Short-term effects of alcohol consumption on:
[Wikipedia]
[Google]
[Amazon]
 The short-term effects of alcohol (more specifically
The short-term effects of alcohol (more specifically
 At low or moderate doses, alcohol acts primarily as a positive allosteric modulator of GABAA. Alcohol binds to several different subtypes of GABAA, but not to others. The main subtypes responsible for the subjective effects of alcohol are the α1β3γ2, α5β3γ2, α4β3δ and α6β3δ subtypes, although other subtypes such as α2β3γ2 and α3β3γ2 are also affected. Activation of these receptors causes most of the effects of alcohol such as relaxation and relief from anxiety, sedation, ataxia and increase in appetite and lowering of inhibitions that can cause a tendency toward violence in some people.
Alcohol has a powerful effect on glutamate as well. Alcohol decreases glutamate's ability to bind with NMDA and acts as an antagonist of the NMDA receptor, which plays a critical role in
At low or moderate doses, alcohol acts primarily as a positive allosteric modulator of GABAA. Alcohol binds to several different subtypes of GABAA, but not to others. The main subtypes responsible for the subjective effects of alcohol are the α1β3γ2, α5β3γ2, α4β3δ and α6β3δ subtypes, although other subtypes such as α2β3γ2 and α3β3γ2 are also affected. Activation of these receptors causes most of the effects of alcohol such as relaxation and relief from anxiety, sedation, ataxia and increase in appetite and lowering of inhibitions that can cause a tendency toward violence in some people.
Alcohol has a powerful effect on glutamate as well. Alcohol decreases glutamate's ability to bind with NMDA and acts as an antagonist of the NMDA receptor, which plays a critical role in
ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
) consumption range from a decrease in anxiety and motor skills and euphoria at lower doses to intoxication (drunkenness), to stupor, unconsciousness, anterograde amnesia
In neurology, anterograde amnesia is the inability to create new memories after the event that caused amnesia, leading to a partial or complete inability to recall the recent past, while long-term memories from before the event remain intact ...
(memory "blackouts"), and central nervous system depression
Central nervous system (CNS) depression is a physiological state that can result in a decreased rate of breathing, decreased heart rate, and loss of consciousness possibly leading to coma or death.
It is the result of inhibited or suppressed brai ...
at higher doses. Cell membrane
The cell membrane (also known as the plasma membrane (PM) or cytoplasmic membrane, and historically referred to as the plasmalemma) is a biological membrane that separates and protects the interior of all cells from the outside environment ( ...
s are highly permeable to alcohol, so once alcohol is in the bloodstream, it can diffuse into nearly every cell in the body.
The concentration of alcohol in blood is measured via blood alcohol content
Blood alcohol content (BAC), also called blood alcohol concentration or blood alcohol level, is a measurement of alcohol intoxication used for legal or medical purposes; it is expressed as mass of alcohol per volume or mass of blood. For example ...
(BAC). The amount and circumstances of consumption play a large role in determining the extent of intoxication; for example, eating a heavy meal before alcohol consumption causes alcohol to absorb more slowly. The amount of alcohol consumed largely determines the extent of hangover
A hangover is the experience of various unpleasant physiological and psychological effects usually following the consumption of alcohol, such as wine, beer, and liquor. Hangovers can last for several hours or for more than 24 hours. Typical sympto ...
s, although hydration also plays a role. After excessive drinking, stupor and unconsciousness can both occur. Extreme levels of consumption can cause alcohol poisoning
Alcohol intoxication, also known as alcohol poisoning, commonly described as drunkenness or inebriation, is the negative behavior and physical effects caused by a recent consumption of alcohol. In addition to the toxicity of ethanol, the main ps ...
and death; in fact, a concentration in the blood stream of 0.36% will kill half of those affected. Alcohol may also cause death indirectly by asphyxiation
Asphyxia or asphyxiation is a condition of deficient supply of oxygen to the body which arises from abnormal breathing. Asphyxia causes generalized hypoxia, which affects primarily the tissues and organs. There are many circumstances that ca ...
, caused from vomiting.
Alcohol can greatly exacerbate sleep problems. During abstinence, residual disruptions in sleep regularity and sleep patterns are the greatest predictors of relapse
In internal medicine, relapse or recidivism is a recurrence of a past (typically medical) condition. For example, multiple sclerosis and malaria often exhibit peaks of activity and sometimes very long periods of dormancy, followed by relapse or ...
.
Effects by dosage
The definition of a unit of alcohol ranges between 8 and 14 grams of pure alcohol/ethanol depending on the country. There is no agreement on definitions of a low, moderate or high dose of alcohol either. The U.S. National Institute on Alcohol Abuse and Alcoholism defines a moderate dose as alcohol intake up to two standard drinks or 28 grams for men and one standard drink or 14 grams for women. The immediate effect of alcohol depends on the drinker's blood alcohol concentration (BAC). BAC can be different for each person depending on their age, sex, pre-existing health condition, even if they drink the same amount of alcohol. Different BACs have different effects. The following lists describe the common effects of alcohol on the body depending on the BAC. However, tolerance varies considerably between individuals, as does individual response to a given dosage; the effects of alcohol differ widely between people. Hence in this context, BAC percentages are just estimates used for illustrative purposes.
Grand Rapids Dip
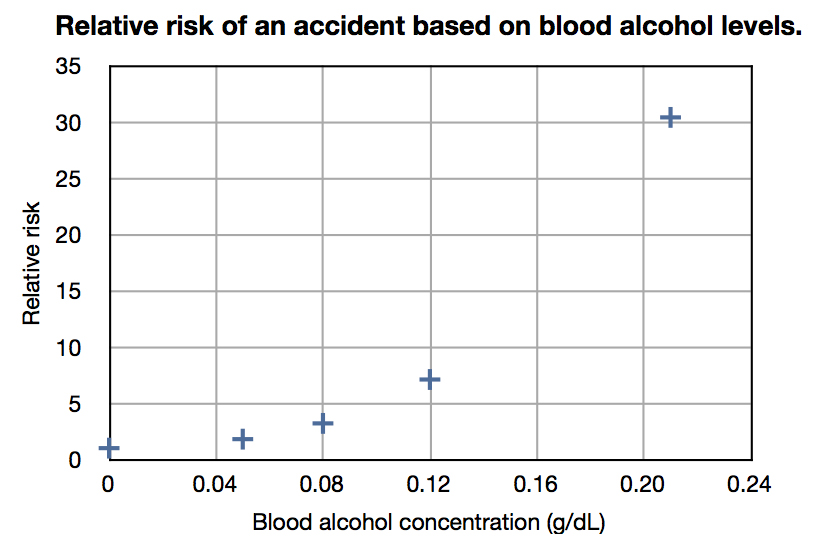
Studies suggest that a BAC of 0.01–0.04% slightly lowers risk of being in a vehicle accident when compared to a BAC of 0.00%, referred to as the ''Grand Rapids Effect'' or ''Grand Rapids Dip''. Some literature has attributed the ''Grand Rapids Effect'' to erroneous data or asserted (without support) that it was possibly due to drivers exerting extra caution at low BAC levels or due to "experience" in drinking. Other explanations are that this effect is at least in part the blocking effect of ethanolexcitotoxicity
In excitotoxicity, nerve cells suffer damage or death when the levels of otherwise necessary and safe neurotransmitters such as glutamate become pathologically high, resulting in excessive stimulation of receptors. For example, when glutamate ...
and the effect of alcohol in essential tremor
Essential tremor (ET), also called benign tremor, familial tremor, and idiopathic tremor, is a medical condition characterized by involuntary rhythmic contractions and relaxations ( oscillations or twitching movements) of certain muscle groups in ...
and other movement disorders, but this remains speculative.
Moderate doses
Ethanol inhibits the ability of glutamate to open the cation channel associated with the N-methyl-D-aspartate (NMDA) subtype of glutamate receptors. Stimulated areas include thecortex
Cortex or cortical may refer to:
Biology
* Cortex (anatomy), the outermost layer of an organ
** Cerebral cortex, the outer layer of the vertebrate cerebrum, part of which is the ''forebrain''
*** Motor cortex, the regions of the cerebral cortex i ...
, hippocampus
The hippocampus (via Latin from Greek , ' seahorse') is a major component of the brain of humans and other vertebrates. Humans and other mammals have two hippocampi, one in each side of the brain. The hippocampus is part of the limbic system, ...
, and nucleus accumbens
The nucleus accumbens (NAc or NAcc; also known as the accumbens nucleus, or formerly as the ''nucleus accumbens septi'', Latin for " nucleus adjacent to the septum") is a region in the basal forebrain rostral to the preoptic area of the hypot ...
, which are all responsible for both thinking
In their most common sense, the terms thought and thinking refer to conscious cognitive processes that can happen independently of sensory stimulation. Their most paradigmatic forms are judging, reasoning, concept formation, problem solving, an ...
and pleasure seeking. Another one of alcohol's agreeable effects is body relaxation, which is possibly caused by neurons transmitting electrical signals in an alpha waves-pattern; such waves are actually observed (with the aid of EEG
Electroencephalography (EEG) is a method to record an electrogram of the spontaneous electrical activity of the brain. The biosignals detected by EEG have been shown to represent the postsynaptic potentials of pyramidal neurons in the neocortex ...
s) whenever the body is relaxed.
Short-term effects of alcohol include the risk of injuries, violence, and fetal damage. Alcohol has also been linked with lowered inhibitions, although it is unclear as to what degree this is chemical or psychological as studies with placebo
A placebo ( ) is a substance or treatment which is designed to have no therapeutic value. Common placebos include inert tablets (like sugar pills), inert injections (like saline), sham surgery, and other procedures.
In general, placebos can af ...
s can often duplicate the social effects of alcohol at either low or moderate doses. Some studies have suggested that intoxicated people have much greater control over their behavior than is generally recognized, though they have a reduced ability to evaluate the consequences of their behavior. Behavioral changes associated with drunkenness are, to some degree, contextual.
Areas of the brain that are responsible for planning and motor learning are sharpened. A related effect, which is caused by even low levels of alcohol, is the tendency for people to become more animated in speech and movement. This is caused by increased metabolism
Metabolism (, from el, μεταβολή ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run c ...
in areas of the brain associated with movement, such as the nigrostriatal pathway The nigrostriatal pathway is a bilateral dopaminergic pathway in the brain that connects the substantia nigra pars compacta (SNc) in the midbrain with the dorsal striatum (i.e., the caudate nucleus and putamen) in the forebrain. It is one of the f ...
. This causes reward systems in the brain to become more active, which may induce certain individuals to behave in an uncharacteristically loud and cheerful manner.
Alcohol has been known to mitigate the production of antidiuretic hormone, which is a hormone that acts on the kidney to favor water reabsorption in the kidneys during filtration. This occurs because alcohol confuses osmoreceptor
An osmoreceptor is a sensory receptor primarily found in the hypothalamus of most homeothermic organisms that detects changes in osmotic pressure. Osmoreceptors can be found in several structures, including two of the circumventricular organs – ...
s in the hypothalamus
The hypothalamus () is a part of the brain that contains a number of small nuclei with a variety of functions. One of the most important functions is to link the nervous system to the endocrine system via the pituitary gland. The hypothalamu ...
, which relay osmotic pressure information to the posterior pituitary
The posterior pituitary (or neurohypophysis) is the posterior lobe of the pituitary gland which is part of the endocrine system. The posterior pituitary is not glandular as is the anterior pituitary. Instead, it is largely a collection of axonal ...
, the site of antidiuretic hormone release. Alcohol causes the osmoreceptors to signal that there is low osmotic pressure in the blood, which triggers an inhibition of the antidiuretic hormone. As a consequence, one's kidneys are no longer able to reabsorb as much water as they should be absorbing, therefore creating excessive volumes of urine and the subsequent overall dehydration
In physiology, dehydration is a lack of total body water, with an accompanying disruption of metabolic processes. It occurs when free water loss exceeds free water intake, usually due to exercise, disease, or high environmental temperature. Mil ...
.
Excessive doses
Acute alcohol intoxication
Alcohol intoxication, also known as alcohol poisoning, commonly described as drunkenness or inebriation, is the negative behavior and physical effects caused by a recent consumption of alcohol. In addition to the toxicity of ethanol, the main ps ...
through excessive doses in general causes short- or long-term health effects. NMDA
''N''-methyl--aspartic acid or ''N''-methyl--aspartate (NMDA) is an amino acid derivative that acts as a specific agonist at the NMDA receptor mimicking the action of glutamate, the neurotransmitter which normally acts at that receptor. Unlike ...
receptors become unresponsive, slowing areas of the brain for which they are responsible. Contributing to this effect is the activity that alcohol induces in the gamma-aminobutyric acid (GABA) system. The GABA system is known to inhibit activity in the brain. GABA could also be responsible for causing the memory impairment
Amnesia is a deficit in memory caused by brain damage or disease,Gazzaniga, M., Ivry, R., & Mangun, G. (2009) Cognitive Neuroscience: The biology of the mind. New York: W.W. Norton & Company. but it can also be caused temporarily by the use ...
that many people experience. It has been asserted that GABA signals interfere with both the ''registration'' and the ''consolidation'' stages of memory formation. As the GABA system is found in the hippocampus (among other areas in the CNS), which is thought to play a large role in memory formation, this is thought to be possible.
Anterograde amnesia
In neurology, anterograde amnesia is the inability to create new memories after the event that caused amnesia, leading to a partial or complete inability to recall the recent past, while long-term memories from before the event remain intact ...
, colloquially referred to as " blacking out", is another symptom of heavy drinking. This is the loss of memory during and after an episode of drinking.
Another classic finding of alcohol intoxication is ataxia
Ataxia is a neurological sign consisting of lack of voluntary coordination of muscle movements that can include gait abnormality, speech changes, and abnormalities in eye movements. Ataxia is a clinical manifestation indicating dysfunction of ...
, in its appendicular, gait, and truncal forms. Appendicular ataxia results in jerky, uncoordinated movements of the limbs, as if each muscle were working independently from the others. Truncal ataxia
Truncal ataxia (or trunk ataxia) is a wide-based "drunken sailor" gait characterised by uncertain starts and stops, lateral deviations and unequal steps. It is an instability of the trunk and often seen during sitting. It is most visible when sh ...
results in postural instability; gait instability is manifested as a disorderly, wide-based gait with inconsistent foot positioning. Ataxia causes the observation that drunk people are clumsy, sway back and forth, and often fall down. It is presumed to be due to alcohol's effect on the cerebellum.
Mellanby effect
The Mellanby effect is the phenomenon that the behavioral impairment due to alcohol is less, at the same BAC, when the BAC is decreasing than when it is increasing. In other words, at a given point on the upward slope of BAC (while drinking), impairment is greater than at a given point on the downward slope (after drinking), even though the BAC is the same at the two points. This effect was confirmed in a 2017 meta-analysis.Effect on different population
Based on sex
Alcohol affects males and females differently because of difference in body fat percentage and water content. On average, for equal body weight, women have a higher body fat percentage than men. Since alcohol is absorbed into body water content and men have more water in their bodies than women, for women there will be a higher blood alcohol concentration from same amount of alcohol consumption. Women are also thought to have less alcohol dehydrogenase (ADH) enzyme which is required to break down alcohol. That is why the drinking guidelines are different for men and women.Based on genetic variation
Alcohol metabolism depends on the enzymes alcohol dehydrogenase (ADH) and aldehyde dehydrogenase (ALDH). Genetic variants of the genes coding for these enzymes can affect the rate of alcohol metabolism. Some ADH gene variants lead to higher metabolic activity, resulting in the accumulation of acetaldehyde, whereas, a null allele in ALDH2 causes an accumulation of acetaldehyde by preventing its catabolism to acetate. The genetic variants of these enzymes can explain the differences in the alcohol metabolism in different races. The different isoforms of ADH showed protection against alcoholic disorders in Han Chinese and Japanese (due to presence of ADH1B*2 ) and in African (due to presence of ADH1B*3). On the other hand, presence of ALDH2*2 in East Asians (a variant of the ALDH gene), can cause blood acetaldehyde levels of 30 to 75 μM or higher, which is more than 10 times the normal level. The excess amount of blood aldehyde produce facial flushing, nausea, rapid heartbeat, and other adverse effects. Presence of these alleles causes rapid conversion of alcohol to acetaldehyde which can be toxic in large amount. So, the East Asians and Africans feel the adverse effects of alcohol early and stop drinking. For Caucasians, ADH1B*1 allele is the most prevalent allele which causes slower conversion of alcohol to acetaldehyde and it makes them more vulnerable to alcohol use disorders.Allergic reaction-like symptoms
Humans metabolizeethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
primarily through NAD-dependent alcohol dehydrogenase
Alcohol dehydrogenases (ADH) () are a group of dehydrogenase enzymes that occur in many organisms and facilitate the interconversion between alcohols and aldehydes or ketones with the reduction of nicotinamide adenine dinucleotide (NAD+) to NA ...
(ADH) class I enzymes (i.e. ADH1A
Alcohol dehydrogenase 1A is an enzyme that in humans is encoded by the ''ADH1A'' gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ...
, ADH1B
Alcohol dehydrogenase 1B is an enzyme that in humans is encoded by the ''ADH1B'' gene.
The protein encoded by this gene is a member of the alcohol dehydrogenase family. Members of this enzyme family metabolize a wide variety of substrates, includ ...
, and ADH1C
Alcohol dehydrogenase 1C is an enzyme that in humans is encoded by the ''ADH1C'' gene.
Function
This gene encodes class I alcohol dehydrogenase
Alcohol dehydrogenases (ADH) () are a group of dehydrogenase enzymes that occur in many organi ...
) to acetaldehyde and then metabolize acetaldehyde primarily by NAD-dependent aldehyde dehydrogenase 2 (ALDH2
Aldehyde dehydrogenase, mitochondrial is an enzyme that in humans is encoded by the ''ALDH2'' gene located on chromosome 12. This protein belongs to the aldehyde dehydrogenase family of enzymes. Aldehyde dehydrogenase is the second enzyme of the ...
) to acetic acid. Eastern Asians reportedly have a deficiency in acetaldehyde metabolism in a surprisingly high percentage (approaching 50%) of their populations. The issue has been most thoroughly investigated in native Japanese where persons with a single-nucleotide polymorphism
In genetics, a single-nucleotide polymorphism (SNP ; plural SNPs ) is a germline substitution of a single nucleotide at a specific position in the genome. Although certain definitions require the substitution to be present in a sufficiently lar ...
(SNP) variant allele
An allele (, ; ; modern formation from Greek ἄλλος ''állos'', "other") is a variation of the same sequence of nucleotides at the same place on a long DNA molecule, as described in leading textbooks on genetics and evolution.
::"The chro ...
of the ALDH2 gene were found; the variant allele, encodes lysine (lys) instead of glutamic acid (glu) at amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha a ...
487; this renders the enzyme essentially inactive in metabolizing acetaldehyde to acetic acid. The variant allele is variously termed glu487lys, ALDH2*2, and ALDH2*504lys. In the overall Japanese population, about 57% of individuals are homozygous
Zygosity (the noun, zygote, is from the Greek "yoked," from "yoke") () is the degree to which both copies of a chromosome or gene have the same genetic sequence. In other words, it is the degree of similarity of the alleles in an organism.
Mo ...
for the normal allele (sometimes termed ALDH2*1), 40% are heterozygous
Zygosity (the noun, zygote, is from the Greek "yoked," from "yoke") () is the degree to which both copies of a chromosome or gene have the same genetic sequence. In other words, it is the degree of similarity of the alleles in an organism.
Mo ...
for glu487lys, and 3% are homozygous for glu487lys. Since ALDH2 assembles and functions as a tetramer
A tetramer () ('' tetra-'', "four" + '' -mer'', "parts") is an oligomer formed from four monomers or subunits. The associated property is called ''tetramery''. An example from inorganic chemistry is titanium methoxide with the empirical formula ...
and since ALDH2 tetramers containing one or more glu487lys proteins are also essentially inactive (i.e. the variant allele behaves as a dominant negative
In genetics, dominance is the phenomenon of one variant (allele) of a gene on a chromosome masking or overriding the effect of a different variant of the same gene on the other copy of the chromosome. The first variant is termed dominant and t ...
), homozygote individuals for glu487lys have undetectable while heterozygote individuals for glu487lys have little ALDH2 activity. In consequence, Japanese individuals homozygous or, to only a slightly lesser extent, heterozygous for glu487lys metabolize ethanol to acetaldehyde normally but metabolize acetaldehyde poorly and are susceptible to a set of adverse responses to the ingestion of, and sometimes even the fumes from, ethanol and ethanol-containing beverages; these responses include the transient accumulation of acetaldehyde in blood and tissues; facial flushing
Flushing may refer to:
Places
* Flushing, Cornwall, a village in the United Kingdom
* Flushing, Queens, New York City
** Flushing Bay, a bay off the north shore of Queens
** Flushing Chinatown (法拉盛華埠), a community in Queens
** Flushin ...
(i.e. the "oriental flushing syndrome" or Alcohol flush reaction
Alcohol flush reaction is a condition in which a person develops flushes or blotches associated with erythema on the face, neck, shoulders, and in some cases, the entire body after consuming alcoholic beverages. The reaction is the result of an a ...
), urticaria
Hives, also known as urticaria, is a kind of skin rash with red, raised, itchy bumps. Hives may burn or sting. The patches of rash may appear on different body parts, with variable duration from minutes to days, and does not leave any long-last ...
, systemic dermatitis
Dermatitis is inflammation of the skin, typically characterized by itchiness, redness and a rash. In cases of short duration, there may be small blisters, while in long-term cases the skin may become thickened. The area of skin involved can ...
, and alcohol-induced respiratory reactions
Alcohol-induced respiratory reactions, also termed alcohol-induced asthma and alcohol-induced respiratory symptoms, are increasingly recognized as a pathological bronchoconstriction response to the consumption of alcohol that afflicts many people w ...
(i.e. rhinitis
Rhinitis, also known as coryza, is irritation and inflammation of the mucous membrane inside the nose. Common symptoms are a stuffy nose, runny nose, sneezing, and post-nasal drip.
The inflammation is caused by viruses, bacteria, irrita ...
and, primarily in patients with a history of asthma
Asthma is a long-term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and easily triggered bronchospasms. Symptoms include episodes of wheezing, co ...
, mild to moderately bronchoconstriction
Bronchoconstriction is the constriction of the airways in the lungs due to the tightening of surrounding smooth muscle, with consequent coughing, wheezing, and shortness of breath.
Causes
The condition has a number of causes, the most common be ...
exacerbations of their asthmatic disease. These allergic reaction-like symptoms, which typically occur within 30–60 minutes of ingesting alcoholic beverages, do not appear to reflect the operation of classical IgE
Immunoglobulin E (IgE) is a type of antibody (or immunoglobulin (Ig) " isotype") that has been found only in mammals. IgE is synthesised by plasma cells. Monomers of IgE consist of two heavy chains (ε chain) and two light chains, with the ε c ...
- or T cell
A T cell is a type of lymphocyte. T cells are one of the important white blood cells of the immune system and play a central role in the adaptive immune response. T cells can be distinguished from other lymphocytes by the presence of a T-cell r ...
-related allergen
An allergen is a type of antigen that produces an abnormally vigorous immune response in which the immune system fights off a perceived threat that would otherwise be harmless to the body. Such reactions are called allergies.
In technical terms ...
-induced reactions but rather are due, at least in large part, to the action of acetaldehyde in stimulating tissues to release histamine
Histamine is an organic nitrogenous compound involved in local immune responses, as well as regulating physiological functions in the gut and acting as a neurotransmitter for the brain, spinal cord, and uterus. Since histamine was discovered ...
, the probable evoker of these symptoms.
The percentages of glu487lys heterozygous plus homozygous genotypes
The genotype of an organism is its complete set of genetic material. Genotype can also be used to refer to the alleles or variants an individual carries in a particular gene or genetic location. The number of alleles an individual can have in a ...
are about 35% in native Caboclo
A caboclo () is a person of mixed Indigenous Brazilian and European ancestry, or, less commonly, a culturally assimilated or detribalized person of full Amerindian descent. In Brazil, a ''caboclo'' generally refers to this specific type of '' ...
of Brazil, 30% in Chinese, 28% in Koreans, 11% in Thai people
Thai people ( th, ชาวไทย; '' endonym''), Central Thai people ( th, คนภาคกลาง, sou, คนใต้, ตามโพร; ''exonym and also domestically'') or Siamese ( th, ชาวสยาม; ''historical exonym an ...
, 7% in Malaysians, 3% in natives of India, 3% in Hungarians, and 1% in Filipinos; percentages are essentially 0 in individuals of Native African descent, Caucasians of Western European descent, Turks, Australian Aborigines, Australians of Western European descent, Swedish Lapps, and Alaskan Eskimos. The prevalence of ethanol-induced allergic symptoms in 0 or low levels of glu487lys genotypes commonly ranges above 5%. These "ethanol reactors" may have other gene-based abnormalities that cause the accumulation of acetaldehyde following the ingestion of ethanol or ethanol-containing beverages. For example, the surveyed incidence of self-reported ethanol-induced flushing reactions in Scandinavians living in Copenhagen as well as Australians of European descent is about 16% in individuals homozygous for the "normal" ADH1B gene but runs to ~23% in individuals with the ADH1-Arg48His SNP variant; in vitro
''In vitro'' (meaning in glass, or ''in the glass'') studies are performed with microorganisms, cells, or biological molecules outside their normal biological context. Colloquially called " test-tube experiments", these studies in biology ...
, this variant metabolizes ethanol rapidly and in humans, it is proposed, may form acetaldehyde at levels that exceed the capacity of ALDH2 to metabolize. Notwithstanding such considerations, experts suggest that the large proportion of alcoholic beverage-induced allergic-like symptoms in populations with a low incidence of the glu487lys genotype reflect true allergic reactions to the natural and/or contaminating allergens particularly those in wines and to a lesser extent beers.
Pathophysiology
LTP
LTP may refer to:
Biology and medicine
* Lateral tibial plateau, part of a leg bone
* Lipid transfer proteins, proteins found in plant tissues
* Long-term potentiation (neurophysiology), a long-lasting enhancement in signal transmission between ...
by allowing Ca2+
Calcium ions (Ca2+) contribute to the physiology and biochemistry of organisms' cells. They play an important role in signal transduction pathways, where they act as a second messenger, in neurotransmitter release from neurons, in contraction of ...
to enter the cell. These inhibitory effects are thought to be responsible for the "memory blanks" that can occur at levels as low as 0.03% blood level. In addition, reduced glutamate release in the dorsal hippocampus has been linked to spatial memory loss. Chronic alcohol users experience an upregulation of NMDA receptors because the brain is attempting to reestablish homeostasis
In biology, homeostasis (British also homoeostasis) (/hɒmɪə(ʊ)ˈsteɪsɪs/) is the state of steady internal, physical, and chemical conditions maintained by living systems. This is the condition of optimal functioning for the organism and ...
. When a chronic alcohol user stops drinking for more than 10 hours, apoptosis can occur due to excitotoxicity
In excitotoxicity, nerve cells suffer damage or death when the levels of otherwise necessary and safe neurotransmitters such as glutamate become pathologically high, resulting in excessive stimulation of receptors. For example, when glutamate ...
. The seizures experienced during alcohol abstinence are thought to be a result of this NMDA upregulation. Alteration of NMDA receptor numbers in chronic alcoholics is likely to be responsible for some of the symptoms seen in delirium tremens during severe alcohol withdrawal, such as delirium and hallucinations. Other targets such as sodium channels can also be affected by high doses of alcohol, and alteration in the numbers of these channels in chronic alcoholics is likely to be responsible for as well as other effects such as cardiac
The heart is a muscular organ in most animals. This organ pumps blood through the blood vessels of the circulatory system. The pumped blood carries oxygen and nutrients to the body, while carrying metabolic waste such as carbon dioxide to t ...
arrhythmia. Other targets that are affected by alcohol include cannabinoid
Cannabinoids () are several structural classes of compounds found in the cannabis plant primarily and most animal organisms (although insects lack such receptors) or as synthetic compounds. The most notable cannabinoid is the phytocannabinoid tet ...
, opioid
Opioids are substances that act on opioid receptors to produce morphine-like effects. Medically they are primarily used for pain relief, including anesthesia. Other medical uses include suppression of diarrhea, replacement therapy for opioid use ...
and dopamine receptors, although it is unclear whether alcohol affects these directly or if they are affected by downstream consequences of the GABA/NMDA effects. People with a family history of alcoholism may exhibit genetic differences in the response of their NMDA glutamate receptors as well as the ratios of GABAA subtypes in their brain. Alcohol inhibits sodium-potassium pumps in the cerebellum and this is likely how it corrupts cerebellar computation and body co-ordination.
Contrary to popular belief, research suggests that acute exposure to alcohol is not neurotoxic
Neurotoxicity is a form of toxicity in which a biological, chemical, or physical agent produces an adverse effect on the structure or function of the central and/or peripheral nervous system. It occurs when exposure to a substance – specificall ...
in adults and actually prevents NMDA antagonist
NMDA receptor antagonists are a class of drugs that work to antagonize, or inhibit the action of, the ''N''-Methyl-D-aspartate receptor ( NMDAR). They are commonly used as anesthetics for animals and humans; the state of anesthesia they induce ...
-induced neurotoxicity.
Alcohol and sleep
Low doses of alcohol (onebeer
Beer is one of the oldest and the most widely consumed type of alcoholic drink in the world, and the third most popular drink overall after water and tea. It is produced by the brewing and fermentation of starches, mainly derived from ce ...
) appear to increase total sleep time and reduce awakening during the night. The sleep-promoting benefits of alcohol dissipate
In thermodynamics, dissipation is the result of an irreversible process that takes place in homogeneous thermodynamic systems. In a dissipative process, energy (internal, bulk flow kinetic, or system potential) transforms from an initial form to ...
at moderate and higher doses of alcohol. Previous experience with alcohol also influences the extent to which alcohol positively or negatively affects sleep. Under free-choice conditions, in which subjects chose between drinking alcohol or water, inexperienced drinkers were sedated while experienced drinkers were stimulated following alcohol consumption. In insomnia
Insomnia, also known as sleeplessness, is a sleep disorder in which people have trouble sleeping. They may have difficulty falling asleep, or staying asleep as long as desired. Insomnia is typically followed by daytime sleepiness, low energy, ...
cs, moderate doses of alcohol improve sleep maintenance.
Moderate alcohol consumption 30–60 minutes before sleep, although decreasing, disrupts sleep architecture. Rebound effects occur once the alcohol has been largely metabolized, causing late night disruptions in sleep maintenance. Under conditions of moderate alcohol consumption where blood alcohol levels average 0.06–0.08 percent and decrease 0.01–0.02 percent per hour, an alcohol clearance rate of 4–5 hours would coincide with disruptions in sleep maintenance in the second half of an 8-hour sleep episode. In terms of sleep architecture, moderate doses of alcohol facilitate "rebounds" in rapid eye movement (REM) following suppression in REM and stage 1 sleep in the first half of an 8-hour sleep episode, REM and stage 1 sleep increase well beyond baseline in the second half. Moderate doses of alcohol also very quickly increase slow wave sleep
Slow-wave sleep (SWS), often referred to as deep sleep, consists of stage three of non-rapid eye movement sleep. It usually lasts between 70 and 90 minutes and takes place during the first hours of the night. Initially, SWS consisted of both S ...
(SWS) in the first half of an 8-hour sleep episode. Enhancements in REM sleep and SWS following moderate alcohol consumption are mediated by reductions in glutamatergic
Glutamatergic means "related to glutamate". A glutamatergic agent (or drug) is a chemical that directly modulates the excitatory amino acid (glutamate/aspartate) system in the body or brain. Examples include excitatory amino acid receptor agonis ...
activity by adenosine in the central nervous system. In addition, tolerance to changes in sleep maintenance and sleep architecture develops within three days of alcohol consumption before bedtime.
Alcohol consumption and balance
Alcohol can affect balance by altering theviscosity
The viscosity of a fluid is a measure of its resistance to deformation at a given rate. For liquids, it corresponds to the informal concept of "thickness": for example, syrup has a higher viscosity than water.
Viscosity quantifies the inte ...
of the endolymph
Endolymph is the fluid contained in the membranous labyrinth of the inner ear. The major cation in endolymph is potassium, with the values of sodium and potassium concentration in the endolymph being 0.91 mM and 154 mM, respectively. I ...
within the otolithic membrane, the fluid inside the semicircular canal
In mathematics (and more specifically geometry), a semicircle is a one-dimensional locus of points that forms half of a circle. The full arc of a semicircle always measures 180° (equivalently, radians, or a half-turn). It has only one line o ...
s inside the ear. The endolymph surrounds the ampullary cupula
The ampullary cupula, or cupula, is a structure in the vestibular system, providing the sense of spatial orientation.
The cupula is located within the ampullae of each of the three semicircular canals. Part of the crista ampullaris, the cupula h ...
which contains hair cells
Hair cells are the sensory receptors of both the auditory system and the vestibular system in the ears of all vertebrates, and in the lateral line organ of fishes. Through mechanotransduction, hair cells detect movement in their environment. ...
within the semicircular canals. When the head is tilted, the endolymph flows and moves the cupula. The hair cells then bend and send signals to the brain indicating the direction in which the head is tilted. By changing the viscosity of the endolymph to become less dense when alcohol enters the system, the hair cells can move more easily within the ear, which sends the signal to the brain and results in exaggerated and overcompensated movements of body. This can also result in vertigo
Vertigo is a condition where a person has the sensation of movement or of surrounding objects moving when they are not. Often it feels like a spinning or swaying movement. This may be associated with nausea, vomiting, sweating, or difficulties w ...
, or "the spins".
Alcohol and postprandial triglycerides
Alcohol taken with a meal increases and prolongs postprandial triglyceridemia. This is true despite the observation that the relationship between alcohol consumption and triglyceridemia is "J-shaped," meaning that fasting triglycerides concentration is lower in people who drink 10–20 g/alcohol a day compared to people who either abstain from alcohol or who drink more per day.Alcohol and blood pressure
A systematic review reported that alcohol has bi-phasic effect on blood pressure. Both systolic and diastolic blood pressure fell when they were measured couple of hours after alcohol consumption. However, the longer term measurement (20 hours average) showed a modest but statistically significant increase in blood pressure: a 2.7 mmHg rise in systolic blood pressure and 1.4 mmHg rise in diastolic blood pressure. A Cochrane systematic review based on only randomized controlled trials which investigates the acute effect of alcohol consumption in healthy and hypertensive adults is in progress.Alcohol and pain
A 2015 literature review found that alcohol administration confers acute pain-inhibitory effects. It also found the relationship between alcohol consumption and pain is curvilinear; moderate alcohol use was associated with positive pain-related outcomes and heavy alcohol use was associated with negative pain-related outcomes.See also
*Alcohol and health
Alcohol (also known as ethanol) has a number of effects on health. Short-term effects of alcohol consumption include intoxication and dehydration. Long-term effects of alcohol include changes in the metabolism of the liver and brain, several typ ...
* Long-term effects of alcohol consumption
The long-term heavy consumption of alcohol (alcohol use disorder) can cause severe detrimental effects. Health effects associated with alcohol intake in large amounts include an increased risk of developing an alcohol use disorder, malnutrition, ...
References
Further reading
* * {{DEFAULTSORT:Short-Term Effects of Alcohol Drinking culture Short