Photosynthesis on:
[Wikipedia]
[Google]
[Amazon]

 Photosynthesis is a process used by
Photosynthesis is a process used by
 Most photosynthetic organisms are
Most photosynthetic organisms are
 In photosynthetic bacteria, the proteins that gather light for photosynthesis are embedded in
In photosynthetic bacteria, the proteins that gather light for photosynthesis are embedded in
 In the light-dependent reactions, one molecule of the
In the light-dependent reactions, one molecule of the
 In plants,
In plants,
 Carbon fixation produces the three-carbon sugar intermediate, which is then converted into the final carbohydrate products. The simple carbon sugars produced by photosynthesis are then used to form other organic compounds, such as the building material
Carbon fixation produces the three-carbon sugar intermediate, which is then converted into the final carbohydrate products. The simple carbon sugars produced by photosynthesis are then used to form other organic compounds, such as the building material
 In hot and dry conditions, plants close their stomata to prevent water loss. Under these conditions, will decrease and oxygen gas, produced by the light reactions of photosynthesis, will increase, causing an increase of photorespiration by the oxygenase activity of ribulose-1,5-bisphosphate carboxylase/oxygenase and decrease in carbon fixation. Some plants have evolved mechanisms to increase the concentration in the leaves under these conditions.
Plants that use the C4 carbon fixation process chemically fix carbon dioxide in the cells of the mesophyll by adding it to the three-carbon molecule
In hot and dry conditions, plants close their stomata to prevent water loss. Under these conditions, will decrease and oxygen gas, produced by the light reactions of photosynthesis, will increase, causing an increase of photorespiration by the oxygenase activity of ribulose-1,5-bisphosphate carboxylase/oxygenase and decrease in carbon fixation. Some plants have evolved mechanisms to increase the concentration in the leaves under these conditions.
Plants that use the C4 carbon fixation process chemically fix carbon dioxide in the cells of the mesophyll by adding it to the three-carbon molecule
 Several groups of animals have formed symbiotic relationships with photosynthetic algae. These are most common in
Several groups of animals have formed symbiotic relationships with photosynthetic algae. These are most common in
 Jan Baptist van Helmont, Jan van Helmont began the research of the process in the mid-17th century when he carefully measured the mass of the soil used by a plant and the mass of the plant as it grew. After noticing that the soil mass changed very little, he hypothesized that the mass of the growing plant must come from the water, the only substance he added to the potted plant. His hypothesis was partially accurate – much of the gained mass comes from carbon dioxide as well as water. However, this was a signaling point to the idea that the bulk of a plant's biomass comes from the inputs of photosynthesis, not the soil itself.
Joseph Priestley, a chemist and minister, discovered that when he isolated a volume of air under an inverted jar and burned a candle in it (which gave off CO2), the candle would burn out very quickly, much before it ran out of wax. He further discovered that a mouse could similarly Breathing, "injure" air. He then showed that the air that had been "injured" by the candle and the mouse could be restored by a plant.
In 1779,
Jan Baptist van Helmont, Jan van Helmont began the research of the process in the mid-17th century when he carefully measured the mass of the soil used by a plant and the mass of the plant as it grew. After noticing that the soil mass changed very little, he hypothesized that the mass of the growing plant must come from the water, the only substance he added to the potted plant. His hypothesis was partially accurate – much of the gained mass comes from carbon dioxide as well as water. However, this was a signaling point to the idea that the bulk of a plant's biomass comes from the inputs of photosynthesis, not the soil itself.
Joseph Priestley, a chemist and minister, discovered that when he isolated a volume of air under an inverted jar and burned a candle in it (which gave off CO2), the candle would burn out very quickly, much before it ran out of wax. He further discovered that a mouse could similarly Breathing, "injure" air. He then showed that the air that had been "injured" by the candle and the mouse could be restored by a plant.
In 1779,
 Melvin Calvin and Andrew Benson, along with James Bassham, elucidated the path of carbon assimilation (the photosynthetic carbon reduction cycle) in plants. The carbon reduction cycle is known as the Calvin cycle, but many scientists refer to it as the Calvin-Benson, Benson-Calvin, or even Calvin-Benson-Bassham (or CBB) Cycle.
Nobel Prize-winning scientist Rudolph A. Marcus was later able to discover the function and significance of the electron transport chain.
Otto Heinrich Warburg and Dean Burk discovered the I-quantum photosynthesis reaction that splits CO2, activated by the respiration.
In 1950, first experimental evidence for the existence of
Melvin Calvin and Andrew Benson, along with James Bassham, elucidated the path of carbon assimilation (the photosynthetic carbon reduction cycle) in plants. The carbon reduction cycle is known as the Calvin cycle, but many scientists refer to it as the Calvin-Benson, Benson-Calvin, or even Calvin-Benson-Bassham (or CBB) Cycle.
Nobel Prize-winning scientist Rudolph A. Marcus was later able to discover the function and significance of the electron transport chain.
Otto Heinrich Warburg and Dean Burk discovered the I-quantum photosynthesis reaction that splits CO2, activated by the respiration.
In 1950, first experimental evidence for the existence of
 There are three main factors affecting photosynthesis and several corollary factors. The three main are:
* Light irradiance and
There are three main factors affecting photosynthesis and several corollary factors. The three main are:
* Light irradiance and
 The process of photosynthesis provides the main input of free energy into the biosphere, and is one of four main ways in which radiation is important for plant life.
The radiation climate within plant communities is extremely variable, in both time and space.
In the early 20th century, Frederick Blackman and Gabrielle Matthaei investigated the effects of light intensity (irradiance) and temperature on the rate of carbon assimilation.
* At constant temperature, the rate of carbon assimilation varies with irradiance, increasing as the irradiance increases, but reaching a plateau at higher irradiance.
* At low irradiance, increasing the temperature has little influence on the rate of carbon assimilation. At constant high irradiance, the rate of carbon assimilation increases as the temperature is increased.
These two experiments illustrate several important points: First, it is known that, in general, photochemical reactions are not affected by temperature. However, these experiments clearly show that temperature affects the rate of carbon assimilation, so there must be two sets of reactions in the full process of carbon assimilation. These are the light-dependent 'photochemical' temperature-independent stage, and the light-independent, temperature-dependent stage. Second, Blackman's experiments illustrate the concept of limiting factors. Another limiting factor is the wavelength of light. Cyanobacteria, which reside several meters underwater, cannot receive the correct wavelengths required to cause photoinduced charge separation in conventional photosynthetic pigments. To combat this problem, a series of proteins with different pigments surround the reaction center. This unit is called a phycobilisome.
The process of photosynthesis provides the main input of free energy into the biosphere, and is one of four main ways in which radiation is important for plant life.
The radiation climate within plant communities is extremely variable, in both time and space.
In the early 20th century, Frederick Blackman and Gabrielle Matthaei investigated the effects of light intensity (irradiance) and temperature on the rate of carbon assimilation.
* At constant temperature, the rate of carbon assimilation varies with irradiance, increasing as the irradiance increases, but reaching a plateau at higher irradiance.
* At low irradiance, increasing the temperature has little influence on the rate of carbon assimilation. At constant high irradiance, the rate of carbon assimilation increases as the temperature is increased.
These two experiments illustrate several important points: First, it is known that, in general, photochemical reactions are not affected by temperature. However, these experiments clearly show that temperature affects the rate of carbon assimilation, so there must be two sets of reactions in the full process of carbon assimilation. These are the light-dependent 'photochemical' temperature-independent stage, and the light-independent, temperature-dependent stage. Second, Blackman's experiments illustrate the concept of limiting factors. Another limiting factor is the wavelength of light. Cyanobacteria, which reside several meters underwater, cannot receive the correct wavelengths required to cause photoinduced charge separation in conventional photosynthetic pigments. To combat this problem, a series of proteins with different pigments surround the reaction center. This unit is called a phycobilisome.
 As carbon dioxide concentrations rise, the rate at which sugars are made by the light-independent reactions increases until limited by other factors.
As carbon dioxide concentrations rise, the rate at which sugars are made by the light-independent reactions increases until limited by other factors.
A collection of photosynthesis pages for all levels from a renowned expert (Govindjee)
* [https://web.archive.org/web/20090428090455/http://scienceaid.co.uk/biology/biochemistry/photosynthesis.html Science Aid: Photosynthesis] Article appropriate for high school science
Metabolism, Cellular Respiration and Photosynthesis – The Virtual Library of Biochemistry and Cell Biology
* [http://www.life.uiuc.edu/govindjee/photosynBook.html Overall Energetics of Photosynthesis]
The source of oxygen produced by photosynthesis
Interactive animation, a textbook tutorial *
Photosynthesis – Light Dependent & Light Independent Stages
Khan Academy, video introduction
{{Authority control Photosynthesis, Agronomy Biological processes Botany Cellular respiration Ecosystems Metabolism Plant nutrition Plant physiology Quantum biology
 Photosynthesis is a process used by
Photosynthesis is a process used by plant
Plants are predominantly photosynthetic eukaryotes of the kingdom Plantae. Historically, the plant kingdom encompassed all living things that were not animals, and included algae and fungi; however, all current definitions of Plantae exclu ...
s and other organisms to convert light energy
Light or visible light is electromagnetic radiation that can be perceived by the human eye. Visible light is usually defined as having wavelengths in the range of 400–700 nanometres (nm), corresponding to frequencies of 750–420 terahe ...
into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored in carbohydrate
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may or m ...
molecules, such as sugars and starches, which are synthesized from carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
and water
Water (chemical formula ) is an Inorganic compound, inorganic, transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living ...
– hence the name ''photosynthesis'', from the Greek
Greek may refer to:
Greece
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group.
*Greek language, a branch of the Indo-European language family.
**Proto-Greek language, the assumed last common ancestor ...
''phōs'' (), "light", and ''synthesis'' (), "putting together". Most plant
Plants are predominantly photosynthetic eukaryotes of the kingdom Plantae. Historically, the plant kingdom encompassed all living things that were not animals, and included algae and fungi; however, all current definitions of Plantae exclu ...
s, algae, and cyanobacteria perform photosynthesis; such organisms are called photoautotrophs. Photosynthesis is largely responsible for producing and maintaining the oxygen content of the Earth's atmosphere, and supplies most of the energy necessary for life on Earth.
Although photosynthesis is performed differently by different species, the process always begins when energy from light is absorbed by protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, res ...
s called reaction center
A photosynthetic reaction center is a complex of several proteins, pigments and other co-factors that together execute the primary energy conversion reactions of photosynthesis. Molecular excitations, either originating directly from sunlight or ...
s that contain green chlorophyll (and other colored) pigments/chromophores. In plants, these proteins are held inside organelles called chloroplasts, which are most abundant in leaf cells, while in bacteria they are embedded in the plasma membrane. In these light-dependent reactions, some energy is used to strip electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
s from suitable substances, such as water, producing oxygen gas. The hydrogen freed by the splitting of water is used in the creation of two further compounds that serve as short-term stores of energy, enabling its transfer to drive other reactions: these compounds are reduced nicotinamide adenine dinucleotide phosphate
Nicotinamide adenine dinucleotide phosphate, abbreviated NADP or, in older notation, TPN (triphosphopyridine nucleotide), is a cofactor used in anabolic reactions, such as the Calvin cycle and lipid and nucleic acid syntheses, which require NAD ...
(NADPH) and adenosine triphosphate (ATP), the "energy currency" of cells.
In plants, algae and cyanobacteria, sugars are synthesized by a subsequent sequence of reactions called the Calvin cycle. In the Calvin cycle, atmospheric carbon dioxide is incorporated into already existing organic carbon compounds, such as ribulose bisphosphate (RuBP). Using the ATP and NADPH produced by the light-dependent reactions, the resulting compounds are then reduced and removed to form further carbohydrates, such as glucose
Glucose is a simple sugar with the molecular formula . Glucose is overall the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is mainly made by plants and most algae during photosynthesis from water and carbon dioxide, u ...
. In other bacteria, different mechanisms such as the reverse Krebs cycle are used to achieve the same end.
The first photosynthetic organisms probably evolved early in the evolutionary history of life
The history of life on Earth traces the processes by which living and fossil organisms evolved, from the earliest emergence of life to present day. Earth formed about 4.5 billion years ago (abbreviated as ''Ga'', for ''gigaannum'') and evide ...
and most likely used reducing agents such as hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
or hydrogen sulfide, rather than water, as sources of electrons. Cyanobacteria appeared later; the excess oxygen they produced contributed directly to the oxygenation of the Earth, which rendered the evolution of complex life possible. Today, the average rate of energy capture by photosynthesis globally is approximately 130 terawatts, which is about eight times the current power consumption of human civilization. Photosynthetic organisms also convert around 100–115 billion tons (91–104 Pg petagrams, or billion metric tons), of carbon into biomass per year. That plants receive some energy from light – in addition to air, soil, and water – was first discovered in 1779 by Jan Ingenhousz
Jan (or John) Ingenhousz or Ingen-Housz FRS (8 December 1730 – 7 September 1799) was a Dutch-born British physiologist, biologist and chemist.
He is best known for discovering photosynthesis by showing that light is essential to the process ...
.
Photosynthesis is vital for climate processes, as it captures carbon dioxide from the air and then binds carbon in plants and further in soils and harvested products. Cereals alone are estimated to bind 3,825 Tg (teragrams) or 3.825 Pg (petagrams) of carbon dioxide every year, i.e. 3.825 billion metric tons.
Overview
photoautotroph Photoautotrophs are organisms that use light energy and inorganic carbon to produce organic materials. Eukaryotic photoautotrophs absorb energy through the chlorophyll molecules in their chloroplasts while prokaryotic photoautotrophs use chlorophyl ...
s, which means that they are able to synthesize food directly from carbon dioxide and water using energy from light. However, not all organisms use carbon dioxide as a source of carbon atoms to carry out photosynthesis; photoheterotroph
Photoheterotrophs ('' Gk'': ''photo'' = light, ''hetero'' = (an)other, ''troph'' = nourishment) are heterotrophic phototrophs – that is, they are organisms that use light for energy, but cannot use carbon dioxide as their sole carbon source. Con ...
s use organic compounds, rather than carbon dioxide, as a source of carbon. In plants, algae, and cyanobacteria, photosynthesis releases oxygen. This oxygenic photosynthesis is by far the most common type of photosynthesis used by living organisms. Although there are some differences between oxygenic photosynthesis in plants
Plants are predominantly photosynthetic eukaryotes of the kingdom Plantae. Historically, the plant kingdom encompassed all living things that were not animals, and included algae and fungi; however, all current definitions of Plantae exclude ...
, algae, and cyanobacteria, the overall process is quite similar in these organisms. There are also many varieties of anoxygenic photosynthesis
Bacterial anoxygenic photosynthesis differs from the better known oxygenic photosynthesis in plants by the reductant used (e.g. hydrogen sulfide instead of water) and the byproduct generated (e.g. elemental sulfur instead of molecular oxygen).
Ba ...
, used mostly by bacteria, which consume carbon dioxide but do not release oxygen.
Carbon dioxide is converted into sugars in a process called carbon fixation; photosynthesis captures energy from sunlight to convert carbon dioxide into carbohydrate
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may or m ...
s. Carbon fixation is an endothermic redox
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a ...
reaction. In general outline, photosynthesis is the opposite of cellular respiration: while photosynthesis is a process of reduction of carbon dioxide to carbohydrates, cellular respiration is the oxidation of carbohydrates or other nutrients to carbon dioxide. Nutrients used in cellular respiration include carbohydrates, amino acids and fatty acids. These nutrients are oxidized to produce carbon dioxide and water, and to release chemical energy to drive the organism's metabolism
Metabolism (, from el, μεταβολή ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run c ...
. Photosynthesis and cellular respiration are distinct processes, as they take place through different sequences of chemical reactions and in different cellular compartment
Cellular compartments in cell biology comprise all of the closed parts within the cytosol of a eukaryotic cell, usually surrounded by a single or double lipid layer membrane. These compartments are often, but not always, defined as membrane-bo ...
s.
The general equation for photosynthesis as first proposed by Cornelis van Niel is:
: + + → + +
Since water is used as the electron donor in oxygenic photosynthesis, the equation for this process is:
: + + → + +
This equation emphasizes that water is both a reactant in the light-dependent reaction
Light-dependent reactions is jargon for certain photochemical reactions that are involved in photosynthesis, the main process by which plants acquire energy. There are two light dependent reactions, the first occurs at photosystem II (PSII) and ...
and a product of the light-independent reaction
The Calvin cycle, light-independent reactions, bio synthetic phase, dark reactions, or photosynthetic carbon reduction (PCR) cycle of photosynthesis is a series of chemical reactions that convert carbon dioxide and hydrogen-carrier compounds into ...
, but canceling ''n'' water molecules from each side gives the net equation:
: + + → +
Other processes substitute other compounds (such as arsenite
In chemistry, an arsenite is a chemical compound containing an arsenic oxyanion where arsenic has oxidation state +3. Note that in fields that commonly deal with groundwater chemistry, arsenite is used generically to identify soluble AsIII anions. ...
) for water in the electron-supply role; for example some microbes use sunlight to oxidize arsenite to arsenate
The arsenate ion is .
An arsenate (compound) is any compound that contains this ion. Arsenates are salts or esters of arsenic acid.
The arsenic atom in arsenate has a valency of 5 and is also known as pentavalent arsenic or As(V).
Arsenate res ...
: The equation for this reaction is:
: + + → + (used to build other compounds in subsequent reactions)
Photosynthesis occurs in two stages. In the first stage, ''light-dependent reactions'' or ''light reactions'' capture the energy of light and use it to make the hydrogen carrier NADPH and the energy-storage molecule ATP. During the second stage, the ''light-independent reactions'' use these products to capture and reduce carbon dioxide.
Most organisms that use oxygenic photosynthesis use visible light for the light-dependent reactions, although at least three use shortwave infrared
Infrared (IR), sometimes called infrared light, is electromagnetic radiation (EMR) with wavelengths longer than those of visible light. It is therefore invisible to the human eye. IR is generally understood to encompass wavelengths from around ...
or, more specifically, far-red radiation.
Some organisms employ even more radical variants of photosynthesis. Some archaea use a simpler method that employs a pigment similar to those used for vision in animals. The bacteriorhodopsin changes its configuration in response to sunlight, acting as a proton pump. This produces a proton gradient more directly, which is then converted to chemical energy. The process does not involve carbon dioxide fixation and does not release oxygen, and seems to have evolved separately from the more common types of photosynthesis.
Photosynthetic membranes and organelles
cell membrane
The cell membrane (also known as the plasma membrane (PM) or cytoplasmic membrane, and historically referred to as the plasmalemma) is a biological membrane that separates and protects the interior of all cells from the outside environment ( ...
s. In its simplest form, this involves the membrane surrounding the cell itself. However, the membrane may be tightly folded into cylindrical sheets called thylakoids, or bunched up into round vesicles called ''intracytoplasmic membranes''. These structures can fill most of the interior of a cell, giving the membrane a very large surface area and therefore increasing the amount of light that the bacteria can absorb.
In plants and algae, photosynthesis takes place in organelles called chloroplasts. A typical plant cell contains about 10 to 100 chloroplasts. The chloroplast is enclosed by a membrane. This membrane is composed of a phospholipid inner membrane, a phospholipid outer membrane, and an intermembrane space. Enclosed by the membrane is an aqueous fluid called the stroma. Embedded within the stroma are stacks of thylakoids (grana), which are the site of photosynthesis. The thylakoids appear as flattened disks. The thylakoid itself is enclosed by the thylakoid membrane, and within the enclosed volume is a lumen or thylakoid space. Embedded in the thylakoid membrane are integral and peripheral membrane protein complexes of the photosynthetic system.
Plants absorb light primarily using the pigment
A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compou ...
chlorophyll. The green part of the light spectrum is not absorbed but is reflected which is the reason that most plants have a green color. Besides chlorophyll, plants also use pigments such as carotenes and xanthophyll
Xanthophylls (originally phylloxanthins) are yellow pigments that occur widely in nature and form one of two major divisions of the carotenoid group; the other division is formed by the carotenes. The name is from Greek (, "yellow") and (, "lea ...
s. Algae also use chlorophyll, but various other pigments are present, such as phycocyanin
Phycocyanin is a pigment-protein complex from the light-harvesting phycobiliprotein family, along with allophycocyanin and phycoerythrin. It is an accessory pigment to chlorophyll. All phycobiliproteins are water-soluble, so they cannot exist w ...
, carotenes, and xanthophyll
Xanthophylls (originally phylloxanthins) are yellow pigments that occur widely in nature and form one of two major divisions of the carotenoid group; the other division is formed by the carotenes. The name is from Greek (, "yellow") and (, "lea ...
s in green algae, phycoerythrin in red algae (rhodophytes) and fucoxanthin in brown algae
Brown algae (singular: alga), comprising the class Phaeophyceae, are a large group of multicellular algae, including many seaweeds located in colder waters within the Northern Hemisphere. Brown algae are the major seaweeds of the temperate and p ...
and diatoms
A diatom (New Latin, Neo-Latin ''diatoma''), "a cutting through, a severance", from el, διάτομος, diátomos, "cut in half, divided equally" from el, διατέμνω, diatémno, "to cut in twain". is any member of a large group com ...
resulting in a wide variety of colors.
These pigments are embedded in plants and algae in complexes called antenna proteins. In such proteins, the pigments are arranged to work together. Such a combination of proteins is also called a light-harvesting complex
A light-harvesting complex consists of a number of chromophores which are complex subunit proteins that may be part of a larger super complex of a photosystem, the functional unit in photosynthesis. It is used by plants and photosynthetic bacteri ...
.
Although all cells in the green parts of a plant have chloroplasts, the majority of those are found in specially adapted structures called leaves. Certain species adapted to conditions of strong sunlight and aridity, such as many ''Euphorbia
''Euphorbia'' is a very large and diverse genus of flowering plants, commonly called spurge, in the family Euphorbiaceae. "Euphorbia" is sometimes used in ordinary English to collectively refer to all members of Euphorbiaceae (in deference to t ...
'' and cactus
A cactus (, or less commonly, cactus) is a member of the plant family Cactaceae, a family comprising about 127 genera with some 1750 known species of the order Caryophyllales. The word ''cactus'' derives, through Latin, from the Ancient Gree ...
species, have their main photosynthetic organs in their stems. The cells in the interior tissues of a leaf, called the mesophyll, can contain between 450,000 and 800,000 chloroplasts for every square millimeter of leaf. The surface of the leaf is coated with a water-resistant wax
Waxes are a diverse class of organic compounds that are lipophilic, malleable solids near ambient temperatures. They include higher alkanes and lipids, typically with melting points above about 40 °C (104 °F), melting to giv ...
y cuticle that protects the leaf from excessive evaporation of water and decreases the absorption of ultraviolet
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation ...
or blue
Blue is one of the three primary colours in the RYB colour model (traditional colour theory), as well as in the RGB (additive) colour model. It lies between violet and cyan on the spectrum of visible light. The eye perceives blue when ...
light
Light or visible light is electromagnetic radiation that can be perceived by the human eye. Visible light is usually defined as having wavelengths in the range of 400–700 nanometres (nm), corresponding to frequencies of 750–420 te ...
to minimize heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is ...
ing. The transparent epidermis layer allows light to pass through to the palisade mesophyll cells where most of the photosynthesis takes place.
Light-dependent reactions
pigment
A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compou ...
chlorophyll absorbs one photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
and loses one electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
. This electron is taken up by a modified form of chlorophyll called pheophytin
Pheophytin or phaeophytin is a chemical compound that serves as the first electron carrier intermediate in the electron transfer pathway of Photosystem II (PS II) in plants, and the type II photosynthetic reaction center (RC P870) found in pur ...
, which passes the electron to a quinone molecule, starting the flow of electrons down an electron transport chain that leads to the ultimate reduction of NADP
Nicotinamide adenine dinucleotide phosphate, abbreviated NADP or, in older notation, TPN (triphosphopyridine nucleotide), is a cofactor used in anabolic reactions, such as the Calvin cycle and lipid and nucleic acid syntheses, which require N ...
to NADPH. In addition, this creates a proton gradient (energy gradient) across the chloroplast membrane
Chloroplasts contain several important membranes, vital for their function. Like mitochondria, chloroplasts have a double-membrane envelope, called the chloroplast envelope, but unlike mitochondria, chloroplasts also have internal membrane struc ...
, which is used by ATP synthase in the synthesis of ATP. The chlorophyll molecule ultimately regains the electron it lost when a water molecule is split in a process called photolysis, which releases oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as ...
.
The overall equation for the light-dependent reactions under the conditions of non-cyclic electron flow in green plants is:
Not all wavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, t ...
s of light can support photosynthesis. The photosynthetic action spectrum depends on the type of accessory pigment Accessory pigments are light-absorbing compounds, found in photosynthetic organisms, that work in conjunction with chlorophyll ''a''. They include other forms of this pigment, such as chlorophyll ''b'' in green algal and higher plant antennae, w ...
s present. For example, in green plants, the action spectrum resembles the absorption spectrum for chlorophylls and carotenoids with absorption peaks in violet-blue and red light. In red algae, the action spectrum is blue-green light, which allows these algae to use the blue end of the spectrum to grow in the deeper waters that filter out the longer wavelengths (red light) used by above-ground green plants. The non-absorbed part of the light spectrum is what gives photosynthetic organisms their color (e.g., green plants, red algae, purple bacteria) and is the least effective for photosynthesis in the respective organisms.
Z scheme
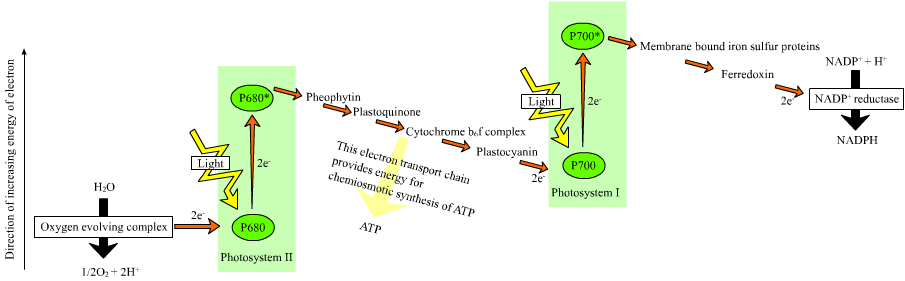
 In plants,
In plants, light-dependent reaction
Light-dependent reactions is jargon for certain photochemical reactions that are involved in photosynthesis, the main process by which plants acquire energy. There are two light dependent reactions, the first occurs at photosystem II (PSII) and ...
s occur in the thylakoid membrane
Thylakoids are membrane-bound compartments inside chloroplasts and cyanobacteria. They are the site of the light-dependent reactions of photosynthesis. Thylakoids consist of a thylakoid membrane surrounding a thylakoid lumen. Chloroplast thyla ...
s of the chloroplasts where they drive the synthesis of ATP and NADPH. The light-dependent reactions are of two forms: cyclic and non-cyclic.
In the non-cyclic reaction, the photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
s are captured in the light-harvesting antenna complexes of photosystem II by chlorophyll and other accessory pigments (see diagram at right). The absorption of a photon by the antenna complex loosens an electron by a process called photoinduced charge separation
Photoinduced charge separation is the process of an electron in an atom or molecule, being excited to a higher energy level by the absorption of a photon and then leaving the atom or molecule to a nearby electron acceptor.
Rutherford model
An atom ...
. The antenna system is at the core of the chlorophyll molecule of the photosystem II reaction center. That loosened electron is taken up by the primary electron-acceptor molecule, pheophytin. As the electrons are shuttled through an electron transport chain (the so-called ''Z-scheme'' shown in the diagram), a chemiosmotic potential is generated by pumping proton cations (H+) across the membrane and into the thylakoid space. An ATP synthase enzyme uses that chemiosmotic potential to make ATP during photophosphorylation In the process of photosynthesis, the phosphorylation of ADP to form ATP using the energy of sunlight is called photophosphorylation. Cyclic photophosphorylation occurs in both aerobic and anaerobic conditions, driven by the main primary source of ...
, whereas NADPH is a product of the terminal redox
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a ...
reaction in the ''Z-scheme''. The electron enters a chlorophyll molecule in Photosystem I
Photosystem I (PSI, or plastocyanin–ferredoxin oxidoreductase) is one of two photosystems in the photosynthetic light reactions of algae, plants, and cyanobacteria. Photosystem I is an integral membrane protein complex that us ...
. There it is further excited by the light absorbed by that photosystem. The electron is then passed along a chain of electron acceptors to which it transfers some of its energy. The energy delivered to the electron acceptors is used to move hydrogen ions across the thylakoid membrane into the lumen. The electron is eventually used to reduce the co-enzyme NADP with a H+ to NADPH (which has functions in the light-independent reaction); at that point, the path of that electron ends.
The cyclic reaction is similar to that of the non-cyclic but differs in that it generates only ATP, and no reduced NADP (NADPH) is created. The cyclic reaction takes place only at photosystem I. Once the electron is displaced from the photosystem, the electron is passed down the electron acceptor molecules and returns to photosystem I, from where it was emitted, hence the name ''cyclic reaction''.
Water photolysis
Linear electron transport through a photosystem will leave the reaction center of that photosystem oxidized. Elevating another electron will first require re-reduction of the reaction center. The excited electrons lost from the reaction center (P700) ofphotosystem I
Photosystem I (PSI, or plastocyanin–ferredoxin oxidoreductase) is one of two photosystems in the photosynthetic light reactions of algae, plants, and cyanobacteria. Photosystem I is an integral membrane protein complex that us ...
are replaced by transfer from plastocyanin
Plastocyanin is a copper-containing protein that mediates electron-transfer. It is found in a variety of plants, where it participates in photosynthesis. The protein is a prototype of the blue copper proteins, a family of intensely blue-colored ...
, whose electrons come from electron transport through photosystem II. Photosystem II, as the first step of the ''Z-scheme'', requires an external source of electrons to reduce its oxidized chlorophyll ''a'' reaction center. The source of electrons for photosynthesis in green plants and cyanobacteria is water. Two water molecules are oxidized by the energy of four successive charge-separation reactions of photosystem II to yield a molecule of diatomic oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as ...
and four hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
ions. The electrons yielded are transferred to a redox-active tyrosine
-Tyrosine or tyrosine (symbol Tyr or Y) or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. The word "tyrosine" is from the G ...
residue that is oxidized by the energy of P680. This resets the ability of P680 to absorb another photon and release another photo-dissociated electron. The oxidation of water is catalyzed in photosystem II by a redox-active structure that contains four manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
ions and a calcium ion; this oxygen-evolving complex
The oxygen-evolving complex (OEC), also known as the water-splitting complex, is the portion of photosystem II where photo-oxidation of water occurs during the light reactions of photosynthesis. The OEC is surrounded by four core protein subunits ...
binds two water molecules and contains the four oxidizing equivalents that are used to drive the water-oxidizing reaction (Kok's S-state diagrams). The hydrogen ions are released in the thylakoid lumen and therefore contribute to the transmembrane chemiosmotic potential that leads to ATP synthesis. Oxygen is a waste product of light-dependent reactions, but the majority of organisms on Earth use oxygen and its energy for cellular respiration, including photosynthetic organisms.
Light-independent reactions
Calvin cycle
In the light-independent (or "dark") reactions, theenzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
RuBisCO
Ribulose-1,5-bisphosphate carboxylase-oxygenase, commonly known by the abbreviations RuBisCo, rubisco, RuBPCase, or RuBPco, is an enzyme () involved in the first major step of carbon fixation, a process by which atmospheric carbon dioxide is con ...
captures CO2 from the atmosphere and, in a process called the Calvin cycle, uses the newly formed NADPH and releases three-carbon sugars, which are later combined to form sucrose and starch. The overall equation for the light-independent reactions in green plants is
cellulose
Cellulose is an organic compound with the formula , a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked D-glucose units. Cellulose is an important structural component of the primary cell w ...
, the precursors for lipid
Lipids are a broad group of naturally-occurring molecules which includes fats, waxes, sterols, fat-soluble vitamins (such as vitamins A, D, E and K), monoglycerides, diglycerides, phospholipids, and others. The functions of lipids includ ...
and amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha a ...
biosynthesis, or as a fuel in cellular respiration. The latter occurs not only in plants but also in animal
Animals are multicellular, eukaryotic organisms in the Kingdom (biology), biological kingdom Animalia. With few exceptions, animals Heterotroph, consume organic material, Cellular respiration#Aerobic respiration, breathe oxygen, are Motilit ...
s when the carbon and energy from plants is passed through a food chain
A food chain is a linear network of links in a food web starting from producer organisms (such as grass or algae which produce their own food via photosynthesis) and ending at an apex predator species (like grizzly bears or killer whales), de ...
.
The fixation or reduction of carbon dioxide is a process in which carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
combines with a five-carbon sugar, ribulose 1,5-bisphosphate
Ribulose 1,5-bisphosphate (RuBP) is an organic substance that is involved in photosynthesis, notably as the principal acceptor in plants. It is a colourless anion, a double phosphate ester of the ketopentose (ketone-containing sugar with five ca ...
, to yield two molecules of a three-carbon compound, glycerate 3-phosphate
3-Phosphoglyceric acid (3PG, 3-PGA, or PGA) is the conjugate acid of 3-phosphoglycerate or glycerate 3-phosphate (GP or G3P). This glycerate is a biochemically significant metabolic intermediate in both glycolysis and the Calvin-Benson cycle. The ...
, also known as 3-phosphoglycerate. Glycerate 3-phosphate, in the presence of ATP and NADPH produced during the light-dependent stages, is reduced to glyceraldehyde 3-phosphate
Glyceraldehyde 3-phosphate, also known as triose phosphate or 3-phosphoglyceraldehyde and abbreviated as G3P, GA3P, GADP, GAP, TP, GALP or PGAL, is a metabolite that occurs as an intermediate in several central pathways of all organisms.Nelson, D ...
. This product is also referred to as 3-phosphoglyceraldehyde ( PGAL) or, more generically, as triose phosphate. Most (5 out of 6 molecules) of the glyceraldehyde 3-phosphate produced are used to regenerate ribulose 1,5-bisphosphate so the process can continue. The triose phosphates not thus "recycled" often condense to form hexose
In chemistry, a hexose is a monosaccharide (simple sugar) with six carbon atoms. The chemical formula for all hexoses is C6H12O6, and their molecular weight is 180.156 g/mol.
Hexoses exist in two forms, open-chain or cyclic, that easily convert ...
phosphates, which ultimately yield sucrose, starch and cellulose
Cellulose is an organic compound with the formula , a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked D-glucose units. Cellulose is an important structural component of the primary cell w ...
. The sugars produced during carbon metabolism
Metabolism (, from el, μεταβολή ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run c ...
yield carbon skeletons that can be used for other metabolic reactions like the production of amino acids and lipids.
Carbon concentrating mechanisms
On land
phosphoenolpyruvate
Phosphoenolpyruvate (2-phosphoenolpyruvate, PEP) is the ester derived from the enol of pyruvate and phosphate. It exists as an anion. PEP is an important intermediate in biochemistry. It has the highest-energy phosphate bond found (−61.9 kJ/m ...
(PEP), a reaction catalyzed by an enzyme called PEP carboxylase, creating the four-carbon organic acid oxaloacetic acid
Oxaloacetic acid (also known as oxalacetic acid or OAA) is a crystalline organic compound with the chemical formula HO2CC(O)CH2CO2H. Oxaloacetic acid, in the form of its conjugate base oxaloacetate, is a metabolic intermediate in many processes ...
. Oxaloacetic acid or malate
Malic acid is an organic compound with the molecular formula . It is a dicarboxylic acid that is made by all living organisms, contributes to the sour taste of fruits, and is used as a food additive. Malic acid has two stereoisomeric forms (L ...
synthesized by this process is then translocated to specialized bundle sheath
A vascular bundle is a part of the transport system in vascular plants. The transport itself happens in the stem, which exists in two forms: xylem and phloem. Both these tissues are present in a vascular bundle, which in addition will includ ...
cells where the enzyme RuBisCO
Ribulose-1,5-bisphosphate carboxylase-oxygenase, commonly known by the abbreviations RuBisCo, rubisco, RuBPCase, or RuBPco, is an enzyme () involved in the first major step of carbon fixation, a process by which atmospheric carbon dioxide is con ...
and other Calvin cycle enzymes are located, and where released by decarboxylation of the four-carbon acids is then fixed by RuBisCO activity to the three-carbon 3-phosphoglyceric acids. The physical separation of RuBisCO from the oxygen-generating light reactions reduces photorespiration and increases fixation and, thus, the photosynthetic capacity of the leaf. plants can produce more sugar than plants in conditions of high light and temperature. Many important crop plants are plants, including maize, sorghum, sugarcane, and millet. Plants that do not use PEP-carboxylase in carbon fixation are called C3 plants because the primary carboxylation reaction, catalyzed by RuBisCO, produces the three-carbon 3-phosphoglyceric acids directly in the Calvin-Benson cycle. Over 90% of plants use carbon fixation, compared to 3% that use carbon fixation; however, the evolution of in over 60 plant lineages makes it a striking example of convergent evolution
Convergent evolution is the independent evolution of similar features in species of different periods or epochs in time. Convergent evolution creates analogous structures that have similar form or function but were not present in the last com ...
. C2 photosynthesis, which involves carbon-concentration by selective breakdown of photorespiratory glycine, is both an evolutionary precursor to and a useful CCM in its own right.
Xerophytes, such as cacti and most succulents
In botany, succulent plants, also known as succulents, are plants with parts that are thickened, fleshy, and engorged, usually to retain water in arid climates or soil conditions. The word ''succulent'' comes from the Latin word ''sucus'', meani ...
, also use PEP carboxylase to capture carbon dioxide in a process called Crassulacean acid metabolism
Crassulacean acid metabolism, also known as CAM photosynthesis, is a carbon fixation pathway that evolved in some plants as an adaptation to arid conditions that allows a plant to photosynthesize during the day, but only exchange gases at night. ...
(CAM). In contrast to metabolism, which ''spatially'' separates the fixation to PEP from the Calvin cycle, CAM ''temporally'' separates these two processes. CAM plants have a different leaf anatomy from plants, and fix the at night, when their stomata are open. CAM plants store the mostly in the form of malic acid via carboxylation of phosphoenolpyruvate
Phosphoenolpyruvate (2-phosphoenolpyruvate, PEP) is the ester derived from the enol of pyruvate and phosphate. It exists as an anion. PEP is an important intermediate in biochemistry. It has the highest-energy phosphate bond found (−61.9 kJ/m ...
to oxaloacetate, which is then reduced to malate. Decarboxylation of malate during the day releases inside the leaves, thus allowing carbon fixation to 3-phosphoglycerate by RuBisCO. CAM is used by 16,000 species of plants.
Calcium oxalate accumulating plants, such as '' Amaranthus hybridus'' and ''Colobanthus quitensis
''Colobanthus quitensis'', the Antarctic pearlwort, is one of two native flowering plants found in the Antarctic region. It has yellow flowers and grows about 5 cm (two inches) tall, with a cushion plant, cushion-like growth habit that gives it a ...
,'' show a variation of photosynthesis where calcium oxalate crystals
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macros ...
function as dynamic carbon pools, supplying carbon dioxide ( CO2) to photosynthetic cells when stomata are partially or totally closed. This process was named Alarm photosynthesis. Under stress conditions (e.g. water deficit) oxalate released from calcium oxalate crystals is converted to CO2 by an oxalate oxidase enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
and the produced CO2 can support the Calvin cycle reactions. Reactive hydrogen peroxide ( H2O2), the byproduct of oxalate oxidase reaction, can be neutralized by catalase. Alarm photosynthesis represents a photosynthetic variant to be added to the well-known C4 and CAM
Calmodulin (CaM) (an abbreviation for calcium-modulated protein) is a multifunctional intermediate calcium-binding messenger protein expressed in all eukaryotic cells. It is an intracellular target of the secondary messenger Ca2+, and the bin ...
pathways. However, alarm photosynthesis, in contrast to these pathways, operates as a biochemical pump that collects carbon from the organ interior (or from the soil) and not from the atmosphere.
In water
Cyanobacteria possesscarboxysome
Carboxysomes are bacterial microcompartments (BMCs) consisting of polyhedral protein shells filled with the enzymes ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO)—the predominant enzyme in carbon fixation and the rate limiting e ...
s, which increase the concentration of around RuBisCO to increase the rate of photosynthesis. An enzyme, carbonic anhydrase, located within the carboxysome releases CO2 from dissolved hydrocarbonate ions (HCO). Before the CO2 diffuses out it is quickly sponged up by RuBisCO, which is concentrated within the carboxysomes. HCO ions are made from CO2 outside the cell by another carbonic anhydrase and are actively pumped into the cell by a membrane protein. They cannot cross the membrane as they are charged, and within the cytosol they turn back into CO2 very slowly without the help of carbonic anhydrase. This causes the HCO ions to accumulate within the cell from where they diffuse into the carboxysomes. Pyrenoid
Pyrenoids are sub-cellular micro-compartments found in chloroplasts of many algae,Giordano, M., Beardall, J., & Raven, J. A. (2005). CO2 concentrating mechanisms in algae: mechanisms, environmental modulation, and evolution. Annu. Rev. Plant Bio ...
s in algae and hornworts also act to concentrate around RuBisCO.
Order and kinetics
The overall process of photosynthesis takes place in four stages:Efficiency
Plants usually convert light into chemical energy with a photosynthetic efficiency of 3–6%. Absorbed light that is unconverted is dissipated primarily as heat, with a small fraction (1–2%) re-emitted aschlorophyll fluorescence
Chlorophyll fluorescence is light re-emitted by chlorophyll molecules during return from excited to non-excited states. It is used as an indicator of photosynthetic energy conversion in plants, algae and bacteria
Bacteria (; singular: b ...
at longer (redder) wavelengths. This fact allows measurement of the light reaction of photosynthesis by using chlorophyll fluorometers.
Actual plants' photosynthetic efficiency varies with the frequency of the light being converted, light intensity, temperature and proportion of carbon dioxide in the atmosphere, and can vary from 0.1% to 8%. By comparison, solar panels convert light into electric energy
Electrical energy is energy related to forces on electrically charged particles and the movement of electrically charged particles (often electrons in wires, but not always). This energy is supplied by the combination of electric current and electr ...
at an efficiency of approximately 6–20% for mass-produced panels, and above 40% in laboratory devices.
Scientists are studying photosynthesis in hopes of developing plants with increased yield.
The efficiency of both light and dark reactions can be measured but the relationship between the two can be complex. For example, the ATP and NADPH energy molecules, created by the light reaction, can be used for carbon fixation or for photorespiration in C3 plants. Electrons may also flow to other electron sinks. For this reason, it is not uncommon for authors to differentiate between work done under non-photorespiratory conditions and under photorespiratory conditions.
Chlorophyll fluorescence of photosystem II can measure the light reaction, and infrared gas analyzers can measure the dark reaction. It is also possible to investigate both at the same time using an integrated chlorophyll fluorometer and gas exchange system, or by using two separate systems together. Infrared gas analyzers and some moisture sensors are sensitive enough to measure the photosynthetic assimilation of CO2, and of ΔH2O using reliable methods CO2 is commonly measured in μmols/(m2/s), parts per million or volume per million and H2O is commonly measured in mmol/(m2/s) or in mbars. By measuring CO2 assimilation, ΔH2O, leaf temperature, barometric pressure, leaf area, and photosynthetically active radiation or PAR, it becomes possible to estimate, "A" or carbon assimilation, "E" or transpiration, "gs" or stomatal conductance, and Ci or intracellular CO2. However, it is more common to used chlorophyll fluorescence for plant stress measurement, where appropriate, because the most commonly used parameters FV/FM and Y(II) or F/FM' can be measured in a few seconds, allowing the investigation of larger plant populations.
Gas exchange systems that offer control of CO2 levels, above and below ambient, allow the common practice of measurement of A/Ci curves, at different CO2 levels, to characterize a plant's photosynthetic response.
Integrated chlorophyll fluorometer – gas exchange systems allow a more precise measure of photosynthetic response and mechanisms. While standard gas exchange photosynthesis systems can measure Ci, or substomatal CO2 levels, the addition of integrated chlorophyll fluorescence measurements allows a more precise measurement of CC to replace Ci. The estimation of CO2 at the site of carboxylation in the chloroplast, or CC, becomes possible with the measurement of mesophyll conductance or gm using an integrated system.
Photosynthesis measurement systems are not designed to directly measure the amount of light absorbed by the leaf. But analysis of chlorophyll-fluorescence, P700- and P515-absorbance and gas exchange measurements reveal detailed information about e.g. the photosystems, quantum efficiency and the CO2 assimilation rates. With some instruments, even wavelength-dependency of the photosynthetic efficiency can be analyzed.
A phenomenon known as quantum walk
Quantum walks are quantum analogues of classical random walks. In contrast to the classical random walk, where the walker occupies definite states and the randomness arises due to stochastic transitions between states, in quantum walks randomness ...
increases the efficiency of the energy transport of light significantly. In the photosynthetic cell of an alga, bacterium, or plant, there are light-sensitive molecules called chromophores arranged in an antenna-shaped structure named a photocomplex. When a photon is absorbed by a chromophore, it is converted into a quasiparticle
In physics, quasiparticles and collective excitations are closely related emergent phenomena arising when a microscopically complicated system such as a solid behaves as if it contained different weakly interacting particles in vacuum.
For exa ...
referred to as an exciton, which jumps from chromophore to chromophore towards the reaction center of the photocomplex, a collection of molecules that traps its energy in a chemical form accessible to the cell's metabolism. The exciton's wave properties enable it to cover a wider area and try out several possible paths simultaneously, allowing it to instantaneously "choose" the most efficient route, where it will have the highest probability of arriving at its destination in the minimum possible time.
Because that quantum walking takes place at temperatures far higher than quantum phenomena usually occur, it is only possible over very short distances. Obstacles in the form of destructive interference cause the particle to lose its wave properties for an instant before it regains them once again after it is freed from its locked position through a classic "hop". The movement of the electron towards the photo center is therefore covered in a series of conventional hops and quantum walks.
Evolution
Early photosynthetic systems, such as those ingreen
Green is the color between cyan and yellow on the visible spectrum. It is evoked by light which has a dominant wavelength of roughly 495570 nm. In subtractive color systems, used in painting and color printing, it is created by a combi ...
and purple sulfur and green
Green is the color between cyan and yellow on the visible spectrum. It is evoked by light which has a dominant wavelength of roughly 495570 nm. In subtractive color systems, used in painting and color printing, it is created by a combi ...
and purple nonsulfur bacteria, are thought to have been anoxygenic, and used various other molecules than water as electron donor
In chemistry, an electron donor is a chemical entity that donates electrons to another compound. It is a reducing agent that, by virtue of its donating electrons, is itself oxidized in the process.
Typical reducing agents undergo permanent chemi ...
s. Green and purple sulfur bacteria are thought to have used hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
and sulfur as electron donors. Green nonsulfur bacteria used various amino
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent ...
and other organic acids as electron donors. Purple nonsulfur bacteria used a variety of nonspecific organic molecules. The use of these molecules is consistent with the geological evidence that Earth's early atmosphere was highly reducing at that time.
Fossils of what are thought to be filamentous photosynthetic organisms have been dated at 3.4 billion years old. More recent studies also suggest that photosynthesis may have begun about 3.4 billion years ago.
The main source of oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as ...
in the Earth's atmosphere
The atmosphere of Earth is the layer of gases, known collectively as air, retained by Earth's gravity that surrounds the planet and forms its planetary atmosphere. The atmosphere of Earth protects life on Earth by creating pressure allowing fo ...
derives from oxygenic photosynthesis, and its appearance is sometimes referred to as the oxygen catastrophe. Geological evidence suggests that oxygenic photosynthesis, such as that in cyanobacteria, became important during the Paleoproterozoic
The Paleoproterozoic Era (;, also spelled Palaeoproterozoic), spanning the time period from (2.5–1.6 Ga), is the first of the three sub-divisions ( eras) of the Proterozoic Eon. The Paleoproterozoic is also the longest era of the Earth's ...
era around 2 billion years ago. Modern photosynthesis in plants and most photosynthetic prokaryotes is oxygenic, using water as an electron donor, which is oxidized to molecular oxygen in the photosynthetic reaction center.
Symbiosis and the origin of chloroplasts
 Several groups of animals have formed symbiotic relationships with photosynthetic algae. These are most common in
Several groups of animals have formed symbiotic relationships with photosynthetic algae. These are most common in coral
Corals are marine invertebrates within the class Anthozoa of the phylum Cnidaria. They typically form compact colonies of many identical individual polyps. Coral species include the important reef builders that inhabit tropical oceans and ...
s, sponge
Sponges, the members of the phylum Porifera (; meaning 'pore bearer'), are a basal animal clade as a sister of the diploblasts. They are multicellular organisms that have bodies full of pores and channels allowing water to circulate throug ...
s and sea anemones. It is presumed that this is due to the particularly simple body plan
A body plan, ( ), or ground plan is a set of morphological features common to many members of a phylum of animals. The vertebrates share one body plan, while invertebrates have many.
This term, usually applied to animals, envisages a "blueprin ...
s and large surface areas of these animals compared to their volumes. In addition, a few marine mollusks '' Elysia viridis'' and '' Elysia chlorotica'' also maintain a symbiotic relationship with chloroplasts they capture from the algae in their diet and then store in their bodies (see Kleptoplasty
Kleptoplasty or kleptoplastidy is a symbiotic phenomenon whereby plastids, notably chloroplasts from algae, are sequestered by host organisms. The word is derived from ''Kleptes'' (κλέπτης) which is Greek for thief. The alga is eaten norma ...
). This allows the mollusks to survive solely by photosynthesis for several months at a time. Some of the genes from the plant cell nucleus have even been transferred to the slugs, so that the chloroplasts can be supplied with proteins that they need to survive.
An even closer form of symbiosis may explain the origin of chloroplasts. Chloroplasts have many similarities with photosynthetic bacteria, including a circular chromosome
A chromosome is a long DNA molecule with part or all of the genetic material of an organism. In most chromosomes the very long thin DNA fibers are coated with packaging proteins; in eukaryotic cells the most important of these proteins are ...
, prokaryotic-type ribosome, and similar proteins in the photosynthetic reaction center. The endosymbiotic theory suggests that photosynthetic bacteria were acquired (by endocytosis) by early eukaryotic
Eukaryotes () are organisms whose Cell (biology), cells have a cell nucleus, nucleus. All animals, plants, fungi, and many unicellular organisms, are Eukaryotes. They belong to the group of organisms Eukaryota or Eukarya, which is one of the ...
cells to form the first plant cells. Therefore, chloroplasts may be photosynthetic bacteria that adapted to life inside plant cells. Like mitochondria, chloroplasts possess their own DNA, separate from the nuclear DNA of their plant host cells and the genes in this chloroplast DNA resemble those found in cyanobacteria. DNA in chloroplasts codes for redox
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a ...
proteins such as those found in the photosynthetic reaction centers. The CoRR Hypothesis proposes that this co-location of genes with their gene products is required for redox regulation of gene expression, and accounts for the persistence of DNA in bioenergetic organelles.
Photosynthetic eukaryotic lineages
Symbiotic and Kleptoplasty, kleptoplastic organisms excluded: *The glaucophytes and the Red algae, red and Viridiplantae, green algae—clade Archaeplastida (uni- and multicellular) *The Cryptophyceae, cryptophytes—clade Cryptista (unicellular) *The haptophytes—clade Haptista (unicellular) *The dinoflagellates and Chromerida, chromerids in the superphylum Myzozoa, and Pseudoblepharisma in the phylum Ciliate, Ciliophora—clade Alveolata (unicellular) *The Ochrophyta, ochrophytes—clade Heterokonta (uni- and multicellular) *The chlorarachniophytes and 3 species of Paulinella in the phylum Cercozoa—clade Rhizaria (unicellular) *The euglenids—clade Excavata (unicellular) Except for the euglenids, which are found within the Excavata, all of these belong to the Plants+HC+SAR megagroup, Diaphoretickes. Archaeplastida and the photosynthetic Paulinella got their plastids, which are surrounded by two membranes, through primary Endosymbiont, endosymbiosis in two separate events, by engulfing a cyanobacterium. The plastids in all the other groups have either a red or green algal origin, and are referred to as the "red lineages" and the "green lineages". The only known exception is the ciliate ''Pseudoblepharisma tenue'', which in addition to its plastids that originated from green algae also has a purple sulfur bacteria, purple sulfur bacterium as symbiont. In dinoflagellates and euglenids the plastids are surrounded by three membranes, and in the remaining lines by four. A nucleomorph, remnants of the original algal nucleus located between the inner and outer membranes of the plastid, is present in the cryptophytes (from a red alga) and chlorarachniophytes (from a green alga). Some dinoflagellates that lost their photosynthetic ability later regained it again through new endosymbiotic events with different algae. While able to perform photosynthesis, many of these eukaryotic groups are mixotrophs and practice heterotrophy to various degrees.Cyanobacteria and the evolution of photosynthesis
The biochemical capacity to use water as the source for electrons in photosynthesis evolved once, in a common ancestor of extant cyanobacteria (formerly called blue-green algae), which are the only prokaryotes performing oxygenic photosynthesis. The geological record indicates that this transforming event took place early in Earth's history, at least 2450–2320 million years ago (Ma), and, it is speculated, much earlier. Because the Earth's atmosphere contained almost no oxygen during the estimated development of photosynthesis, it is believed that the first photosynthetic cyanobacteria did not generate oxygen. Available evidence from geobiological studies of Archean (>2500 Ma) sedimentary rocks indicates that life existed 3500 Ma, but the question of when oxygenic photosynthesis evolved is still unanswered. A clear paleontological window on cyanobacterial evolution opened about 2000 Ma, revealing an already-diverse biota of cyanobacteria. Cyanobacteria remained the principal primary producers of oxygen throughout the Proterozoic Eon (2500–543 Ma), in part because the redox structure of the oceans favored photoautotrophs capable of nitrogen fixation. Green algae joined cyanobacteria as the major primary producers of oxygen on continental shelves near the end of the Proterozoic, but only with the Mesozoic (251–66 Ma) radiations of dinoflagellates, coccolithophorids, and diatoms did the primary production of oxygen in marine shelf waters take modern form. Cyanobacteria remain critical to marine ecosystems as Oxygen cycle, primary producers of oxygen in oceanic gyres, as agents of biological nitrogen fixation, and, in modified form, as the plastids of marine algae.Experimental history
Discovery
Although some of the steps in photosynthesis are still not completely understood, the overall photosynthetic equation has been known since the 19th century. Jan Baptist van Helmont, Jan van Helmont began the research of the process in the mid-17th century when he carefully measured the mass of the soil used by a plant and the mass of the plant as it grew. After noticing that the soil mass changed very little, he hypothesized that the mass of the growing plant must come from the water, the only substance he added to the potted plant. His hypothesis was partially accurate – much of the gained mass comes from carbon dioxide as well as water. However, this was a signaling point to the idea that the bulk of a plant's biomass comes from the inputs of photosynthesis, not the soil itself.
Joseph Priestley, a chemist and minister, discovered that when he isolated a volume of air under an inverted jar and burned a candle in it (which gave off CO2), the candle would burn out very quickly, much before it ran out of wax. He further discovered that a mouse could similarly Breathing, "injure" air. He then showed that the air that had been "injured" by the candle and the mouse could be restored by a plant.
In 1779,
Jan Baptist van Helmont, Jan van Helmont began the research of the process in the mid-17th century when he carefully measured the mass of the soil used by a plant and the mass of the plant as it grew. After noticing that the soil mass changed very little, he hypothesized that the mass of the growing plant must come from the water, the only substance he added to the potted plant. His hypothesis was partially accurate – much of the gained mass comes from carbon dioxide as well as water. However, this was a signaling point to the idea that the bulk of a plant's biomass comes from the inputs of photosynthesis, not the soil itself.
Joseph Priestley, a chemist and minister, discovered that when he isolated a volume of air under an inverted jar and burned a candle in it (which gave off CO2), the candle would burn out very quickly, much before it ran out of wax. He further discovered that a mouse could similarly Breathing, "injure" air. He then showed that the air that had been "injured" by the candle and the mouse could be restored by a plant.
In 1779, Jan Ingenhousz
Jan (or John) Ingenhousz or Ingen-Housz FRS (8 December 1730 – 7 September 1799) was a Dutch-born British physiologist, biologist and chemist.
He is best known for discovering photosynthesis by showing that light is essential to the process ...
repeated Priestley's experiments. He discovered that it was the influence of sunlight on the plant that could cause it to revive a mouse in a matter of hours.
In 1796, Jean Senebier, a Swiss pastor, botanist, and naturalist, demonstrated that green plants consume carbon dioxide and release oxygen under the influence of light. Soon afterward, Nicolas-Théodore de Saussure showed that the increase in mass of the plant as it grows could not be due only to uptake of CO2 but also to the incorporation of water. Thus, the basic reaction by which photosynthesis is used to produce food (such as glucose) was outlined.
Refinements
Cornelis Van Niel made key discoveries explaining the chemistry of photosynthesis. By studying purple sulfur bacteria and green bacteria he was the first to demonstrate that photosynthesis is a light-dependent redox reaction, in which hydrogen Reducing agent, reduces (donates its atoms as electrons and protons to) carbon dioxide. Robert Emerson (scientist), Robert Emerson discovered two light reactions by testing plant productivity using different wavelengths of light. With the red alone, the light reactions were suppressed. When blue and red were combined, the output was much more substantial. Thus, there were two photosystems, one absorbing up to 600 nm wavelengths, the other up to 700 nm. The former is known as PSII, the latter is PSI. PSI contains only chlorophyll "a", PSII contains primarily chlorophyll "a" with most of the available chlorophyll "b", among other pigments. These include phycobilins, which are the red and blue pigments of red and blue algae, respectively, and fucoxanthol for brown algae and diatoms. The process is most productive when the absorption of quanta is equal in both PSII and PSI, assuring that input energy from the antenna complex is divided between the PSI and PSII systems, which in turn powers the photochemistry. Robin Hill (biochemist), Robert Hill thought that a complex of reactions consisted of an intermediate to cytochrome b6 (now a plastoquinone), and that another was from cytochrome f to a step in the carbohydrate-generating mechanisms. These are linked by plastoquinone, which does require energy to reduce cytochrome f. Further experiments to prove that the oxygen developed during the photosynthesis of green plants came from water were performed by Hill in 1937 and 1939. He showed that isolated chloroplasts give off oxygen in the presence of unnatural reducing agents like iron oxalate, ferricyanide or benzoquinone after exposure to light. In the Hill reaction: :2 H2O + 2 A + (light, chloroplasts) → 2 AH2 + O2 A is the electron acceptor. Therefore, in light, the electron acceptor is reduced and oxygen is evolved. Sam Ruben, Samuel Ruben and Martin Kamen used Radionuclide, radioactive isotopes to determine that the oxygen liberated in photosynthesis came from the water. Melvin Calvin and Andrew Benson, along with James Bassham, elucidated the path of carbon assimilation (the photosynthetic carbon reduction cycle) in plants. The carbon reduction cycle is known as the Calvin cycle, but many scientists refer to it as the Calvin-Benson, Benson-Calvin, or even Calvin-Benson-Bassham (or CBB) Cycle.
Nobel Prize-winning scientist Rudolph A. Marcus was later able to discover the function and significance of the electron transport chain.
Otto Heinrich Warburg and Dean Burk discovered the I-quantum photosynthesis reaction that splits CO2, activated by the respiration.
In 1950, first experimental evidence for the existence of
Melvin Calvin and Andrew Benson, along with James Bassham, elucidated the path of carbon assimilation (the photosynthetic carbon reduction cycle) in plants. The carbon reduction cycle is known as the Calvin cycle, but many scientists refer to it as the Calvin-Benson, Benson-Calvin, or even Calvin-Benson-Bassham (or CBB) Cycle.
Nobel Prize-winning scientist Rudolph A. Marcus was later able to discover the function and significance of the electron transport chain.
Otto Heinrich Warburg and Dean Burk discovered the I-quantum photosynthesis reaction that splits CO2, activated by the respiration.
In 1950, first experimental evidence for the existence of photophosphorylation In the process of photosynthesis, the phosphorylation of ADP to form ATP using the energy of sunlight is called photophosphorylation. Cyclic photophosphorylation occurs in both aerobic and anaerobic conditions, driven by the main primary source of ...
''in vivo'' was presented by Otto Kandler using intact ''Chlorella'' cells and interpreting his findings as light-dependent ATP formation.
In 1954, Daniel I. Arnon et al. discovered photophosphorylation ''in vitro'' in isolated chloroplasts with the help of P32.
Louis N.M. Duysens and Jan Amesz discovered that chlorophyll "a" will absorb one light, oxidize cytochrome f, while chlorophyll "a" (and other pigments) will absorb another light but will reduce this same oxidized cytochrome, stating the two light reactions are in series.
Development of the concept
In 1893, Charles Reid Barnes proposed two terms, ''photosyntax'' and ''photosynthesis'', for the biological process of ''synthesis of complex carbon compounds out of carbonic acid, in the presence of chlorophyll, under the influence of light''. Over time, the term ''photosynthesis'' came into common usage. Later discovery of anoxygenic photosynthetic bacteria and photophosphorylation necessitated redefinition of the term.C3 : C4 photosynthesis research
In the late 1940s at the University of California, Berkeley, the details of photosynthetic carbon metabolism were sorted out by the chemists Melvin Calvin, Andrew Benson, James Bassham and a score of students and researchers utilizing the carbon-14 isotope and paper chromatography techniques. The pathway of CO2 fixation by the algae ''Chlorella'' in a fraction of a second in light resulted in a 3 carbon molecule called phosphoglyceric acid (PGA). For that original and ground-breaking work, a Nobel Prize in Chemistry was awarded to Melvin Calvin in 1961. In parallel, plant physiologists studied leaf gas exchanges using the new method of infrared gas analysis and a leaf chamber where the net photosynthetic rates ranged from 10 to 13 μmol CO2·m−2·s−1, with the conclusion that all terrestrial plants have the same photosynthetic capacities, that are light saturated at less than 50% of sunlight. Later in 1958–1963 at Cornell University, field grown maize was reported to have much greater leaf photosynthetic rates of 40 μmol CO2·m−2·s−1 and not be saturated at near full sunlight. This higher rate in maize was almost double of those observed in other species such as wheat and soybean, indicating that large differences in photosynthesis exist among higher plants. At the University of Arizona, detailed gas exchange research on more than 15 species of Monocotyledon, monocot and Dicotyledon, dicot uncovered for the first time that differences in leaf anatomy are crucial factors in differentiating photosynthetic capacities among species. In tropical grasses, including maize, sorghum, sugarcane, Bermuda grass and in the dicot amaranthus, leaf photosynthetic rates were around 38−40 μmol CO2·m−2·s−1, and the leaves have two types of green cells, i. e. outer layer of mesophyll cells surrounding a tightly packed cholorophyllous vascular bundle sheath cells. This type of anatomy was termed Kranz anatomy in the 19th century by the botanist Gottlieb Haberlandt while studying leaf anatomy of sugarcane. Plant species with the greatest photosynthetic rates and Kranz anatomy showed no apparent photorespiration, very low CO2 compensation point, high optimum temperature, high stomatal resistances and lower mesophyll resistances for gas diffusion and rates never saturated at full sun light. The research at Arizona was designated a Citation Classic in 1986. These species were later termed C4 plants as the first stable compound of CO2 fixation in light has 4 carbons as malate and aspartate. Other species that lack Kranz anatomy were termed C3 type such as cotton and sunflower, as the first stable carbon compound is the 3-carbon PGA. At 1000 ppm CO2 in measuring air, both the C3 and C4 plants had similar leaf photosynthetic rates around 60 μmol CO2·m−2·s−1 indicating the suppression of photorespiration in C3 plants.Factors
 There are three main factors affecting photosynthesis and several corollary factors. The three main are:
* Light irradiance and
There are three main factors affecting photosynthesis and several corollary factors. The three main are:
* Light irradiance and wavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, t ...
* Carbon dioxide concentration
* Temperature.
Total photosynthesis is limited by a range of environmental factors. These include the amount of light available, the amount of leaf area a plant has to capture light (shading by other plants is a major limitation of photosynthesis), the rate at which carbon dioxide can be supplied to the chloroplasts to support photosynthesis, the availability of water, and the availability of suitable temperatures for carrying out photosynthesis.
Light intensity (irradiance), wavelength and temperature
Carbon dioxide levels and photorespiration
RuBisCO
Ribulose-1,5-bisphosphate carboxylase-oxygenase, commonly known by the abbreviations RuBisCo, rubisco, RuBPCase, or RuBPco, is an enzyme () involved in the first major step of carbon fixation, a process by which atmospheric carbon dioxide is con ...
, the enzyme that captures carbon dioxide in the light-independent reactions, has a binding affinity for both carbon dioxide and oxygen. When the concentration of carbon dioxide is high, RuBisCO will fix carbon dioxide. However, if the carbon dioxide concentration is low, RuBisCO will bind oxygen instead of carbon dioxide. This process, called photorespiration, uses energy, but does not produce sugars.
RuBisCO oxygenase activity is disadvantageous to plants for several reasons:
# One product of oxygenase activity is phosphoglycolate (2 carbon) instead of 3-phosphoglycerate (3 carbon). Phosphoglycolate cannot be metabolized by the Calvin-Benson cycle and represents carbon lost from the cycle. A high oxygenase activity, therefore, drains the sugars that are required to recycle ribulose 5-bisphosphate and for the continuation of the Calvin-Benson cycle.
# Phosphoglycolate is quickly metabolized to glycolate that is toxic to a plant at a high concentration; it inhibits photosynthesis.
# Salvaging glycolate is an energetically expensive process that uses the glycolate pathway, and only 75% of the carbon is returned to the Calvin-Benson cycle as 3-phosphoglycerate. The reactions also produce ammonia (NH3), which is able to Molecular diffusion, diffuse out of the plant, leading to a loss of nitrogen.
::A highly simplified summary is:
:::2 glycolate + ATP → 3-phosphoglycerate + carbon dioxide + ADP + NH3
The salvaging pathway for the products of RuBisCO oxygenase activity is more commonly known as photorespiration, since it is characterized by light-dependent oxygen consumption and the release of carbon dioxide.
See also
* Jan Anderson (scientist) * Artificial photosynthesis * Calvin-Benson cycle * Carbon fixation * Cellular respiration * Chemosynthesis * Daily light integral * Hill reaction * Integrated fluorometer * Light-dependent reaction * Organic reaction * Photobiology * Photoinhibition * Photosynthetic reaction center * Photosynthetically active radiation * Photosystem *Photosystem I
Photosystem I (PSI, or plastocyanin–ferredoxin oxidoreductase) is one of two photosystems in the photosynthetic light reactions of algae, plants, and cyanobacteria. Photosystem I is an integral membrane protein complex that us ...
* Photosystem II
* Quantasome
* Quantum biology
* Radiosynthesis (metabolism), Radiosynthesis
* Red edge
* Vitamin D
References
Further reading
Books
* * * *Papers
* *External links
A collection of photosynthesis pages for all levels from a renowned expert (Govindjee)
* [https://web.archive.org/web/20090428090455/http://scienceaid.co.uk/biology/biochemistry/photosynthesis.html Science Aid: Photosynthesis] Article appropriate for high school science
Metabolism, Cellular Respiration and Photosynthesis – The Virtual Library of Biochemistry and Cell Biology
* [http://www.life.uiuc.edu/govindjee/photosynBook.html Overall Energetics of Photosynthesis]
The source of oxygen produced by photosynthesis
Interactive animation, a textbook tutorial *
Photosynthesis – Light Dependent & Light Independent Stages
Khan Academy, video introduction
{{Authority control Photosynthesis, Agronomy Biological processes Botany Cellular respiration Ecosystems Metabolism Plant nutrition Plant physiology Quantum biology