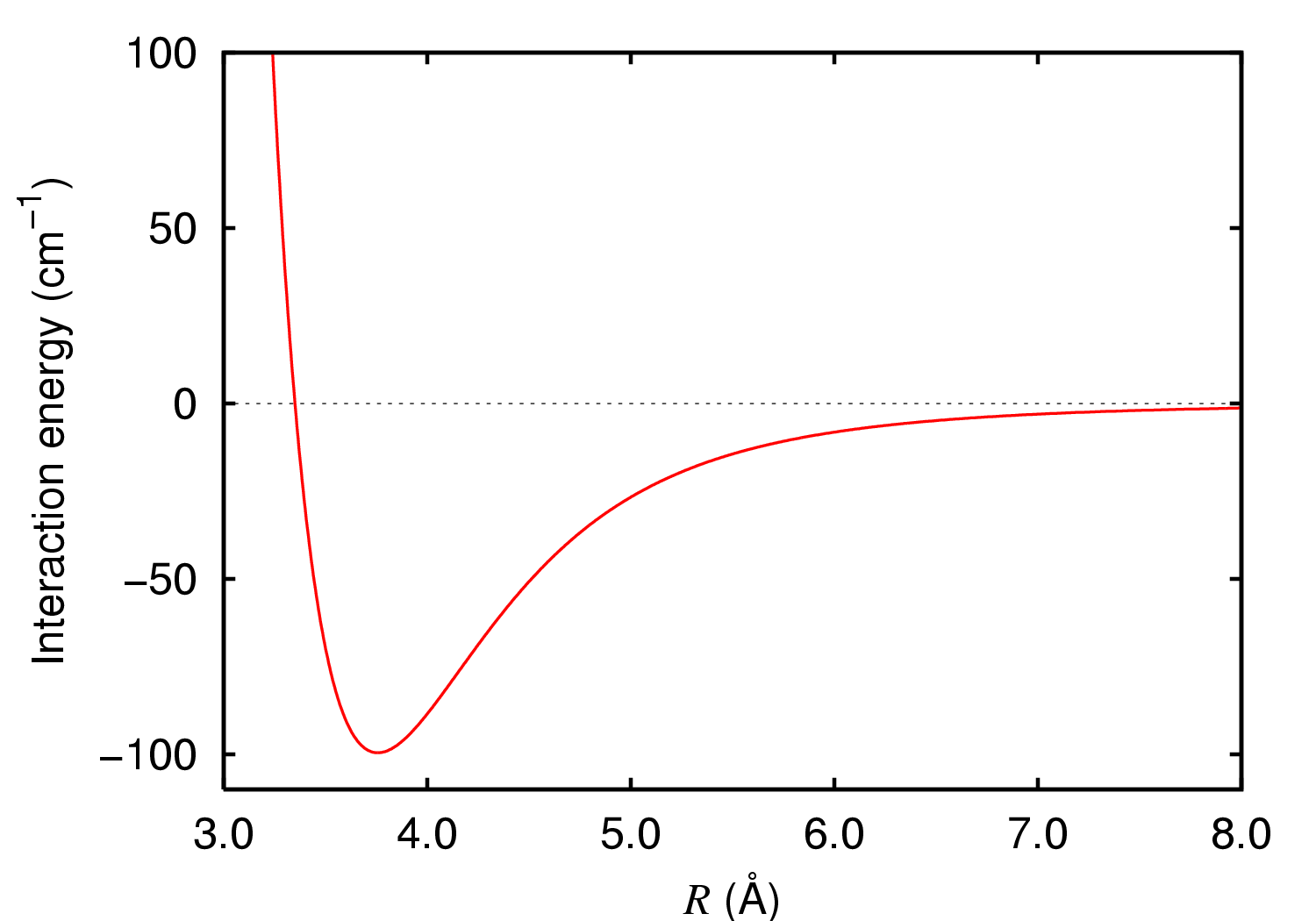
London Dispersion Forces
London dispersion forces (LDF, also known as dispersion forces, London forces, instantaneous dipole–induced dipole forces, fluctuating induced dipole bonds or loosely as van der Waals forces) are a type of intermolecular force acting between atoms and molecules that are normally electrically symmetric; that is, the electrons are symmetrically distributed with respect to the nucleus. They are part of the van der Waals forces. The LDF is named after the German physicist Fritz London. They are the weakest of the intermolecular forces. Introduction The electron distribution around an atom or molecule undergoes fluctuations in time. These fluctuations create instantaneous electric fields which are felt by other nearby atoms and molecules, which in turn adjust the spatial distribution of their own electrons. The net effect is that the fluctuations in electron positions in one atom induce a corresponding redistribution of electrons in other atoms, such that the electron motions beco ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intermolecular Forces
An intermolecular force (IMF; also secondary force) is the force that mediates interaction between molecules, including the electromagnetic forces of attraction or repulsion which act between atoms and other types of neighbouring particles (e.g. atoms or ions). Intermolecular forces are weak relative to intramolecular forces – the forces which hold a molecule together. For example, the covalent bond, involving sharing electron pairs between atoms, is much stronger than the forces present between neighboring molecules. Both sets of forces are essential parts of force fields frequently used in molecular mechanics. The first reference to the nature of microscopic forces is found in Alexis Clairaut's work ''Théorie de la figure de la Terre,'' published in Paris in 1743. Other scientists who have contributed to the investigation of microscopic forces include: Laplace, Gauss, Maxwell, Boltzmann and Pauling. Attractive intermolecular forces are categorized into the following type ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bond Dipole Moment
In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively charged end. Polar molecules must contain one or more polar bonds due to a difference in electronegativity between the bonded atoms. Molecules containing polar bonds have no molecular polarity if the bond dipoles cancel each other out by symmetry. Polar molecules interact through dipole-dipole intermolecular forces and hydrogen bonds. Polarity underlies a number of physical properties including surface tension, solubility, and melting and boiling points. Polarity of bonds Not all atoms attract electrons with the same force. The amount of "pull" an atom exerts on its electrons is called its electronegativity. Atoms with high electronegativitiessuch as fluorine, oxygen, and nitrogenexert a greater pull on electrons than atoms with lower electronegativities such as alkali metals and alkaline e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,2-dichloroethene
1,2-Dichloroethylene or 1,2-DCE is the name for a pair of organochlorine compounds with the molecular formula . The two compounds are isomers, each being colorless liquids with a sweet odor. It can exist as either of two geometric isomers, ''cis''-1,2-dichloroethene or ''trans''-1,2-dichloroethene, but is often used as a mixture of the two. They have modest solubility in water. These compounds have some applications as a degreasing solvent. In contrast to most ''cis-trans'' compounds, the ''Z'' isomer (''cis'') is more stable than the ''E'' isomer (''trans'') by 0.4 kcal/mol. Production, uses and reactions ''cis''-DCE, the ''Z'' isomer, is obtainable by the controlled chlorination of acetylene: :CH + Cl → CHCl Industrially both isomers arise as byproducts of the production of vinyl chloride, which is produced on a vast scale. Unlike 1,1-dichloroethylene, the 1,2-dichloroethylene isomers do not polymerize. ''trans''-1,2-DCE has applications including electronics c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pentene
Pentenes are alkenes with the chemical formula . Each molecule contains one double bond within its molecular structure. Six different compounds are in this class, differing from each other by whether the carbon atoms are attached linearly or in a branched structure and whether the double bond has a ''cis'' or ''trans'' form. Straight-chain isomers 1-Pentene is an alpha-olefin. Most often, 1-pentene is made as a byproduct of catalytic or thermal cracking of petroleum or during the production of ethylene and propylene via thermal cracking of hydrocarbon fractions. As of 2010s, the only commercial manufacturer of 1-pentene was Sasol Ltd., where it is separated from crude by the Fischer-Tropsch process. 2-Pentene has two geometric isomers: ''cis''-2-pentene and ''trans''-2-pentene. ''Cis''-2-Pentene is used in olefin metathesis. Branched-chain isomers The branched isomers are 2-methylbut-1-ene, 3-methylbut-1-ene (isopentene), and 2-methylbut-2-ene (isoamylene). Isoamyl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |