Titanium dioxide on:
[Wikipedia]
[Google]
[Amazon]
Titanium dioxide, also known as titanium(IV) oxide or titania , is the
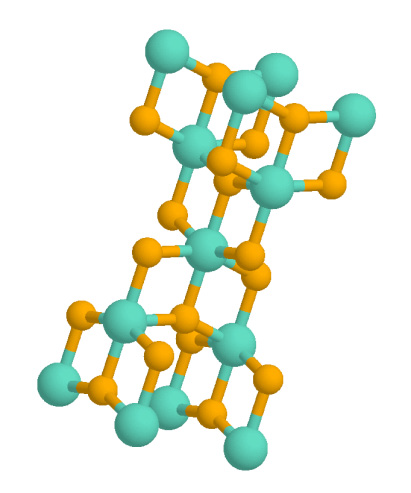 In all three of its main dioxides,
In all three of its main dioxides,

 The largest pigment processors are
The largest pigment processors are
''Japan Nanonet Bulletin'' Issue 44, 12 May 2005. In

File:Chiral TiO2 nanofibers 2.jpg, SEM (top) and TEM (bottom) images of
International Chemical Safety Card 0338
(if inhaled as a powder) * ttps://web.archive.org/web/20070112210620/http://www.threebond.co.jp/en/technical/technicalnews/pdf/tech62.pdf A description of TiO2 photocatalysis
Titanium and titanium dioxide production data (US and World)
{{Authority control Dye-sensitized solar cells E-number additives Excipients Food colorings IARC Group 2B carcinogens Inorganic pigments Sunscreening agents Titanium(IV) compounds Transition metal oxides
inorganic compound
An inorganic compound is typically a chemical compound that lacks carbon–hydrogen bondsthat is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as ''inorganic chemistry''.
Inorgan ...
derived from titanium
Titanium is a chemical element; it has symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resistant to corrosion in ...
with the chemical formula . When used as a pigment
A pigment is a powder used to add or alter color or change visual appearance. Pigments are completely or nearly solubility, insoluble and reactivity (chemistry), chemically unreactive in water or another medium; in contrast, dyes are colored sub ...
, it is called titanium white, Pigment White 6 (PW6), or CI 77891. It is a white solid that is insoluble in water, although mineral forms can appear black. As a pigment, it has a wide range of applications, including paint
Paint is a material or mixture that, when applied to a solid material and allowed to dry, adds a film-like layer. As art, this is used to create an image or images known as a painting. Paint can be made in many colors and types. Most paints are ...
, sunscreen
Sunscreen, also known as sunblock, sun lotion or sun cream, is a photoprotection, photoprotective topical product for the Human skin, skin that helps protect against sunburn and prevent skin cancer. Sunscreens come as lotions, sprays, gels, fo ...
, and food coloring
Food coloring, color additive or colorant is any dye, pigment, or substance that imparts color when it is added to food or beverages. Colorants can be supplied as liquids, powders, gels, or pastes. Food coloring is commonly used in commercia ...
. When used as a food coloring, it has E number
E numbers, short for Europe numbers, are codes for substances used as food additives, including those found naturally in many foods, such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly ...
E171. World production in 2014 exceeded 9 million tonnes. It has been estimated that titanium dioxide is used in two-thirds of all pigments, and pigments based on the oxide have been valued at a price of $13.2 billion.
Structure
 In all three of its main dioxides,
In all three of its main dioxides, titanium
Titanium is a chemical element; it has symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resistant to corrosion in ...
exhibits octahedral geometry, being bonded to six oxide anions. The oxides in turn are bonded to three Ti centers. The overall crystal structures of rutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at vis ...
and anatase
Anatase is a metastable mineral form of titanium dioxide (TiO2) with a Tetragonal crystal system, tetragonal crystal structure. Although colorless or white when pure, anatase in nature is usually a black solid due to impurities. Three other Pol ...
are tetragonal in symmetry whereas brookite
Brookite is the Orthorhombic crystal system, orthorhombic variant of titanium dioxide (TiO2), which occurs in four known natural Polymorphism (materials science), polymorphic forms (minerals with the same composition but different structure). The ...
is orthorhombic. The oxygen substructures are all slight distortions of close packing
In geometry, close-packing of equal spheres is a dense arrangement of congruent spheres in an infinite, regular arrangement (or lattice). Carl Friedrich Gauss
Johann Carl Friedrich Gauss (; ; ; 30 April 177723 February 1855) was a German m ...
: in rutile, the oxide anions are arranged in distorted hexagonal close-packing, whereas they are close to cubic close-packing in anatase and to "double hexagonal close-packing" for brookite. The rutile structure is widespread for other metal dioxides and difluorides, e.g. RuO2 and ZnF2.
Molten titanium dioxide has a local structure in which each Ti is coordinated to, on average, about 5 oxygen atoms. This is distinct from the crystalline forms in which Ti coordinates to 6 oxygen atoms.
Synthetic and geologic occurrence
Synthetic TiO2 is mainly produced from the mineralilmenite
Ilmenite is a titanium-iron oxide mineral with the idealized formula . It is a weakly magnetic black or steel-gray solid. Ilmenite is the most important ore of titanium and the main source of titanium dioxide, which is used in paints, printi ...
. Rutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at vis ...
, and anatase
Anatase is a metastable mineral form of titanium dioxide (TiO2) with a Tetragonal crystal system, tetragonal crystal structure. Although colorless or white when pure, anatase in nature is usually a black solid due to impurities. Three other Pol ...
, naturally occurring TiO2, occur widely also, e.g. rutile as a 'heavy mineral' in beach sand. Leucoxene, fine-grained anatase formed by natural alteration of ilmenite, is yet another ore. Star sapphires and rubies get their asterism from oriented inclusions of rutile needles.
Mineralogy and uncommon polymorphs
Titanium dioxide occurs in nature as the mineralsrutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at vis ...
and anatase
Anatase is a metastable mineral form of titanium dioxide (TiO2) with a Tetragonal crystal system, tetragonal crystal structure. Although colorless or white when pure, anatase in nature is usually a black solid due to impurities. Three other Pol ...
. Additionally two high-pressure forms are known minerals: a monoclinic
In crystallography, the monoclinic crystal system is one of the seven crystal systems. A crystal system is described by three Vector (geometric), vectors. In the monoclinic system, the crystal is described by vectors of unequal lengths, as in t ...
baddeleyite
Baddeleyite is a rare zirconium oxide mineral (ZrO2 or zirconia), occurring in a variety of monoclinic prismatic crystal forms. It is transparent to translucent, has high Index of refraction, indices of refraction, and ranges from colorless to yel ...
-like form known as akaogiite, and the other has a slight monoclinic distortion of the orthorhombic
In crystallography, the orthorhombic crystal system is one of the 7 crystal systems. Orthorhombic Lattice (group), lattices result from stretching a cubic crystal system, cubic lattice along two of its orthogonal pairs by two different factors, res ...
α-PbO2 structure and is known as riesite. Both of which can be found at the Ries crater in Bavaria
Bavaria, officially the Free State of Bavaria, is a States of Germany, state in the southeast of Germany. With an area of , it is the list of German states by area, largest German state by land area, comprising approximately 1/5 of the total l ...
. It is mainly sourced from ilmenite
Ilmenite is a titanium-iron oxide mineral with the idealized formula . It is a weakly magnetic black or steel-gray solid. Ilmenite is the most important ore of titanium and the main source of titanium dioxide, which is used in paints, printi ...
, which is the most widespread titanium dioxide-bearing ore around the world. Rutile is the next most abundant and contains around 98% titanium dioxide in the ore. The metastable anatase and brookite phases convert irreversibly to the equilibrium rutile phase upon heating above temperatures in the range .
Titanium dioxide has twelve known polymorphs – in addition to rutile, anatase, brookite, akaogiite and riesite, three metastable phases can be produced synthetically (monoclinic
In crystallography, the monoclinic crystal system is one of the seven crystal systems. A crystal system is described by three Vector (geometric), vectors. In the monoclinic system, the crystal is described by vectors of unequal lengths, as in t ...
, tetragonal
In crystallography, the tetragonal crystal system is one of the 7 crystal systems. Tetragonal crystal lattices result from stretching a cubic lattice along one of its lattice vectors, so that the Cube (geometry), cube becomes a rectangular Pri ...
, and orthorhombic ramsdellite-like), and four high-pressure forms (α-PbO2-like, cotunnite-like, orthorhombic OI, and cubic phases) also exist:
The cotunnite-type phase was claimed to be the hardest known oxide with the Vickers hardness
The Vickers hardness test was developed in 1921 by Robert L. Smith and George E. Sandland at Vickers Ltd as an alternative to the Brinell method to measure the hardness of materials. The Vickers test is often easier to use than other hardness ...
of 38 GPa and the bulk modulus
The bulk modulus (K or B or k) of a substance is a measure of the resistance of a substance to bulk compression. It is defined as the ratio of the infinitesimal pressure increase to the resulting ''relative'' decrease of the volume.
Other mo ...
of 431 GPa (i.e. close to diamond's value of 446 GPa) at atmospheric pressure. However, later studies came to different conclusions with much lower values for both the hardness (7–20 GPa, which makes it softer than common oxides like corundum Al2O3 and rutile TiO2) and bulk modulus (~300 GPa).
Titanium dioxide (B) is found as a mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. (2011): Mi ...
in magmatic rocks and hydrothermal veins, as well as weathering rims on perovskite
Perovskite (pronunciation: ) is a calcium titanium oxide mineral composed of calcium titanate (chemical formula ). Its name is also applied to the class of compounds which have the same type of crystal structure as , known as the perovskite (stru ...
. TiO2 also forms lamellae
Lamella (: lamellae) means a small plate or flake in Latin, and in English may refer to:
Biology
* Lamella (mycology), a papery rib beneath a mushroom cap
* Lamella (botany)
* Lamella (surface anatomy), a plate-like structure in an animal
* Lame ...
in other minerals.
Production

Chemours
The Chemours Company (, ) is an American chemical industry, chemical company that was founded in July 2015 as a Corporate spin-off, spin-off from DuPont. It has its corporate headquarters in Wilmington, Delaware, United States. Chemours is the m ...
, Venator, , and Tronox
Tronox Limited is an American worldwide chemical company involved in the titanium products industry with approximately 6,500 employees. Following its acquisition of the mineral sands business formerly belonging to South Africa's Exxaro Resources, ...
. Major paint and coating company end users for pigment grade titanium dioxide include Akzo Nobel, PPG Industries
PPG Industries, Inc. is an American Fortune 500, ''Fortune'' 500 company and global supplier of paints, coatings, and specialty materials. With headquarters in Pittsburgh, Pennsylvania, PPG operates in more than 70 countries around the globe. By ...
, Sherwin Williams, BASF
BASF SE (), an initialism of its original name , is a European Multinational corporation, multinational company and the List of largest chemical producers, largest chemical producer in the world. Its headquarters are located in Ludwigshafen, Ge ...
, Kansai Paints and Valspar
The Valspar Corporation is an American manufacturer of paint and coatings based in Minneapolis, Minnesota. With over 11,000 employees in 26 countries and a company history that spans two centuries, it was the sixth largest paint and coating corpo ...
.Hayes (2011), p. 3 Global pigment demand for 2010 was 5.3 Mt with annual growth expected to be about 3–4%.
The production method depends on the feedstock. In addition to ores, other feedstocks include upgraded slag
The general term slag may be a by-product or co-product of smelting (pyrometallurgical) ores and recycled metals depending on the type of material being produced. Slag is mainly a mixture of metal oxides and silicon dioxide. Broadly, it can be c ...
. Both the chloride process and the sulfate process (both described below) produce titanium dioxide pigment in the rutile crystal form, but the sulfate process can be adjusted to produce the anatase
Anatase is a metastable mineral form of titanium dioxide (TiO2) with a Tetragonal crystal system, tetragonal crystal structure. Although colorless or white when pure, anatase in nature is usually a black solid due to impurities. Three other Pol ...
form. Anatase, being softer, is used in fiber and paper applications. The sulfate process is run as a batch process; the chloride process is run as a continuous process.
Chloride process
In chloride process, the ore is treated with chlorine and carbon to givetitanium tetrachloride
Titanium tetrachloride is the inorganic compound with the formula . It is an important intermediate in the production of titanium metal and the pigment titanium dioxide. is a volatile liquid. Upon contact with humid air, it forms thick clouds o ...
, a volatile liquid that is further purified by distillation. The TiCl4 is treated with oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
to regenerate chlorine and produce the titanium dioxide.
Sulfate process
In the sulfate process, ilmenite is treated withsulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
to extract iron(II) sulfate pentahydrate. This process requires concentrated ilmenite (45–60% TiO2) or pretreated feedstocks as a suitable source of titanium. The resulting synthetic rutile is further processed according to the specifications of the end user, i.e. pigment grade or otherwise.
Examples of plants using the sulfate process are the Sorel-Tracy
Sorel-Tracy (; ) is a city in southwestern Quebec, Canada and the geographical end point of the Champlain Valley. It is located at the confluence of the Richelieu River and the St. Lawrence River, on the western edge of Lac Saint-Pierre, down ...
plant of QIT-Fer et Titane and the Eramet Titanium & Iron smelter in Tyssedal Norway.
Becher process
The Becher process is another method for the production of synthetic rutile from ilmenite. It first oxidizes the ilmenite as a means to separate the iron component.Specialized methods
For specialty applications, TiO2 films are prepared by various specialized chemistries. Sol-gel routes involve the hydrolysis of titaniumalkoxide
In chemistry, an alkoxide is the conjugate base of an alcohol and therefore consists of an organic group bonded to a negatively charged oxygen atom. They are written as , where R is the organyl substituent. Alkoxides are strong bases and, whe ...
s such as titanium ethoxide:
: Ti(OEt)4 + 2 H2O → TiO2 + 4 EtOH
A related approach that also relies on molecular precursors involves chemical vapor deposition
Chemical vapor deposition (CVD) is a vacuum deposition method used to produce high-quality, and high-performance, solid materials. The process is often used in the semiconductor industry to produce thin films.
In typical CVD, the wafer (electro ...
. In this method, the alkoxide is volatilized and then decomposed on contact with a hot surface:
: Ti(OEt)4 → TiO2 + 2 Et2O
Applications
Pigment
First mass-produced in 1916, titanium dioxide is the most widely used white pigment because of its brightness and very highrefractive index
In optics, the refractive index (or refraction index) of an optical medium is the ratio of the apparent speed of light in the air or vacuum to the speed in the medium. The refractive index determines how much the path of light is bent, or refrac ...
, in which it is surpassed only by a few other materials (see ''list of indices of refraction
A list is a set of discrete items of information collected and set forth in some format for utility, entertainment, or other purposes. A list may be memorialized in any number of ways, including existing only in the mind of the list-maker, but ...
''). Titanium dioxide crystal size is ideally around 220 nm (measured by electron microscope) to optimize the maximum reflection of visible light. However, abnormal grain growth is often observed in titanium dioxide, particularly in its rutile phase.
The occurrence of abnormal grain growth brings about a deviation of a small number of crystallites from the mean crystal size and modifies the physical behaviour of TiO2. The optical properties of the finished pigment are highly sensitive to purity. As little as a few parts per million (ppm) of certain metals (Cr, V, Cu, Fe, Nb) can disturb the crystal lattice so much that the effect can be detected in quality control. Approximately 4.6 million tons of pigmentary TiO2 are used annually worldwide, and this number is expected to increase as use continues to rise.
TiO2 is also an effective opacifier in powder form, where it is employed as a pigment to provide whiteness and opacity to products such as paints, coatings, plastics, papers, inks, foods, supplements, medicines (i.e. pills and tablets), and most toothpastes; in 2019 it was present in two-thirds of toothpastes on the French market. In paint, it is often referred to offhandedly as "brilliant white", "the perfect white", "the whitest white", or other similar terms. Opacity is improved by optimal sizing of the titanium dioxide particles.
Additive for food
Often used as color in food, it is commonly found in ice creams, chocolates, all types of candy, creamers, desserts, marshmallows, chewing gum, pastries, spreads, dressings, cakes, some cheeses, and many other foods. It is permitted in many countries, but was banned for use in food by the European Union in 2022. While permitted in the United States,Mars
Mars is the fourth planet from the Sun. It is also known as the "Red Planet", because of its orange-red appearance. Mars is a desert-like rocky planet with a tenuous carbon dioxide () atmosphere. At the average surface level the atmosph ...
removed it from their Skittles confectionery in 2025, although a class-action lawsuit against the use of titanium dioxide in Skittles had been dismissed in 2022.
Thin films
When deposited as athin film
A thin film is a layer of materials ranging from fractions of a nanometer ( monolayer) to several micrometers in thickness. The controlled synthesis of materials as thin films (a process referred to as deposition) is a fundamental step in many ...
, its refractive index and colour make it an excellent reflective optical coating for dielectric mirror
A dielectric mirror, also known as a Bragg mirror, is a type of mirror composed of multiple thin film, thin layers of dielectric material, typically deposited on a substrate of glass or some other optical material. By careful choice of the type a ...
s; it is also used in generating decorative thin films such as found in "mystic fire topaz".
Some grades of modified titanium based pigments as used in sparkly paints, plastics, finishes and cosmetics – these are man-made pigments whose particles have two or more layers of various oxides – often titanium dioxide, iron oxide
An iron oxide is a chemical compound composed of iron and oxygen. Several iron oxides are recognized. Often they are non-stoichiometric. Ferric oxyhydroxides are a related class of compounds, perhaps the best known of which is rust.
Iron ...
or alumina
Aluminium oxide (or aluminium(III) oxide) is a chemical compound of aluminium and oxygen with the chemical formula . It is the most commonly occurring of several aluminium oxides, and specifically identified as aluminium oxide. It is commonly ...
– in order to have glittering, iridescent
Iridescence (also known as goniochromism) is the phenomenon of certain surfaces that appear gradually to change colour as the angle of view or the angle of illumination changes. Iridescence is caused by wave interference of light in microstruc ...
and or pearlescent effects similar to crushed mica
Micas ( ) are a group of silicate minerals whose outstanding physical characteristic is that individual mica crystals can easily be split into fragile elastic plates. This characteristic is described as ''perfect basal cleavage''. Mica is co ...
or guanine
Guanine () (symbol G or Gua) is one of the four main nucleotide bases found in the nucleic acids DNA and RNA, the others being adenine, cytosine, and thymine ( uracil in RNA). In DNA, guanine is paired with cytosine. The guanine nucleoside ...
-based products. In addition to these effects a limited colour change is possible in certain formulations depending on how and at which angle the finished product is illuminated and the thickness of the oxide layer in the pigment particle; one or more colours appear by reflection while the other tones appear due to interference of the transparent titanium dioxide layers. In some products, the layer of titanium dioxide is grown in conjunction with iron oxide by calcination of titanium salts (sulfates, chlorates) around 800 °C One example of a pearlescent pigment is Iriodin, based on mica coated with titanium dioxide or iron (III) oxide.
The iridescent effect in these titanium oxide particles is unlike the opaque effect obtained with usual ground titanium oxide pigment obtained by mining, in which case only a certain diameter of the particle is considered and the effect is due only to scattering.
Sunscreen and UV blocking pigments
In cosmetic and skin care products, titanium dioxide is used as a pigment, sunscreen and a thickener. As a sunscreen, ultrafine TiO2 is used, which is notable in that combined with ultrafine zinc oxide, it is considered to be an effective sunscreen that lowers the incidence of sun burns and minimizes the prematurephotoaging
Photoaging or photoageing (also known as "dermatoheliosis") is a term used for the characteristic changes to skin induced by chronic Ultraviolet, UVA and UVB exposure.
Effects of UV light
Molecular and genetic changes
UVB rays are a primary ...
, photocarcinogenesis and immunosuppression
Immunosuppression is a reduction of the activation or efficacy of the immune system. Some portions of the immune system itself have immunosuppressive effects on other parts of the immune system, and immunosuppression may occur as an adverse react ...
associated with long term excess sun exposure. Sometimes these UV blockers are combined with iron oxide pigments in sunscreen to increase visible light protection.
Titanium dioxide and zinc oxide are generally considered to be less harmful to coral reef
A coral reef is an underwater ecosystem characterized by reef-building corals. Reefs are formed of colonies of coral polyps held together by calcium carbonate. Most coral reefs are built from stony corals, whose polyps cluster in group ...
s than sunscreens that include chemicals such as oxybenzone
Oxybenzone or benzophenone-3 or BP-3 (trade names Milestab 9, Eusolex 4360, Escalol 567, KAHSCREEN BZ-3) is an organic compound belonging to the class of aromatic ketones known as benzophenones. It takes the form of pale-yellow crystals that are ...
, octocrylene and octinoxate
Octyl methoxycinnamate or ethylhexyl methoxycinnamate (INCI) or octinoxate (United States Adopted Name, USAN), trade names Eusolex 2292 and Uvinul MC80, is an organic compound that is an ingredient in some sunscreens and lip balms. It is an este ...
.
Nanosized titanium dioxide is found in the majority of physical sunscreens because of its strong UV light absorbing capabilities and its resistance to discolouration under ultraviolet
Ultraviolet radiation, also known as simply UV, is electromagnetic radiation of wavelengths of 10–400 nanometers, shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight and constitutes about 10% of ...
light. This advantage enhances its stability and ability to protect the skin from ultraviolet light. Nano-scaled (particle size of 20–40 nm) titanium dioxide particles are primarily used in sunscreen lotion because they scatter visible light much less than titanium dioxide pigments, and can give UV protection. Sunscreens designed for infants or people with sensitive skin are often based on titanium dioxide and/or zinc oxide
Zinc oxide is an inorganic compound with the Chemical formula, formula . It is a white powder which is insoluble in water. ZnO is used as an additive in numerous materials and products including cosmetics, Zinc metabolism, food supplements, rubbe ...
, as these mineral UV blockers are believed to cause less skin irritation than other UV absorbing chemicals. Nano-TiO2, which blocks both UV-A and UV-B radiation, is used in sunscreens and other cosmetic products.
The EU Scientific Committee on Consumer Safety considered nano sized titanium dioxide to be safe for skin applications, in concentrations of up to 25 percent based on animal testing. The risk assessment of different titanium dioxide nanomaterials in sunscreen is currently evolving since nano-sized TiO2 is different from the well-known micronized form. The rutile form is generally used in cosmetic and sunscreen products due to it not possessing any observed ability to damage the skin under normal conditions and having a higher UV absorption. In 2016 Scientific Committee on Consumer Safety (SCCS) tests concluded that the use of nano titanium dioxide (95–100% rutile, ≦5% anatase) as a UV filter can be considered to not pose any risk of adverse effects in humans post-application on healthy skin, except in the case the application method would lead to substantial risk of inhalation (ie; powder or spray formulations). This safety opinion applied to nano TiO2 in concentrations of up to 25%.
Initial studies indicated that nano-TiO2 particles could penetrate the skin, causing concern over its use. These studies were later refuted, when it was discovered that the testing methodology couldn't differentiate between penetrated particles and particles simply trapped in hair follicles and that having a diseased or physically damaged dermis could be the true cause of insufficient barrier protection.
SCCS research found that when nanoparticles had certain photostable coatings (e.g., alumina
Aluminium oxide (or aluminium(III) oxide) is a chemical compound of aluminium and oxygen with the chemical formula . It is the most commonly occurring of several aluminium oxides, and specifically identified as aluminium oxide. It is commonly ...
, silica
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , commonly found in nature as quartz. In many parts of the world, silica is the major constituent of sand. Silica is one of the most complex and abundant f ...
, cetyl phosphate, triethoxycaprylylsilane, manganese dioxide
Manganese dioxide is the inorganic compound with the formula . This blackish or brown solid occurs naturally as the mineral pyrolusite, which is the main ore of manganese and a component of manganese nodules. The principal use for is for dry-cel ...
), the photocatalytic activity was attenuated and no notable skin penetration was observed; the sunscreen in this research was applied at amounts of 10 mg/cm2 for exposure periods of 24 hours. Coating TiO2 with alumina, silica, zircon
Zircon () is a mineral belonging to the group of nesosilicates and is a source of the metal zirconium. Its chemical name is zirconium(IV) silicate, and its corresponding chemical formula is Zr SiO4. An empirical formula showing some of th ...
or various polymer
A polymer () is a chemical substance, substance or material that consists of very large molecules, or macromolecules, that are constituted by many repeat unit, repeating subunits derived from one or more species of monomers. Due to their br ...
s can minimize avobenzone
Avobenzone (trade names Parsol 1789, Milestab 1789, Eusolex 9020, Escalol 517, Neo Heliopan 357 and others, International Nomenclature of Cosmetic Ingredients, INCI Butyl Methoxydibenzoylmethane) is an organic molecule and an oil-soluble ingredie ...
degradation and enhance UV absorption by adding an additional light diffraction mechanism.
is used extensively in plastics and other applications as a white pigment or an opacifier and for its UV resistant properties where the powder disperses light – unlike organic UV absorbers – and reduces UV damage, due mostly to the particle's high refractive index.
Other uses of titanium dioxide
Inceramic glaze
Ceramic glaze, or simply glaze, is a glassy coating on ceramics. It is used for decoration, to ensure the item is impermeable to liquids and to minimize the adherence of pollutants.
Glazing renders earthenware impermeable to water, sealing th ...
s, titanium dioxide acts as an opacifier and seeds crystal
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macros ...
formation.
It is used as a tattoo
A tattoo is a form of body modification made by inserting tattoo ink, dyes, or pigments, either indelible or temporary, into the dermis layer of the skin to form a design. Tattoo artists create these designs using several tattooing processes ...
pigment and in styptic pencils. Titanium dioxide is produced in varying particle sizes which are both oil and water dispersible, and in certain grades for the cosmetic industry. It is also a common ingredient in toothpaste.
The exterior of the Saturn V
The Saturn V is a retired American super heavy-lift launch vehicle developed by NASA under the Apollo program for human exploration of the Moon. The rocket was human-rated, had multistage rocket, three stages, and was powered by liquid-propel ...
rocket was painted with titanium dioxide; this later allowed astronomers to determine that J002E3 was likely the S-IVB
The S-IVB (pronounced "S-four-B") was the third stage on the Saturn V and second stage on the Saturn IB launch vehicles. Built by the Douglas Aircraft Company, it had one J-2 rocket engine. For lunar missions it was fired twice: first for Earth ...
stage from Apollo 12 and not an asteroid
An asteroid is a minor planet—an object larger than a meteoroid that is neither a planet nor an identified comet—that orbits within the Solar System#Inner Solar System, inner Solar System or is co-orbital with Jupiter (Trojan asteroids). As ...
.
Titanium dioxide is an n-type semiconductor
N-type, N type or Type N may refer to:
* N-type semiconductor is a key material in the manufacture of transistors and integrated circuits
* An N-type connector is a threaded RF connector used to join coaxial cables
* The MG N-type Magnette was p ...
and is used in dye-sensitized solar cells. It is also used in other electronics components such as electrode
An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit (e.g. a semiconductor, an electrolyte, a vacuum or a gas). In electrochemical cells, electrodes are essential parts that can consist of a varie ...
s in batteries.
Research
Patenting activities
Between 2002 and 2022, there were 459 patent families that describe the production of titanium dioxide fromilmenite
Ilmenite is a titanium-iron oxide mineral with the idealized formula . It is a weakly magnetic black or steel-gray solid. Ilmenite is the most important ore of titanium and the main source of titanium dioxide, which is used in paints, printi ...
. The majority of these patents describe pre-treatment processes, such as using smelting and magnetic separation to increase titanium concentration in low-grade ores, leading to titanium concentrates or slags. Other patents describe processes to obtain titanium dioxide, either by a direct hydrometallurgical process or through the main industrial production processes, the sulfate process and the chloride process. The sulfate process represents 40% of the world’s titanium dioxide production and is protected in 23% of patent families. The chloride process is only mentioned in 8% of patent families, although it provides 60% of the worldwide industrial production of titanium dioxide.
Key contributors to patents on the production of titanium dioxide are companies from China, Australia and the United States, reflecting the major contribution of these countries to industrial production. Chinese companies Pangang and Lomon Billions Groups hold major patent portfolios.
Photocatalyst
Nanosized titanium dioxide, particularly in the anatase form, exhibits photocatalytic activity under ultraviolet (UV) irradiation. This photoactivity is reportedly most pronounced at the planes of anatase, although the planes are thermodynamically more stable and thus more prominent in most synthesised and natural anatase, as evident by the often observed tetragonal dipyramidal growth habit. Interfaces between rutile and anatase are further considered to improve photocatalytic activity by facilitating charge carrier separation and as a result, biphasic titanium dioxide is often considered to possess enhanced functionality as a photocatalyst. It has been reported that titanium dioxide, when doped with nitrogen ions or doped with metal oxide like tungsten trioxide, exhibits excitation also under visible light. The strong oxidative potential of the positive holes oxidizes water to createhydroxyl radical
The hydroxyl radical, •HO, is the neutral form of the hydroxide ion (HO–). Hydroxyl radicals are highly reactive and consequently short-lived; however, they form an important part of radical chemistry. Most notably hydroxyl radicals are pr ...
s. It can also oxidize oxygen or organic materials directly. Hence, in addition to its use as a pigment, titanium dioxide can be added to paints, cements, windows, tiles, or other products for its sterilizing, deodorizing, and anti-fouling properties, and is used as a hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water ...
catalyst
Catalysis () is the increase in rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quick ...
. It is also used in dye-sensitized solar cells
A dye-sensitized solar cell (DSSC, DSC, DYSC or Grätzel cell) is a low-cost solar cell belonging to the group of thin film solar cells. It is based on a semiconductor formed between a photo-sensitized anode and an electrolyte, a ''Photoelectro ...
, which are a type of chemical solar cell (also known as a Graetzel cell).
The photocatalytic properties of nanosized titanium dioxide were discovered by Akira Fujishima in 1967 and published in 1972. The process on the surface of the titanium dioxide was called the ."Discovery and applications of photocatalysis – Creating a comfortable future by making use of light energy"''Japan Nanonet Bulletin'' Issue 44, 12 May 2005. In
thin film
A thin film is a layer of materials ranging from fractions of a nanometer ( monolayer) to several micrometers in thickness. The controlled synthesis of materials as thin films (a process referred to as deposition) is a fundamental step in many ...
and nanoparticle
A nanoparticle or ultrafine particle is a particle of matter 1 to 100 nanometres (nm) in diameter. The term is sometimes used for larger particles, up to 500 nm, or fibers and tubes that are less than 100 nm in only two directions. At ...
form, titanium dioxide has the potential for use in energy production: As a photocatalyst, it can break water into hydrogen and oxygen. With the hydrogen collected, it could be used as a fuel. The efficiency of this process can be greatly improved by doping the oxide with carbon. Further efficiency and durability has been obtained by introducing disorder to the lattice structure of the surface layer of titanium dioxide nanocrystals, permitting infrared absorption. Visible-light-active nanosized anatase and rutile has been developed for photocatalytic applications.
In 1995 Fujishima and his group discovered the superhydrophilicity phenomenon for titanium dioxide coated glass exposed to sun light. This resulted in the development of self-cleaning glass and anti-fogging coatings.
Nanosized TiO2 incorporated into outdoor building materials, such as paving stones in noxer blocks or paints, could reduce concentrations of airborne pollutants such as volatile organic compound
Volatile organic compounds (VOCs) are organic compounds that have a high vapor pressure at room temperature. They are common and exist in a variety of settings and products, not limited to Indoor mold, house mold, Upholstery, upholstered furnitur ...
s and nitrogen oxide
Nitrogen oxide may refer to a binary compound of oxygen and nitrogen, or a mixture of such compounds:
Charge-neutral
*Nitric oxide (NO), nitrogen(II) oxide, or nitrogen monoxide
* Nitrogen dioxide (), nitrogen(IV) oxide
* Nitrogen trioxide (), o ...
s. A TiO2-containing cement has been produced.
Using TiO2 as a photocatalyst, attempts have been made to mineralize pollutants (to convert into CO2 and H2O) in waste water. The photocatalytic destruction of organic matter could also be exploited in coatings with antimicrobial applications.
Hydroxyl radical formation
Although nanosized anatase TiO2 does not absorb visible light, it does strongly absorbultraviolet
Ultraviolet radiation, also known as simply UV, is electromagnetic radiation of wavelengths of 10–400 nanometers, shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight and constitutes about 10% of ...
(UV) radiation (''hv''), leading to the formation of hydroxyl radicals. This occurs when photo-induced valence bond holes (h+vb) are trapped at the surface of TiO2 leading to the formation of trapped holes (h+tr) that cannot oxidize water.
: TiO2 + ''hv'' → e− + h+vb
: h+vb → h+tr
: O2 + e− → O2•−
: O2•− + O2•−+ 2 → H2O2 + O2
: O2•− + h+vb → O2
: O2•− + h+tr → O2
: + h+vb → HO•
: e− + h+tr → recombination
: Note: Wavelength (λ)= 387 nm This reaction has been found to mineralize and decompose undesirable compounds in the environment, specifically the air and in wastewater.
Nanotubes
Anatase can be converted into non-carbon nanotubes andnanowire
file:SnSe@SWCNT.jpg, upright=1.2, Crystalline 2×2-atom tin selenide nanowire grown inside a single-wall carbon nanotube (tube diameter ≈1 nm).
A nanowire is a nanostructure in the form of a wire with the diameter of the order of a nanometre ( ...
s. Hollow TiO2 nanofibers can be also prepared by coating carbon nanofibers by first applying titanium butoxide.
chiral
Chirality () is a property of asymmetry important in several branches of science. The word ''chirality'' is derived from the Greek language, Greek (''kheir''), "hand", a familiar chiral object.
An object or a system is ''chiral'' if it is dist ...
TiO2 nanofibers
File:TiO2nanotube.jpg, Titanium oxide nanotubes, SEM image
File:The army of titanium dioxide nanotubes.jpg, Nanotubes of titanium dioxide (TiO2-Nt) obtained by electrochemical synthesis. The SEM image shows an array of vertical self-ordered TiO2-Nt with closed bottom ends of tubes.
Solubility
Titanium dioxide is insoluble in water, organic solvents, and inorganic acids. It is slightly soluble inalkali
In chemistry, an alkali (; from the Arabic word , ) is a basic salt of an alkali metal or an alkaline earth metal. An alkali can also be defined as a base that dissolves in water. A solution of a soluble base has a pH greater than 7.0. The a ...
, soluble in saturated potassium acid carbonate, and can be completely dissolved in strong sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
and hydrofluoric acid
Hydrofluoric acid is a solution of hydrogen fluoride (HF) in water. Solutions of HF are colorless, acidic and highly corrosive. A common concentration is 49% (48–52%) but there are also stronger solutions (e.g. 70%) and pure HF has a boiling p ...
after boiling for a long time.
Health and safety
Widely-occurring minerals and even gemstones are composed of TiO2. All natural titanium, comprising more than 0.5% of the Earth's crust, exists as oxides.Food additive
As of 2024, titanium dioxide is considered safe by the US FDA as a color ingredient for oral human consumption as long as it is 1% or less of the total food composition. A 2021 ban by the EU EFSA has been criticized as based on errors regarding the safety of titanium dioxide (E171) particles as a food additive, and according to a 2022 review, existing evidence does not support a direct DNA damaging mechanism for titanium dioxide.Government policies
TiO2 whitener in food was banned in France from 2020, due to uncertainty about safe quantities for human consumption. In 2021, theEuropean Food Safety Authority
The European Food Safety Authority (EFSA) is the agency of the European Union (EU) that provides independent scientific advice and communicates on existing and emerging risks associated with the food chain. EFSA was established in February 2002 ...
(EFSA) ruled that as a consequence of new understandings of nanoparticle
A nanoparticle or ultrafine particle is a particle of matter 1 to 100 nanometres (nm) in diameter. The term is sometimes used for larger particles, up to 500 nm, or fibers and tubes that are less than 100 nm in only two directions. At ...
s, titanium dioxide could "no longer be considered safe as a food additive", and the EU health commissioner announced plans to ban its use across the EU, with discussions beginning in June 2021. EFSA concluded that genotoxicity
Genotoxicity is the chemical property, property of chemical agents that damage the genetic information within a cell causing mutations, which may lead to cancer. While genotoxicity is often confused with mutagenicity, all mutagens are genotoxic, bu ...
—which could lead to carcinogenic
A carcinogen () is any agent that promotes the development of cancer. Carcinogens can include synthetic chemicals, naturally occurring substances, physical agents such as ionizing and non-ionizing radiation, and Biological agent, biologic agent ...
effects—could not be ruled out, and that a "safe level for daily intake of the food additive could not be established". In 2022, the UK Food Standards Agency and Food Standards Scotland announced their disagreement with the EFSA ruling, and did not follow the EU in banning titanium dioxide as a food additive. Health Canada similarly reviewed the available evidence in 2022 and decided not to change their position on titanium dioxide as a food additive.
The European Union removed the authorization to use titanium dioxide (E 171) in foods, effective 7 February 2022, with a six months grace period.
As of May 2023, following the European Union 2022 ban, the U.S. states California
California () is a U.S. state, state in the Western United States that lies on the West Coast of the United States, Pacific Coast. It borders Oregon to the north, Nevada and Arizona to the east, and shares Mexico–United States border, an ...
and New York were considering banning the use of titanium dioxide in foods.
As of 2024, the Food and Drug Administration
The United States Food and Drug Administration (FDA or US FDA) is a List of United States federal agencies, federal agency of the United States Department of Health and Human Services, Department of Health and Human Services. The FDA is respo ...
(FDA) in the United States permits titanium dioxide as a food additive. It may be used to increase whiteness and opacity in dairy products (some cheeses, ice cream, and yogurt), candies, frostings, fillings, and many other foods. The FDA regulates the labeling of products containing titanium dioxide, allowing the product's ingredients list to identify titanium dioxide either as "color added" or "artificial colors" or "titanium dioxide;" it does not require that titanium dioxide be explicitly named. In 2023, the Consumer Healthcare Products Association, a manufacturer's trade group, defended the substance as safe at certain limits while allowing that additional studies could provide further insight, saying an immediate ban would be a "knee-jerk" reaction.
Industry response
Dunkin' Donuts
DD IP Holder LLC, doing business as Dunkin', and originally Dunkin' Donuts, is an American multinational coffee and doughnut company, as well as a quick service restaurant. It was founded by Bill Rosenberg in Quincy, Massachusetts, in 19 ...
dropped titanium dioxide from their merchandise in 2015 after public pressure.
Research as an ingestible nanomaterial
Size distribution analyses showed that batches of food-grade TiO₂, which is produced with a target particle size in the 200300nm range for optimal pigmentation qualities, include a nanoparticle-sized fraction as inevitable byproduct of the manufacturing processes.Inhalation
Titanium dioxide dust, when inhaled, has been classified by theInternational Agency for Research on Cancer
The International Agency for Research on Cancer (IARC; ) is an intergovernmental agency forming part of the World Health Organization of the United Nations.
Its role is to conduct and coordinate research into the causes of cancer. It also cance ...
(IARC) as an IARC Group 2B carcinogen, meaning it is ''possibly carcinogenic to humans''. The US National Institute for Occupational Safety and Health
The National Institute for Occupational Safety and Health (NIOSH, ) is the List of United States federal agencies, United States federal agency responsible for conducting research and making recommendations for the prevention of work-related occ ...
recommends two separate exposure limits. NIOSH recommends that fine particles be set at an exposure limit of 2.4 mg/m3, while ultrafine be set at an exposure limit of 0.3 mg/m3, as time-weighted average concentrations up to 10 hours a day for a 40-hour work week.
Although no evidence points to acute toxicity, recurring concerns have been expressed about nanophase forms of these materials. Studies of workers with high exposure to TiO2 particles indicate that even at high exposure there is no adverse effect to human health.
Environmental waste introduction
Titanium dioxide (TiO₂) is mostly introduced into the environment asnanoparticle
A nanoparticle or ultrafine particle is a particle of matter 1 to 100 nanometres (nm) in diameter. The term is sometimes used for larger particles, up to 500 nm, or fibers and tubes that are less than 100 nm in only two directions. At ...
s via wastewater treatment plants. Cosmetic pigments including titanium dioxide enter the wastewater when the product is washed off into sinks after cosmetic use. Once in the sewage treatment plants, pigments separate into sewage sludge which can then be released into the soil when injected into the soil or distributed on its surface. 99% of these nanoparticles wind up on land rather than in aquatic environments due to their retention in sewage sludge. In the environment, titanium dioxide nanoparticles have low to negligible solubility and have been shown to be stable once particle aggregates are formed in soil and water surroundings. In the process of dissolution, water-soluble ions typically dissociate from the nanoparticle into solution when thermodynamically unstable. TiO2 dissolution increases when there are higher levels of dissolved organic matter and clay in the soil. However, aggregation is promoted by pH at the isoelectric point of TiO2 (pH= 5.8) which renders it neutral and solution ion concentrations above 4.5 mM.
See also
* Delustrant * Dye-sensitized solar cell *List of inorganic pigments
The following list includes commercially or artistically important inorganic pigments of natural and synthetic origin..
Purple pigments
Aluminosilicate pigments
* Ultramarine violet (): a synthetic or naturally occurring sulfur containing silica ...
* Noxer blocks, TiO2-coated pavers that remove pollutants from the air
* Suboxide
* Surface properties of transition metal oxides
* Titanium dioxide nanoparticle
Sources
References
External links
International Chemical Safety Card 0338
(if inhaled as a powder) * ttps://web.archive.org/web/20070112210620/http://www.threebond.co.jp/en/technical/technicalnews/pdf/tech62.pdf A description of TiO2 photocatalysis
Titanium and titanium dioxide production data (US and World)
{{Authority control Dye-sensitized solar cells E-number additives Excipients Food colorings IARC Group 2B carcinogens Inorganic pigments Sunscreening agents Titanium(IV) compounds Transition metal oxides