|
Amides
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent any group, typically organyl groups or hydrogen atoms. The amide group is called a peptide bond when it is part of the main chain of a protein, and an isopeptide bond when it occurs in a side chain, as in asparagine and glutamine. It can be viewed as a derivative of a carboxylic acid () with the hydroxyl group () replaced by an amino group (); or, equivalently, an acyl (alkanoyl) group () joined to an amino group. Common amides are formamide (), acetamide (), benzamide (), and dimethylformamide (). Some uncommon examples of amides are ''N''-chloroacetamide () and chloroformamide (). Amides are qualified as primary, secondary, and tertiary according to the number of acyl groups bounded to the nitrogen atom. Nomenclature The core of amides is called the amide group (specifically, carboxamide group). In the usual no ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amino
In chemistry, amines (, ) are organic compounds that contain carbon-nitrogen bonds. Amines are formed when one or more hydrogen atoms in ammonia are replaced by alkyl or aryl groups. The nitrogen atom in an amine possesses a lone pair of electrons. Amines can also exist as hetero cyclic compounds. Aniline is the simplest aromatic amine, consisting of a benzene ring bonded to an amino group. Amines are classified into three types: primary (1°), secondary (2°), and tertiary (3°) amines. Primary amines (1°) contain one alkyl or aryl substituent and have the general formula RNH2. Secondary amines (2°) have two alkyl or aryl groups attached to the nitrogen atom, with the general formula R2NH. Tertiary amines (3°) contain three substituent groups bonded to the nitrogen atom, and are represented by the formula R3N. The functional group present in primary amines is called the amino group. Classification of amines Amines can be classified according to the nature and number o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acyl Group
In chemistry, an acyl group is a moiety derived by the removal of one or more hydroxyl groups from an oxoacid, including inorganic acids. It contains a double-bonded oxygen atom and an organyl group () or hydrogen in the case of formyl group (). In organic chemistry, the acyl group (IUPAC name alkanoyl if the organyl group is alkyl) is usually derived from a carboxylic acid, in which case it has the formula , where R represents an organyl group or hydrogen. Although the term is almost always applied to organic compounds, acyl groups can in principle be derived from other types of acids such as sulfonic acids and phosphonic acids. In the most common arrangement, acyl groups are attached to a larger molecular fragment, in which case the carbon and oxygen atoms are linked by a double bond. Reactivity trends There are five main types of acyl derivatives. Acid halides are the most reactive towards nucleophiles, followed by anhydrides, esters, and amides. Carboxylate ions are esse ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylformamide
Dimethylformamide, DMF is an organic compound with the chemical formula . Its structure is . Commonly abbreviated as DMF (although this initialism is sometimes used for 2,5-dimethylfuran, dimethylfuran, or dimethyl fumarate), this colourless liquid is Miscibility, miscible with Water (molecule), water and the majority of organic liquids. DMF is a common solvent for chemical reactions. Dimethylformamide is odorless, but chemical purity, technical-grade or degraded samples often have a fishy smell due to impurity of dimethylamine. Dimethylamine degradation impurities can be removed by Sparging (chemistry), sparging samples with an inert gas such as argon or by sonication, sonicating the samples under reduced pressure. As its name indicates, it is structurally related to formamide, having two methyl groups in the place of the two hydrogens. DMF is a polar molecule, polar (hydrophilic) aprotic solvent with a high boiling point. It facilitates reactions that follow polar mechanisms, such ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboxylic Acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an Substituent, R-group. The general formula of a carboxylic acid is often written as or , sometimes as with R referring to an organyl group (e.g., alkyl, alkenyl, aryl), or hydrogen, or other groups. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion. Examples and nomenclature Carboxylic acids are commonly identified by their trivial names. They often have the suffix ''-ic acid''. IUPAC-recommended names also exist; in this system, carboxylic acids have an ''-oic acid'' suffix. For example, butyric acid () is butanoic acid by IUPAC guidelines. For nomenclature of complex molecules containing a carboxylic acid, the carboxyl can be considered position one of the parent chain even if there are other substituents, such as 3-chloropropanoic acid. Alternately, it can be named ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylamine
Dimethylamine is an organic compound with the formula (CH3)2NH. This secondary amine is a colorless, flammable gas with an ammonia-like odor. Dimethylamine is commonly encountered commercially as a solution in water at concentrations up to around 40%. An estimated 270,000 tons were produced in 2005. Structure and synthesis The molecule consists of a nitrogen atom with two methyl substituents and one hydrogen. Dimethylamine is a weak base and the pKa of the ammonium CH3--CH3 is 10.73, a value above methylamine (10.64) and trimethylamine (9.79). Dimethylamine reacts with acids to form salts, such as dimethylamine hydrochloride, an odorless white solid with a melting point of 171.5 °C. Dimethylamine is produced by catalytic reaction of methanol and ammonia at elevated temperatures and high pressure: : Natural occurrence Dimethylamine is found quite widely distributed in animals and plants, and is present in many foods at the level of a few mg/kg. Uses Dimethylamine is a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethanamide
Acetamide (systematic name: ethanamide) is an organic compound with the formula CH3CONH2. It is an amide derived from ammonia and acetic acid. It finds some use as a plasticizer and as an industrial solvent. The related compound ''N'',''N''-dimethylacetamide (DMA) is more widely used, but it is not prepared from acetamide. Acetamide can be considered an intermediate between acetone, which has two methyl (CH3) groups either side of the carbonyl (CO), and urea which has two amide (NH2) groups in those locations. Acetamide is also a naturally occurring mineral with the IMA symbol: Ace. Production Laboratory scale Acetamide can be produced in the laboratory from ammonium acetate by dehydration: : H4CH3CO2] → CH3C(O)NH2 + H2O Alternatively acetamide can be obtained in excellent yield via ammonolysis of acetylacetone under conditions commonly used in reductive amination. It can also be made from anhydrous acetic acid, acetonitrile and very well dried hydrogen chloride gas, u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetic Acid
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula (also written as , , or ). Vinegar is at least 4% acetic acid by volume, making acetic acid the main component of vinegar apart from water. Historically, vinegar was produced from the third century BC and was likely the first acid to be produced in large quantities. Acetic acid is the second simplest carboxylic acid (after formic acid). It is an important Reagent, chemical reagent and industrial chemical across various fields, used primarily in the production of cellulose acetate for photographic film, polyvinyl acetate for wood Adhesive, glue, and synthetic fibres and fabrics. In households, diluted acetic acid is often used in descaling agents. In the food industry, acetic acid is controlled by the E number, food additive code E260 as an acidity regulator and as a condiment. In biochemistry, the acetyl group, derived from acetic acid, is funda ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
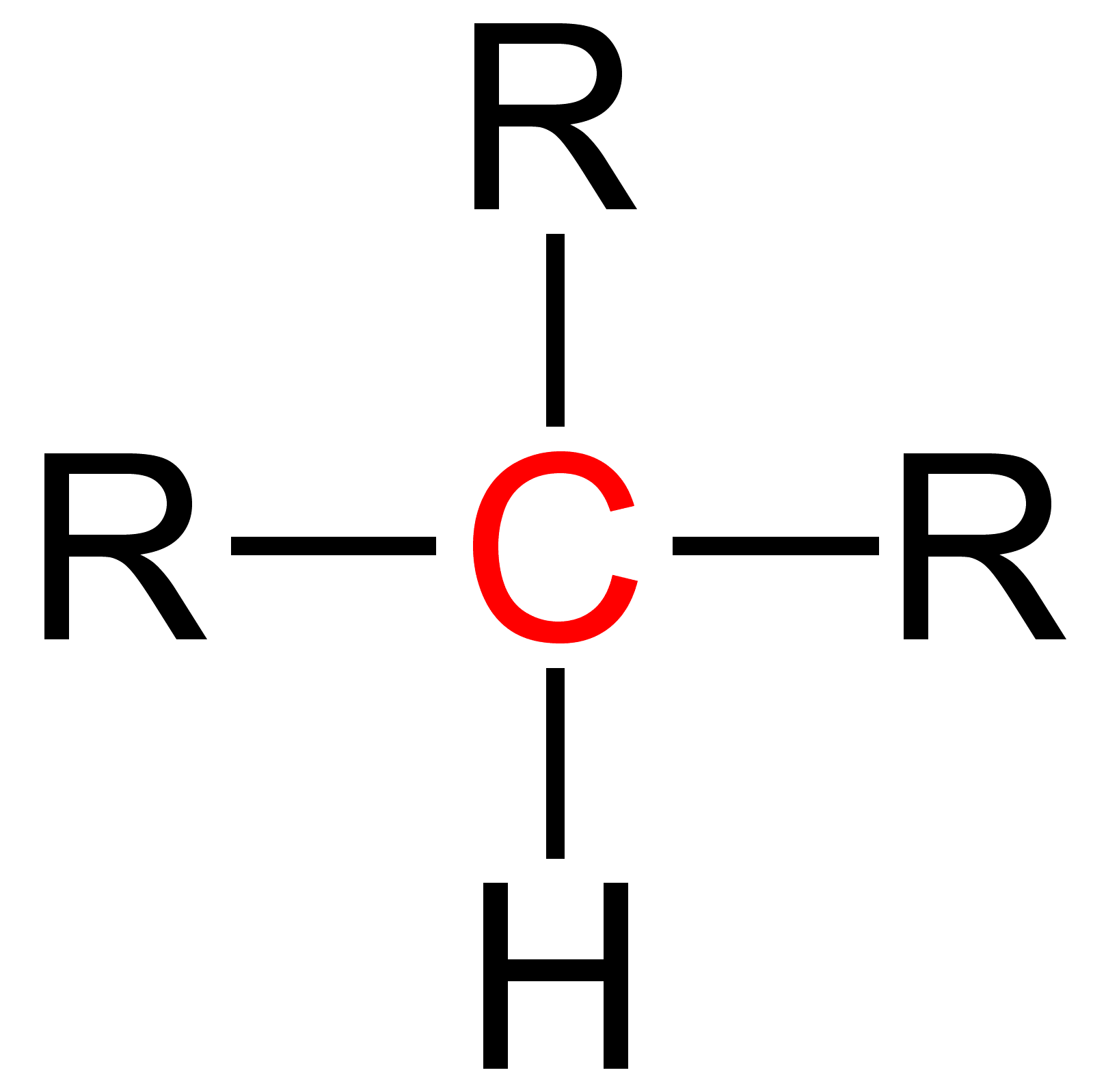
Tertiary (chemistry)
Tertiary is a term used in organic chemistry to classify various types of compounds (e. g. alcohols, alkyl halides, amines) or reactive intermediates (e. g. alkyl radicals, carbocations). See also * Primary (chemistry) Primary is a term used in organic chemistry to classify various types of compounds (e.g. alcohols, alkyl halides, amines) or reactive intermediates (e.g. alkyl radicals, carbocations). {{clear See also * Secondary (chemistry) * Tertiary (che ... * Secondary (chemistry) * Quaternary (chemistry) References {{Navbox stereochemistry Chemical nomenclature ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
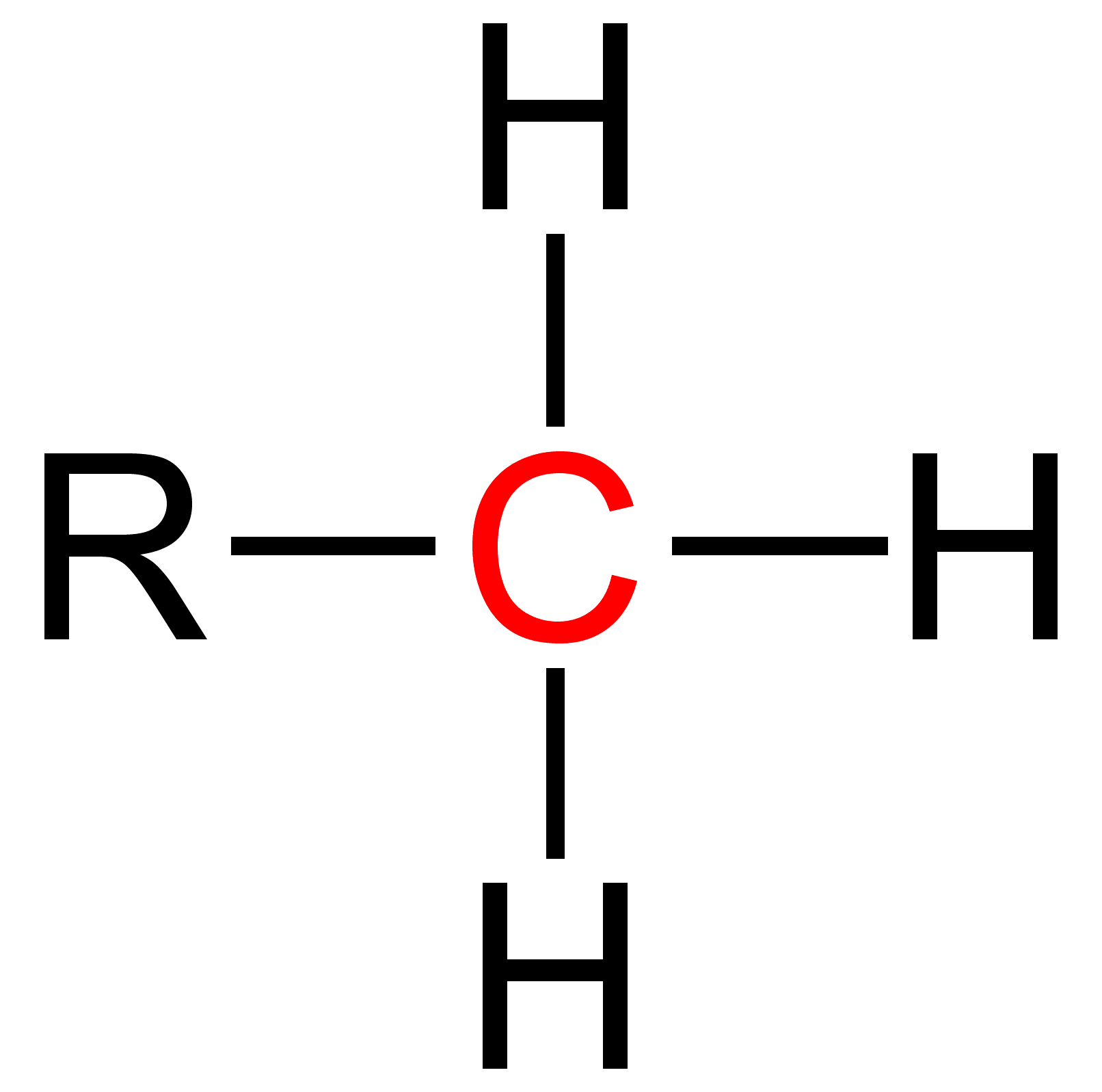
Secondary (chemistry)
Secondary is a term used in organic chemistry to classify various types of compounds (e. g. alcohols, alkyl halides, amines) or reactive intermediates (e. g. alkyl radicals, carbocations). An atom is considered secondary if it has two 'R' Groups attached to it. An 'R' group is a carbon containing group such as a methyl (CH3). A secondary compound is most often classified on an alpha carbon (middle carbon) or a nitrogen. The word secondary comes from the root word 'second' which means two. This nomenclature can be used in many cases and further used to explain relative reactivity. The reactivity of molecules varies with respect to the attached atoms. Thus, a primary, secondary, tertiary and quaternary molecule of the same function group will have different reactivities. Secondary alcohols Secondary alcohols have the formula RCH(OH)R' where R and R' are organyl. Secondary amines A secondary amine has the formula RR'NH where R and R' are organyl. Secondary amides Se ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Primary (chemistry)
Primary is a term used in organic chemistry to classify various types of compounds (e.g. alcohols, alkyl halides, amines) or reactive intermediates (e.g. alkyl radicals, carbocations). {{clear See also * Secondary (chemistry) * Tertiary (chemistry) * Quaternary (chemistry) References [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzamide
Benzamide is an organic compound with the chemical formula of C7H7NO. It is the simplest amide derivative of benzoic acid. In powdered form, it appears as a white solid, while in crystalline form, it appears as colourless crystals. It is slightly soluble in water, and soluble in many organic solvents. It is a natural alkaloid Alkaloids are a broad class of natural product, naturally occurring organic compounds that contain at least one nitrogen atom. Some synthetic compounds of similar structure may also be termed alkaloids. Alkaloids are produced by a large varie ... found in the herbs of '' Berberis pruinosa''. Chemical derivatives A number of substituted benzamides are commercial drugs, including: See also * References External links Physical characteristics {{Authority control Phenyl compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |