|
Bohn–Schmidt Reaction
The Bohn–Schmidt reaction, a named reaction in chemistry, introduces a hydroxy group at an anthraquinone system. The anthraquinone must already have at least one hydroxy group. The reaction was first described in 1889 by René Bohn (1862–1922) and in 1891 by Robert Emanuel Schmidt (1864–1938), two German industrial chemists. René Bohn is one of the few industrial chemists after whom a reaction is named. In 1901, he made indanthrone from 2-aminoanthraquinone and thus laid the basis for a new group of dyes. Reaction mechanism The postulated reaction mechanism is explained below for the example of 2-hydroxyanthraquinone: The sulfuric acid protonates the keto group of the anthraquinone 1. This causes a shift of the electrons to the oxonium ion in molecule 2. This shift enables the sulfuric acid to attack the carbenium ion 3 which is formed. The sulfuric acid oxidizes the resulting hydroxyanthracenone 5, which is then Protonation, protonated and the reaction starts all ove ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Named Reaction
A name reaction (or named reaction) is a chemical reaction named after its discoverer(s) or developer(s). Among the tens of thousands of organic reactions that are known, hundreds of such reactions are typically identified by the eponym. Well-known examples include the Grignard reaction, the Sabatier reaction, the Wittig reaction, the Claisen condensation, the Friedel–Crafts acylation, and the Diels–Alder reaction. Books have been published devoted exclusively to name reactions;Alfred Hassner, C. Stumer. ''Organic syntheses based on name reactions''. Elsevier, 2002. Li, Jie Jack. ''Name Reactions: A Collection of Detailed Reaction Mechanisms''. Springer, 2003. the ''Merck Index'', a chemical encyclopedia, also includes an appendix on name reactions. As organic chemistry developed during the 20th century, chemists started associating synthetically useful reactions with the names of their discoverers or developers. In many cases, the name is merely a mnemonic. Some reactions suc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxonium Ion
In chemistry, an oxonium ion is any cation containing an oxygen atom that has three chemical bond, bonds and 1+ formal charge. The simplest oxonium ion is the hydronium ion (). Alkyloxonium Hydronium is one of a series of oxonium ions with the formula R''n''H3−''n''O+. Oxygen is usually pyramidal with an sp3 orbital hybridisation, hybridization. Those with ''n'' = 1 are called primary oxonium ions, an example being protonated alcohol (e.g. methanol). In acidic media, the oxonium functional group produced by protonating an alcohol can be a leaving group in the elimination reaction, E2 elimination reaction. The product is an alkene. Extreme acidity, heat, and dehydrating conditions are usually required. Other hydrocarbon oxonium ions are formed by protonation or alkylation of alcohol (chemistry), alcohols or ethers (R−C−−R1R2). Secondary oxonium ions have the formula R2OH+, an example being protonated ethers. Tertiary oxonium ions have the formula R3O+, an example ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wolffenstein–Böters Reaction
The Wolffenstein–Böters reaction is an organic reaction converting benzene to picric acid by a mixture of aqueous nitric acid and mercury(II) nitrate. The reaction, which involves simultaneous nitration and oxidation, was first reported by the German chemists Richard Wolffenstein and Oskar Böters in 1906. According to one series of studies the mercury nitrate first takes benzene to the corresponding nitroso compound and through the diazonium salt to the phenol. The presence of nitrite is essential for the reaction; picric acid formation is prevented when urea, a trap for nitrous acid, is added to the mixture. From then on the reaction proceeds as a regular aromatic nitration.''The Oxynitration of Benzene. I. Studies Relating to the Reaction Mechanisms''Marvin Carmack, Manuel M. Baizer, G. Richard Handrick, L. W. Kissinger, and Edward H. Specht J. Am. Chem. Soc.; 1947; 69(4) pp 785 - 790; A conceptually related reaction is the Bohn–Schmidt reaction, dating to 1889, which ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of Biomolecule, biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Esterification
In chemistry, an ester is a compound derived from an acid (either organic or inorganic) in which the hydrogen atom (H) of at least one acidic hydroxyl group () of that acid is replaced by an organyl group (R). These compounds contain a distinctive functional group. Analogues derived from oxygen replaced by other chalcogens belong to the ester category as well. According to some authors, organyl derivatives of acidic hydrogen of other acids are esters as well (e.g. amides), but not according to the IUPAC. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Lactones are cyclic carboxylic esters; naturally occurring lactones are mainly 5- and 6-membered ring lactones. Lactones contribute to the aroma of fruits, butter, cheese, vegetables like celery and other foods. Esters can be formed from oxoacids (e.g. esters of acetic acid, carbonic acid, sulfu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atom Economy
Atom economy (atom efficiency/percentage) is the conversion efficiency of a chemical process in terms of all atoms involved and the desired products produced. The simplest definition was introduced by Barry Trost in 1991 and is equal to the ratio between the mass of desired product to the total mass of reactants, expressed as a percentage. The concept of atom economy (AE) and the idea of making it a primary criterion for improvement in chemistry, is a part of the green chemistry movement that was championed by Paul Anastas from the early 1990s. Atom economy is an important concept of green chemistry philosophy, and one of the most widely used metrics for measuring the "greenness" of a process or synthesis. Good atom economy means most of the atoms of the reactants are incorporated in the desired products and only small amounts of unwanted byproducts are formed, reducing the economic and environmental impact of waste disposal. Atom economy can be written as: \text = \frac \tim ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protonation
In chemistry, protonation (or hydronation) is the adding of a proton (or hydron, or hydrogen cation), usually denoted by H+, to an atom, molecule, or ion, forming a conjugate acid. (The complementary process, when a proton is removed from a Brønsted–Lowry acid, is deprotonation.) Some examples include * The protonation of water by sulfuric acid: *: H2SO4 + H2O H3O+ + * The protonation of isobutene in the formation of a carbocation: *: (CH3)2C=CH2 + HBF4 (CH3)3C+ + * The protonation of ammonia in the formation of ammonium chloride from ammonia and hydrogen chloride: *: NH3( g) + HCl( g) → NH4Cl( s) Protonation is a fundamental chemical reaction and is a step in many stoichiometric and catalytic processes. Some ions and molecules can undergo more than one protonation and are labeled polybasic, which is true of many biological macromolecules. Protonation and deprotonation (removal of a proton) occur in most acid–base reactions; they are the core of most acid–base r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
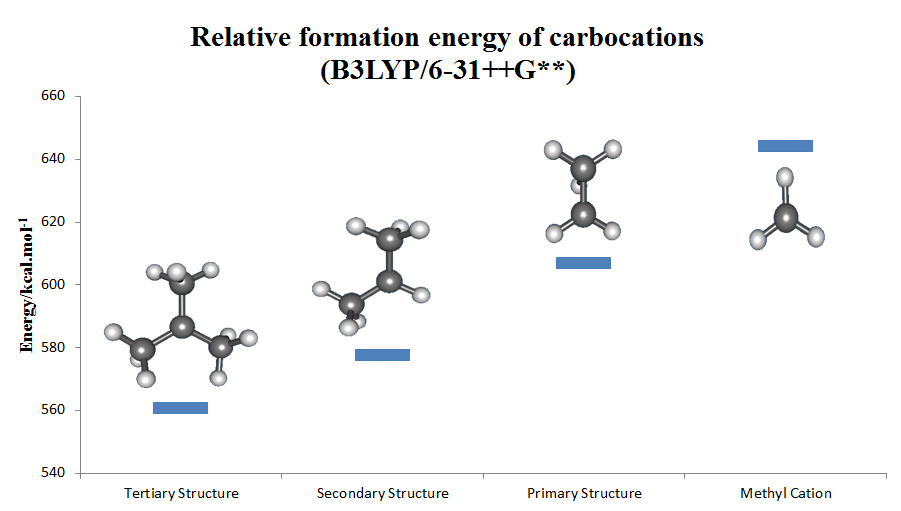
Carbenium Ion
The carbenium ion is a kind of cation, positive ion with the structure RR′R″C+, that is, a chemical species with carbon atom having three covalent bonds, and it bears a +1 formal charge. Carbenium ions are a major subset of carbocations, which is a general term for diamagnetic carbon-based cations. In parallel with carbenium ions is another subset of carbocations, the carbonium ions with the formula R5+. In carbenium ions charge is localized. They are isoelectronic with monoboranes such as B(CH3)3. Nomenclature Reactivity Carbenium ions are generally highly reactive due to having an incomplete octet rule, octet of electrons; however, certain carbenium ions, such as the tropylium cation, tropylium ion, are relatively stable due to the positive charge being delocalised between the carbon atoms.(It can even exist stably in aqueous solution.) Rearrangements Carbenium ions sometimes rearrangement reaction, rearrange readily. For example, when pentan-3-ol is heated with aq ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Keto Group
In organic chemistry, a ketone is an organic compound with the structure , where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond C=O). The simplest ketone is acetone (where R and R' are methyl), with the formula . Many ketones are of great importance in biology and industry. Examples include many sugars (ketoses), many steroids, ''e.g.'', testosterone, and the solvent acetone. Nomenclature and etymology The word ''ketone'' is derived from ''Aketon'', an old German word for ''acetone''. According to the rules of IUPAC nomenclature, ketone names are derived by changing the suffix ''-ane'' of the parent alkane to ''-anone''. Typically, the position of the carbonyl group is denoted by a number, but traditional nonsystematic names are still generally used for the most important ketones, for example acetone and benzophenone. These nonsystematic names are considered retained IUPAC names, although some introdu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxy Group
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy groups. Both the negatively charged anion , called hydroxide, and the neutral radical , known as the hydroxyl radical, consist of an unbonded hydroxy group. According to IUPAC definitions, the term ''hydroxyl'' refers to the hydroxyl radical () only, while the functional group is called a ''hydroxy group''. Properties Water, alcohols, carboxylic acids, and many other hydroxy-containing compounds can be readily deprotonated due to a large difference between the electronegativity of oxygen (3.5) and that of hydrogen (2.1). Hydroxy-containing compounds engage in intermolecular hydrogen bonding increasing the electrostatic attraction between molecules and thus to higher boiling and melting points than found for compounds that lack thi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |