Signal peptidase on:
[Wikipedia]
[Google]
[Amazon]
 Signal peptidases are
Signal peptidases are
 Signal peptidases are
Signal peptidases are enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
s that convert secretory and some membrane proteins to their mature or pro forms by cleaving their signal peptide
A signal peptide (sometimes referred to as signal sequence, targeting signal, localization signal, localization sequence, transit peptide, leader sequence or leader peptide) is a short peptide (usually 16-30 amino acids long) present at the N-te ...
s from their N-termini.
Signal peptidases were initially observed in endoplasmic reticulum
The endoplasmic reticulum (ER) is, in essence, the transportation system of the eukaryotic cell, and has many other important functions such as protein folding. It is a type of organelle made up of two subunits – rough endoplasmic reticulum ...
(ER)-derived membrane fractions isolated from mouse myeloma cells. The key observation by César Milstein
César Milstein, CH, FRS (8 October 1927 – 24 March 2002) was an Argentine biochemist in the field of antibody research. Milstein shared the Nobel Prize in Physiology or Medicine in 1984 with Niels Kaj Jerne and Georges J. F. Köhler for d ...
and colleagues was that immunoglobulin light chains were produced in a higher molecular weight form, which became processed by the ER membrane fraction. This finding was directly followed by the discovery of the translocation machinery. Signal peptidases are also found in prokaryotes
A prokaryote () is a single-celled organism that lacks a nucleus and other membrane-bound organelles. The word ''prokaryote'' comes from the Greek πρό (, 'before') and κάρυον (, 'nut' or 'kernel').Campbell, N. "Biology:Concepts & Con ...
as well as the protein import machinery of mitochondria
A mitochondrion (; ) is an organelle found in the cells of most Eukaryotes, such as animals, plants and fungi. Mitochondria have a double membrane structure and use aerobic respiration to generate adenosine triphosphate (ATP), which is used ...
and chloroplasts
A chloroplast () is a type of membrane-bound organelle known as a plastid that conducts photosynthesis mostly in plant and algal cells. The photosynthetic pigment chlorophyll captures the energy from sunlight, converts it, and stores it in ...
.
All signal peptidases described so far are serine proteases. The active site that endoproteolytically cleaves signal peptides
A signal peptide (sometimes referred to as signal sequence, targeting signal, localization signal, localization sequence, transit peptide, leader sequence or leader peptide) is a short peptide (usually 16-30 amino acids long) present at the N-ter ...
from translocated precursor proteins is located at the extracytoplasmic site of the membrane. The eukaryotic signal peptidase is an integral membrane protein
Membrane proteins are common proteins that are part of, or interact with, biological membranes. Membrane proteins fall into several broad categories depending on their location. Integral membrane proteins are a permanent part of a cell membrane ...
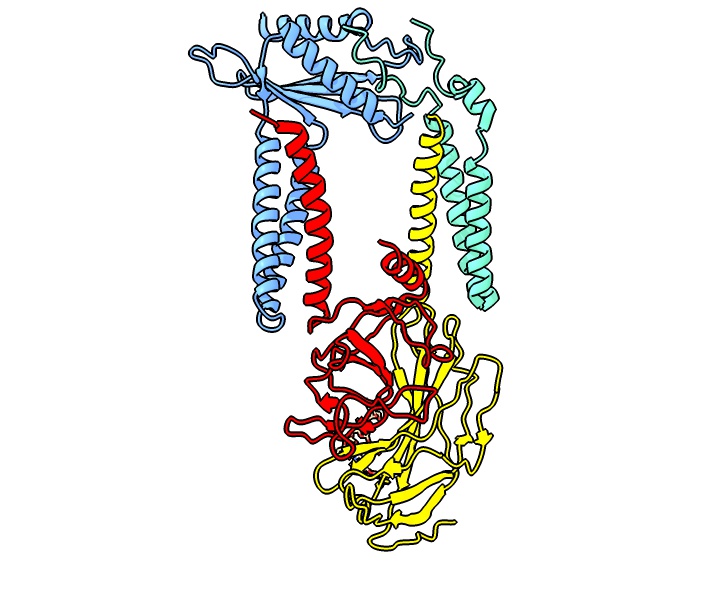
complex. The first subunit, which was identified by yeast genetics is Sec11, a 17 kDa membrane protein that is associated with three subunits termed Spc3p (21 kDa), Spc2p (18 kDa) and Spc1p (11 kDa). Sec11 is the only essential factor for signal peptide processing as can be deduced from a growth defect upon its deletion. The functional signal peptidase complex was first purified from a canine ER membrane fraction. The five mammalian subunits, originally named according to their molecular weight are referred to as SPCS1 (SPC12), SEC11A (SPC18), SEC11C (SPC21), SPCS3 (SPC22/23) and SPCS2 (SPC25). These subunits assemble into two distinct paralogous complexes differing in their catalytic subunit SEC11A and SEC11C, respectively, which exhibit largely identical structures. The SPC structure suggests that the enzyme has a transmembrane domain that is only accessible to signal peptides with their characteristically short helical segment.
References
External links
*Further reading
* {{Portal bar, Biology, border=no EC 3.4.21 EC 3.4.23