Polyurea on:
[Wikipedia]
[Google]
[Amazon]
 Polyurea is a type of elastomer that is derived from the reaction product of an
Polyurea is a type of elastomer that is derived from the reaction product of an
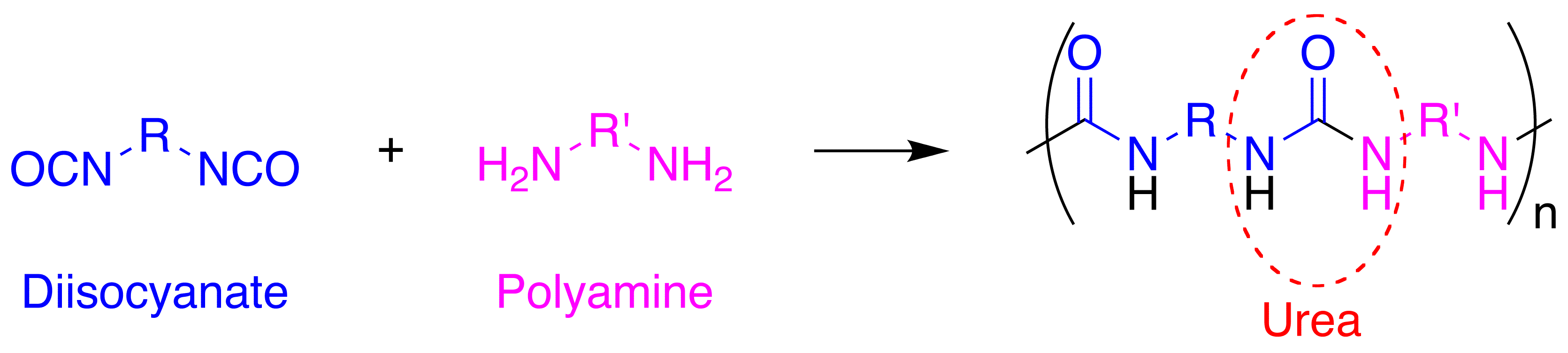 The word polyurea is derived from the Greek words πολυ- - poly- meaning "many"; and ουρίας - oûron meaning "to urinate" (referring to the substance
The word polyurea is derived from the Greek words πολυ- - poly- meaning "many"; and ουρίας - oûron meaning "to urinate" (referring to the substance
isocyanate
In organic chemistry, isocyanate is the functional group with the formula . Organic compounds that contain an isocyanate group are referred to as isocyanates. An organic compound with two isocyanate groups is known as a diisocyanate. Diisocyan ...
component and a synthetic resin
Synthetic resins are industrially produced resins, typically viscous substances that convert into rigid polymers by the process of curing. In order to undergo curing, resins typically contain reactive end groups, such as acrylates or epoxides. ...
blend component through step-growth polymerization. The isocyanate
In organic chemistry, isocyanate is the functional group with the formula . Organic compounds that contain an isocyanate group are referred to as isocyanates. An organic compound with two isocyanate groups is known as a diisocyanate. Diisocyan ...
can be aromatic
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to satur ...
or aliphatic
In organic chemistry, hydrocarbons ( compounds composed solely of carbon and hydrogen) are divided into two classes: aromatic compounds and aliphatic compounds (; G. ''aleiphar'', fat, oil). Aliphatic compounds can be saturated, like hexane, ...
in nature. It can be monomer
In chemistry, a monomer ( ; '' mono-'', "one" + ''-mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization.
Classification
...
, polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
, or any variant reaction of isocyanates, quasi-prepolymer or a prepolymer
In polymer chemistry, the term prepolymer or pre-polymer, refers to a monomer or system of monomers that have been reacted to an intermediate-molecular mass state. This material is capable of further polymerization by reactive groups to a fully c ...
. The prepolymer, or quasi-prepolymer, can be made of an amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen
Hydrogen is the chemical element wi ...
-terminated polymer resin, or a hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydro ...
-terminated polymer resin.
The resin blend may be made up of amine-terminated polymer resins, and/or amine-terminated chain extenders. The amine-terminated polymer resins do not have any intentional hydroxyl moieties. Any hydroxyls are the result of incomplete conversion to the amine-terminated polymer resins. The resin blend may also contain additives or non-primary components. These additives may contain hydroxyls, such as pre-dispersed pigments
A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compoun ...
in a polyol
In organic chemistry, a polyol is an organic compound containing multiple hydroxyl groups (). The term "polyol" can have slightly different meanings depending on whether it is used in food science or polymer chemistry. Polyols containing two, thr ...
carrier. Normally, the resin blend does not contain a catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
(s).
Polymer structure
 The word polyurea is derived from the Greek words πολυ- - poly- meaning "many"; and ουρίας - oûron meaning "to urinate" (referring to the substance
The word polyurea is derived from the Greek words πολυ- - poly- meaning "many"; and ουρίας - oûron meaning "to urinate" (referring to the substance urea
Urea, also known as carbamide, is an organic compound with chemical formula . This amide has two amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid.
Urea serves an important ...
, found in urine
Urine is a liquid by-product of metabolism in humans and in many other animals. Urine flows from the kidneys through the ureters to the urinary bladder. Urination results in urine being excreted from the body through the urethra.
Cellular ...
). Urea or carbamide is an organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. T ...
with the chemical formula (NH2)2CO. The molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioche ...
has two amine groups (–NH2) joined by a carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containi ...
functional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the re ...
(C=O). In a polyurea, alternating monomer
In chemistry, a monomer ( ; '' mono-'', "one" + ''-mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization.
Classification
...
units of isocyanates and amines react with each other to form urea linkages. Ureas can also be formed from the reaction of isocyanates and water which forms a carbamic acid
Carbamic acid, which might also be called aminoformic acid or aminocarboxylic acid, is the chemical compound with the formula . It can be obtained by the reaction of ammonia and carbon dioxide at very low temperatures, which also yields an equ ...
intermediate. This acid quickly decomposes by splitting off carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
and leaving behind an amine. This amine then reacts with another isocyanate group to form the polyurea linkage. This two step reaction is used to make what is commonly but improperly called polyurethane foam. The carbon dioxide that is liberated in this reaction is the primary blowing (foaming) agent especially in many polyurethane foams which more precisely should be called polyurethane/urea foams.
Uses
Polyurea andpolyurethane
Polyurethane (; often abbreviated PUR and PU) refers to a class of polymers composed of organic units joined by carbamate (urethane) links. In contrast to other common polymers such as polyethylene and polystyrene, polyurethane is produced from ...
are copolymer
In polymer chemistry, a copolymer is a polymer derived from more than one species of monomer. The polymerization of monomers into copolymers is called copolymerization. Copolymers obtained from the copolymerization of two monomer species are some ...
s used in the manufacture of spandex
Spandex, Lycra, or elastane is a synthetic fiber known for its exceptional elasticity. It is a polyether-polyurea copolymer that was invented in 1958 by chemist Joseph Shivers at DuPont's Benger Laboratory in Waynesboro, Virginia, US.
The g ...
, which was invented in 1959.
Polyurea was originally developed to protect tabletop edges which led to the development of two-component polyurethane and polyurea spray elastomers took place in the 1990s by Mark S. Barton and Mark Schlichter (). Its fast reactivity and relative moisture insensitivity made it useful for coatings on large surface area projects, such as secondary containment, manhole and tunnel coatings, tank liners, and truck bed liners. Excellent adhesion to concrete and steel is obtained with the proper primer and surface treatment. They can also be used for spray molding and armor. Some polyureas reach strengths of 40 MPa (6000 psi) tensile and over 500% elongation making it a tough coating. The quick cure time allows many coats to be built up quickly.
In 2014, a polyurea elastomer-based material was shown to be self-healing, melding together after being cut in half. The material also includes inexpensive commercially available compounds. The elastomer molecules were tweaked, making the bonds between them longer. The resulting molecules are easier to pull apart from one another and better able to rebond at room temperature with almost the same strength. The rebonding can be repeated. Stretchy, self-healing paints and other coatings recently took a step closer to common use, thanks to research being conducted at the University of Illinois. Scientists there have used "off-the-shelf" components to create a polymer that melds back together after being cut in half, without the addition of other chemicals.
Polyurea has become a preferred long term solution for narrowboats. The traditional coating with bitumen, known as "blacking" is being replaced with the practice of polyurea coatings. The clearest advantage is that it is not necessary to reapply a coat every 3–4 years. It is thought that polyurea coatings last 25–30 years.
Commercial trademarks for Polyurea include Line-X, GLS 100R, and Pentens SPU-1000, to name a few. There are several formulations of Polyurea. The Polyurea Development Association is a trade association that represents the interests of polyurea coating manufacturers.
References
{{Reflist Polyamides Synthetic resins Plastics PolyurethanesSee also
* Polyaspartic