Sulphite on:
[Wikipedia]
[Google]
[Amazon]
 Sulfites or sulphites are compounds that contain the sulfite ion (or the sulfate(IV) ion, from its correct systematic name), . The sulfite ion is the
Sulfites or sulphites are compounds that contain the sulfite ion (or the sulfate(IV) ion, from its correct systematic name), . The sulfite ion is the
REGULATION (EU) No 1169/2011 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL
/ref>
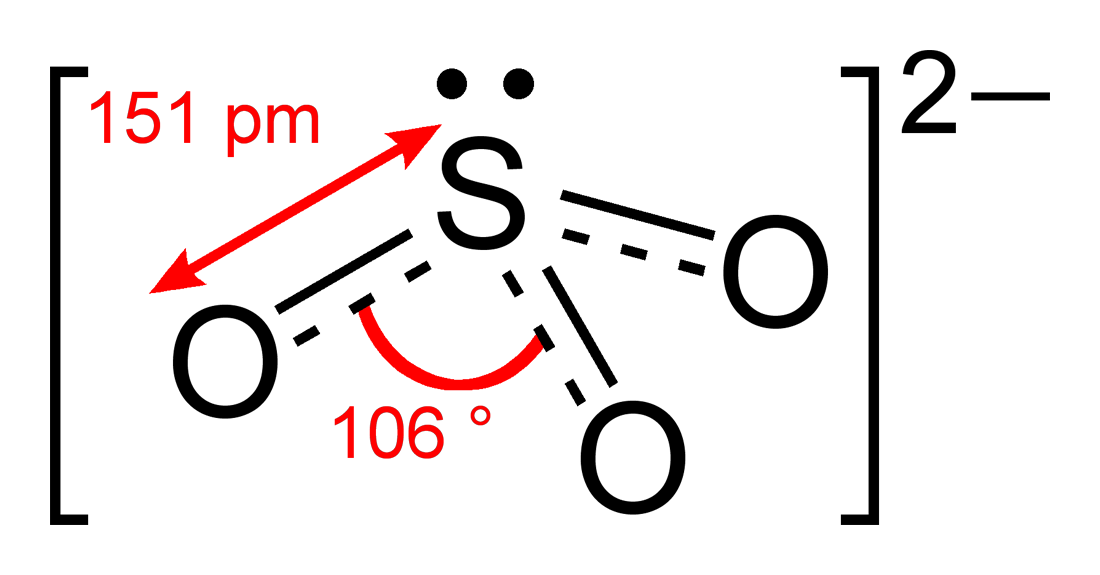
 The structure of the sulfite anion can be described with three equivalent
The structure of the sulfite anion can be described with three equivalent  Evidence from 17O NMR spectroscopic data suggests that protonation of the sulfite ion gives a mixture of isomers:
Evidence from 17O NMR spectroscopic data suggests that protonation of the sulfite ion gives a mixture of isomers:

 Sulfites or sulphites are compounds that contain the sulfite ion (or the sulfate(IV) ion, from its correct systematic name), . The sulfite ion is the
Sulfites or sulphites are compounds that contain the sulfite ion (or the sulfate(IV) ion, from its correct systematic name), . The sulfite ion is the conjugate base
A conjugate acid, within the Brønsted–Lowry acid–base theory, is a chemical compound formed when an acid donates a proton () to a base—in other words, it is a base with a hydrogen ion added to it, as in the reverse reaction it loses a ...
of bisulfite
The bisulfite ion (IUPAC-recommended nomenclature: hydrogensulfite) is the ion . Salts containing the ion are also known as "sulfite lyes". Sodium bisulfite is used interchangeably with sodium metabisulfite (Na2S2O5). Sodium metabisulfite diss ...
. Although its acid (sulfurous acid
Sulfurous acid (also sulfuric(IV) acid, sulphurous acid (UK), sulphuric(IV) acid (UK)) is the chemical compound with the formula . There is no evidence that sulfurous acid exists in solution, but the molecule has been detected in the gas phase ...
) is elusive, its salts are widely used.
Sulfites are substances that naturally occur in some foods and the human body. They are also used as regulated food additives. When in food or drink, sulfites are often lumped together with sulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic a ...
.SeREGULATION (EU) No 1169/2011 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL
/ref>
Structure

 The structure of the sulfite anion can be described with three equivalent
The structure of the sulfite anion can be described with three equivalent resonance structures
In chemistry, resonance, also called mesomerism, is a way of describing bonding in certain molecules or polyatomic ions by the combination of several contributing structures (or ''forms'', also variously known as ''resonance structures'' or '' ...
. In each resonance structure, the sulfur atom is double-bonded to one oxygen atom with a formal charge
In chemistry, a formal charge (F.C. or q), in the covalent view of chemical bonding, is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electro ...
of zero (neutral), and sulfur is singly bonded to the other two oxygen atoms, which each carry a formal charge of −1, together accounting for the −2 charge on the anion. There is also a non-bonded lone pair
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bondIUPAC '' Gold Book'' definition''lone (electron) pair''/ref> and is sometimes called an unshared pair or non-bonding pair. L ...
on the sulfur, so the structure predicted by VSEPR theory
Valence shell electron pair repulsion (VSEPR) theory ( , ), is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. It is also named the Gillespie-Nyholm the ...
is trigonal pyramidal, as in ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous ...
(NH3). In the hybrid resonance structure, the S−O bonds are equivalently of bond order
In chemistry, bond order, as introduced by Linus Pauling, is defined as the difference between the number of bonds and anti-bonds.
The bond order itself is the number of electron pairs ( covalent bonds) between two atoms. For example, in diat ...
one and one-third.
 Evidence from 17O NMR spectroscopic data suggests that protonation of the sulfite ion gives a mixture of isomers:
Evidence from 17O NMR spectroscopic data suggests that protonation of the sulfite ion gives a mixture of isomers:

Commercial uses
Sulfites are used as a foodpreservative
A preservative is a substance or a chemical that is added to products such as food products, beverages, pharmaceutical drugs, paints, biological samples, cosmetics, wood, and many other products to prevent decomposition by microbial growth or b ...
or enhancer. They may come in various forms, such as:
* Sulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic a ...
, which is not a sulfite, but a closely related chemical oxide
* Potassium bisulfite
Potassium bisulfite (or potassium hydrogen sulfite) is a chemical mixture with the approximate chemical formula KHSO3. Potassium bisulfite in fact is not a real compound, but a mixture of salts that dissolve in water to give solutions composed o ...
or potassium metabisulfite
Potassium metabisulfite, K2S2O5, also known as potassium pyrosulfite, is a white crystalline powder with a pungent odour. It is mainly used as an antioxidant or chemical sterilant. As a disulfite, it is chemically very similar to sodium meta ...
* Sodium bisulfite
Sodium bisulfite (or sodium bisulphite, sodium hydrogen sulfite) is a chemical mixture with the approximate chemical formula NaHSO3. Sodium bisulfite in fact is not a real compound, but a mixture of salts that dissolve in water to give solutio ...
, sodium metabisulfite or sodium sulfite
Sodium sulfite (sodium sulphite) is the inorganic compound with the chemical formula Na2 SO3. A white, water-soluble solid, it is used commercially as an antioxidant and preservative. A heptahydrate is also known but it is less useful because o ...
Wine
Sulfites occur naturally in allwine
Wine is an alcoholic drink typically made from Fermentation in winemaking, fermented grapes. Yeast in winemaking, Yeast consumes the sugar in the grapes and converts it to ethanol and carbon dioxide, releasing heat in the process. Different ...
s to some extent. Sulfites are commonly introduced to arrest fermentation
Fermentation is a metabolic process that produces chemical changes in organic substrates through the action of enzymes. In biochemistry, it is narrowly defined as the extraction of energy from carbohydrates in the absence of oxygen. In food p ...
at a desired time, and may also be added to wine as preservative
A preservative is a substance or a chemical that is added to products such as food products, beverages, pharmaceutical drugs, paints, biological samples, cosmetics, wood, and many other products to prevent decomposition by microbial growth or b ...
s to prevent spoilage and oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or ...
at several stages of the winemaking
Winemaking or vinification is the production of wine, starting with the selection of the fruit, its fermentation into alcohol, and the bottling of the finished liquid. The history of wine-making stretches over millennia. The science of wine and ...
. Sulfur dioxide (SO2) protects wine not only from oxidation, but also from bacteria.
Organic
Organic may refer to:
* Organic, of or relating to an organism, a living entity
* Organic, of or relating to an anatomical organ
Chemistry
* Organic matter, matter that has come from a once-living organism, is capable of decay or is the product ...
wines are not necessarily sulfite-free, but generally have lower amounts and regulations stipulate lower maximum sulfite contents for these wines. In general, white wines contain more sulfites than red wines and sweeter wines contain more sulfites than drier ones.
In the United States, wines bottled after mid-1987 must have a label stating that they contain sulfites if they contain more than 10 parts per million (ppm).
In the European Union
The European Union (EU) is a supranational union, supranational political union, political and economic union of Member state of the European Union, member states that are located primarily in Europe, Europe. The union has a total area of ...
an equivalent regulation came into force in November 2005. This includes sulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic a ...
, and the limit is on the milligrams per kilogram or per litre of sulfur dioxide equivalent. In 2012, a new regulation for organic wines came into force. In the United Kingdom, similar laws apply. Bottles of wine that contain over 10 mg/L (ppm) of "sulfites" (or sulfur dioxide) are required to bear "contains sulphites" on the label. This does not differ if sulfites are naturally occurring or added in the winemaking process.
Other foods
Sulfites are often used aspreservative
A preservative is a substance or a chemical that is added to products such as food products, beverages, pharmaceutical drugs, paints, biological samples, cosmetics, wood, and many other products to prevent decomposition by microbial growth or b ...
s in dried fruit
Dried fruit is fruit from which the majority of the original water content has been removed either naturally, through sun drying, or through the use of specialized dryers or dehydrators. Dried fruit has a long tradition of use dating back to th ...
s, preserved radish
The radish (''Raphanus raphanistrum'' subsp. ''sativus'') is an edible root vegetable of the family Brassicaceae that was domesticated in Asia prior to Roman times.
Radishes are grown and consumed throughout the world, being mostly eaten raw ...
, and dried potato
The potato is a starchy food, a tuber of the plant ''Solanum tuberosum'' and is a root vegetable native to the Americas. The plant is a perennial in the nightshade family Solanaceae.
Wild potato species can be found from the southern Uni ...
products.
Most beer
Beer is one of the oldest and the most widely consumed type of alcoholic drink in the world, and the third most popular drink overall after water and tea. It is produced by the brewing and fermentation of starches, mainly derived from ce ...
s no longer contain sulfites, although some alcoholic ciders contain them. Although shrimp
Shrimp are crustaceans (a form of shellfish) with elongated bodies and a primarily swimming mode of locomotion – most commonly Caridea and Dendrobranchiata of the decapod order, although some crustaceans outside of this order are ref ...
are sometimes treated with sulfites on fishing vessels, the chemical may not appear on the label. In 1986, the Food and Drug Administration
The United States Food and Drug Administration (FDA or US FDA) is a List of United States federal agencies, federal agency of the United States Department of Health and Human Services, Department of Health and Human Services. The FDA is respon ...
in the United States banned the addition of sulfites to all fresh fruit and vegetables that are eaten raw.
E numbers
E numbers
E numbers ("E" stands for "Europe") are codes for substances used as food additives, including those found naturally in many foods such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly f ...
for sulfites as food additives
Food additives are substances added to food to preserve flavor or enhance taste, appearance, or other sensory qualities. Some additives have been used for centuries as part of an effort to preserve food, for example vinegar (pickling), salt (sal ...
are:
Health effects
Allergic reactions to sulfites appear to be very rare in the general population, but more common in hyperallergic individuals. Sulfites are counted among the top nine food allergens, but a reaction to sulfite is not a true allergy. Some people have positive skin allergy tests to sulfites indicating true (IgE
Immunoglobulin E (IgE) is a type of antibody (or immunoglobulin (Ig) " isotype") that has been found only in mammals. IgE is synthesised by plasma cells. Monomers of IgE consist of two heavy chains (ε chain) and two light chains, with the ε c ...
-mediated) allergy. Chronic skin conditions in the hands, perineum
The perineum in humans is the space between the anus and scrotum in the male, or between the anus and the vulva in the female. The perineum is the region of the body between the pubic symphysis (pubic arch) and the coccyx (tail bone), includi ...
, and face have been reported in individuals that regularly use cosmetics or medications containing sulfites. Occupational exposure to sulfites has been reported to cause persistent skin symptoms.
It may cause breathing difficulty within minutes after eating a food containing it. Asthma
Asthma is a long-term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and easily triggered bronchospasms. Symptoms include episodes of wheezing, co ...
tics and possibly people with salicylate sensitivity (or aspirin
Aspirin, also known as acetylsalicylic acid (ASA), is a nonsteroidal anti-inflammatory drug (NSAID) used to reduce pain, fever, and/or inflammation, and as an antithrombotic. Specific inflammatory conditions which aspirin is used to treat inc ...
sensitivity) are at an elevated risk for reaction to sulfites. Anaphylaxis
Anaphylaxis is a serious, potentially fatal allergic reaction and medical emergency that is rapid in onset and requires immediate medical attention regardless of use of emergency medication on site. It typically causes more than one of the foll ...
and life-threatening reactions are rare. Other potential symptoms include sneezing
A sneeze (also known as sternutation) is a semi-autonomous, convulsive expulsion of air from the lungs through the nose and mouth, usually caused by foreign particles irritating the nasal mucosa. A sneeze expels air forcibly from the mouth and ...
, swelling of the throat
In vertebrate anatomy, the throat is the front part of the neck, internally positioned in front of the vertebrae. It contains the pharynx and larynx. An important section of it is the epiglottis, separating the esophagus from the trachea (windpip ...
, hives, and migraine.
A 2017 study has shown negative impacts of sulfites on bacteria found in the human microbiome
A microbiome () is the community of microorganisms that can usually be found living together in any given habitat. It was defined more precisely in 1988 by Whipps ''et al.'' as "a characteristic microbial community occupying a reasonably wel ...
.
Use and labeling regulations
In 1986, the U.S. Food and Drug Administration banned the use of sulfites as preservatives on foods intended to be eaten fresh (such as salad ingredients). This has contributed to the increased use of erythorbic acid and itssalts
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively c ...
as preservatives.
They also cannot be added to foods high in vitamin B1
Thiamine, also known as thiamin and vitamin B1, is a vitamin, an essential micronutrient, that cannot be made in the body. It is found in food and commercially synthesized to be a dietary supplement or medication. Phosphorylated forms of thia ...
such as meats because sulfites can destroy vitamin B1 from foods
Generally, U.S. labeling regulations do not require products to indicate the presence of sulfites in foods unless it is added specifically as a preservative; still, many companies voluntarily label sulfite-containing foods. Sulfites used in food processing (but not as a preservative) are required to be listed if they are not incidental additives (21 CFR 101.100(a)(3)), and if there are more than 10 ppm in the finished product (21 CFR 101.100(a)(4))
Sulfites that are allowed to be added in food in the US are sulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic a ...
, sodium sulfite
Sodium sulfite (sodium sulphite) is the inorganic compound with the chemical formula Na2 SO3. A white, water-soluble solid, it is used commercially as an antioxidant and preservative. A heptahydrate is also known but it is less useful because o ...
, sodium bisulfite
Sodium bisulfite (or sodium bisulphite, sodium hydrogen sulfite) is a chemical mixture with the approximate chemical formula NaHSO3. Sodium bisulfite in fact is not a real compound, but a mixture of salts that dissolve in water to give solutions ...
, potassium bisulfite
Potassium bisulfite (or potassium hydrogen sulfite) is a chemical mixture with the approximate chemical formula KHSO3. Potassium bisulfite in fact is not a real compound, but a mixture of salts that dissolve in water to give solutions composed of ...
, sodium metabisulfite, and potassium metabisulfite
Potassium metabisulfite, K2S2O5, also known as potassium pyrosulfite, is a white crystalline powder with a pungent odour. It is mainly used as an antioxidant or chemical sterilant. As a disulfite, it is chemically very similar to sodium meta ...
.
Products likely to contain sulfites at less than 10 ppm (fruits and alcoholic beverages) do not require ingredients labels, and the presence of sulfites usually is undisclosed.
In Australia and New Zealand, sulfites must be declared in the statement of ingredients when present in packaged foods in concentrations of 10 mg/kg (ppm) or more as an ingredient; or as an ingredient of a compound ingredient; or as a food additive or component of a food additive; or as a processing aid or component of a processing aid.
Sulfites that can be added to foods in Canada are potassium bisulfite, potassium metabisulfite, sodium bisulfite, sodium dithionite
Sodium dithionite (also known as sodium hydrosulfite) is a white crystalline powder with a sulfurous odor. Although it is stable in dry air, it decomposes in hot water and in acid solutions.
Structure
The structure has been examined by Raman s ...
, sodium metabisulfite, sodium sulfite, sulfur dioxide and sulfurous acid
Sulfurous acid (also sulfuric(IV) acid, sulphurous acid (UK), sulphuric(IV) acid (UK)) is the chemical compound with the formula . There is no evidence that sulfurous acid exists in solution, but the molecule has been detected in the gas phase ...
. These can also be declared using the common names sulfites, sulfates, sulfiting agents.
In the European Union
The European Union (EU) is a supranational union, supranational political union, political and economic union of Member state of the European Union, member states that are located primarily in Europe, Europe. The union has a total area of ...
, "EU law requires food labels to indicate “contains sulfites” (when exceeding 10 milligrams per kilogram or per litre) without specifying the amount".
Metabolic diseases
High sulfite content in the blood and urine of babies can be caused bymolybdenum cofactor deficiency
Molybdenum cofactor deficiency is a rare human disease in which the absence of molybdopterin – and consequently its molybdenum complex, commonly called molybdenum cofactor – leads to accumulation of toxic levels of sulphite and neurological da ...
disease which leads to neurological damage and early death unless treated. Treatment, requiring daily injections, became available in 2009.
See also
*HSObisulfite
The bisulfite ion (IUPAC-recommended nomenclature: hydrogensulfite) is the ion . Salts containing the ion are also known as "sulfite lyes". Sodium bisulfite is used interchangeably with sodium metabisulfite (Na2S2O5). Sodium metabisulfite diss ...
ion
*S2O metabisulfite
A disulfite, commonly known as metabisulfite or pyrosulfite, is a chemical compound containing the ion . It is a colorless dianion that is primarily marketed in the form of sodium metabisulfite or potassium metabisulfite. When dissolved in wat ...
ion
*H2SO3 sulfurous acid
Sulfurous acid (also sulfuric(IV) acid, sulphurous acid (UK), sulphuric(IV) acid (UK)) is the chemical compound with the formula . There is no evidence that sulfurous acid exists in solution, but the molecule has been detected in the gas phase ...
*SO persulfate ion
*SO sulfate
The sulfate or sulphate ion is a polyatomic anion with the empirical formula . Salts, acid derivatives, and peroxides of sulfate are widely used in industry. Sulfates occur widely in everyday life. Sulfates are salts of sulfuric acid and many ...
ion
*SO hyposulfite ion
*S2O thiosulfate
Thiosulfate ( IUPAC-recommended spelling; sometimes thiosulphate in British English) is an oxyanion of sulfur with the chemical formula . Thiosulfate also refers to the compounds containing this anion, which are the salts of thiosulfuric acid, ...
ion
*SO2 sulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic a ...
*SO3 sulfur trioxide (a sulfate
The sulfate or sulphate ion is a polyatomic anion with the empirical formula . Salts, acid derivatives, and peroxides of sulfate are widely used in industry. Sulfates occur widely in everyday life. Sulfates are salts of sulfuric acid and many ...
precursor)
* Sulfites for a list of sulfites.
*''Grant v The Australian Knitting Mills
''Grant v Australian Knitting Mills'',; . is a landmark case in consumer and negligence law from 1935, holding that where a manufacturer knows that a consumer may be injured if the manufacturer does not take reasonable care, the manufacturer ...
''
References
{{Reflist * Sulfur oxyanions "Allergies: Sulfite Sensitivity". WebMD., 1 February 2007. What Is Sulfite Sensitivity? Foods and Ingredients To Avoid (webmd.com)