Neonatal infection on:
[Wikipedia]
[Google]
[Amazon]
Neonatal infections are infections of the
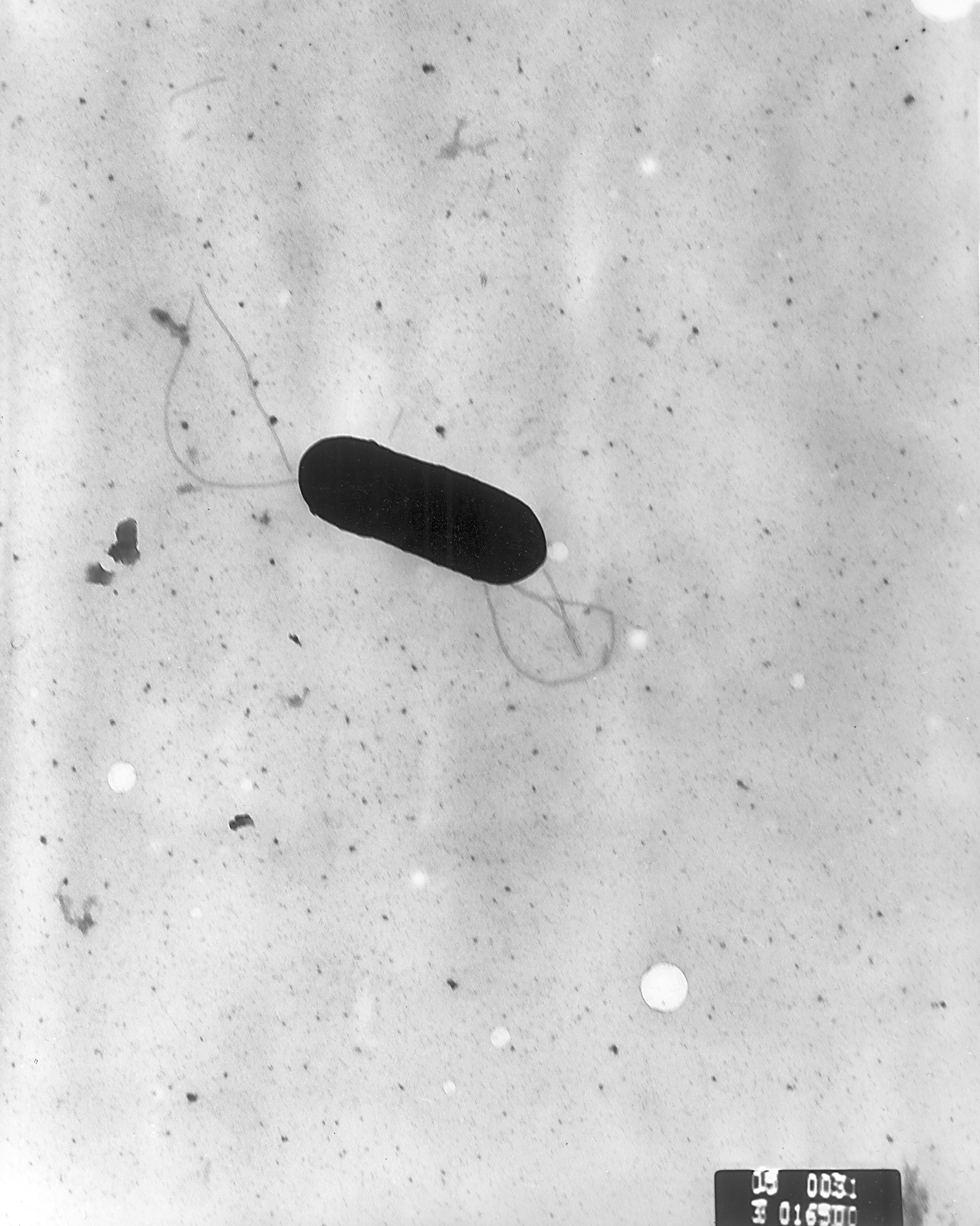 Group B streptococcus are typically identified as the cause of the majority of early-onset infections in the neonate.
This pathogen is vertically transmitted (transmitted directly from the mother) to the infant.
Group B streptococcus are typically identified as the cause of the majority of early-onset infections in the neonate.
This pathogen is vertically transmitted (transmitted directly from the mother) to the infant.
 Inflammation accompanies infection and is likely to complicate treatment and recovery. Inflammation is linked to reduced growth of the lungs of the premature baby.
Inflammation accompanies infection and is likely to complicate treatment and recovery. Inflammation is linked to reduced growth of the lungs of the premature baby.
neonate
An infant or baby is the very young offspring of human beings. ''Infant'' (from the Latin word ''infans'', meaning 'unable to speak' or 'speechless') is a formal or specialised synonym for the common term ''baby''. The terms may also be used to ...
(newborn) acquired during prenatal development or in the first four weeks of life (neonatal period). Neonatal infections may be contracted by mother to child transmission, in the birth canal
In mammals, the vagina is the elastic, muscular part of the female genital tract. In humans, it extends from the vestibule to the cervix. The outer vaginal opening is normally partly covered by a thin layer of mucosal tissue called the hyme ...
during childbirth
Childbirth, also known as labour and delivery, is the ending of pregnancy where one or more babies exits the internal environment of the mother via vaginal delivery or caesarean section. In 2019, there were about 140.11 million births glob ...
, or contracted after birth. Some neonatal infections are apparent soon after delivery, while others may develop in the postnatal period. Some neonatal infections such as HIV
The human immunodeficiency viruses (HIV) are two species of ''Lentivirus'' (a subgroup of retrovirus) that infect humans. Over time, they cause acquired immunodeficiency syndrome (AIDS), a condition in which progressive failure of the immune ...
, hepatitis B
Hepatitis B is an infectious disease caused by the '' Hepatitis B virus'' (HBV) that affects the liver; it is a type of viral hepatitis. It can cause both acute and chronic infection.
Many people have no symptoms during an initial infection. ...
, and malaria
Malaria is a mosquito-borne infectious disease that affects humans and other animals. Malaria causes symptoms that typically include fever, tiredness, vomiting, and headaches. In severe cases, it can cause jaundice, seizures, coma, or death. S ...
do not become apparent until much later.
There is a higher risk of infection for preterm
Preterm birth, also known as premature birth, is the birth of a baby at fewer than 37 weeks gestational age, as opposed to full-term delivery at approximately 40 weeks. Extreme preterm is less than 28 weeks, very early preterm birth is between ...
or low birth weight
Low birth weight (LBW) is defined by the World Health Organization as a birth weight of an
infant of or less, regardless of gestational age. Infants born with LBW have added health risks which require close management, often in a neonatal inten ...
neonates. Infant respiratory distress syndrome
Infantile respiratory distress syndrome (IRDS), also called respiratory distress syndrome of newborn, or increasingly surfactant deficiency disorder (SDD), and previously called hyaline membrane disease (HMD), is a syndrome in premature infants ...
is often a condition of preterm neonates that can have long-term negative consequences, it can also arise following an infection. In some instances, neonatal respiratory tract diseases may increase the susceptibility to future respiratory infections and inflammatory responses related to lung disease.
Antibiotics
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention o ...
can be effective for neonatal infections, especially when the pathogen
In biology, a pathogen ( el, πάθος, "suffering", "passion" and , "producer of") in the oldest and broadest sense, is any organism or agent that can produce disease. A pathogen may also be referred to as an infectious agent, or simply a germ ...
is quickly identified. Instead of relying solely on culturing techniques, pathogen identification has improved substantially with advancing technology; however, neonate mortality reduction has not kept pace and remains 20% to 50%. While preterm neonates are at a particularly high risk, all neonates can develop infection. Neonatal infection may also be associated with premature rupture of membranes (breakage of the amniotic sac
The amniotic sac, also called the bag of waters or the membranes, is the sac in which the embryo and later fetus develops in amniotes. It is a thin but tough transparent pair of membranes that hold a developing embryo (and later fetus) until s ...
) which substantially increases the risk of neonatal sepsis
Neonatal sepsis is a type of neonatal infection and specifically refers to the presence in a newborn baby of a bacterial blood stream infection (BSI) (such as meningitis, pneumonia, pyelonephritis, or gastroenteritis) in the setting of fever. ...
by allowing passage for bacteria to enter the womb prior to the birth of the infant. Neonatal infection can be distressing to the family and it initiates concentrated effort to treat it by clinicians. Research to improve treatment of infections and prophylactic treatment of the mother to avoid infections of the infant is ongoing.
Causes
In industrialized countries, treatment for neonatal infections takes place in the neonatal intensive care unit (NICU). The causes and reasons for neonatal infection are many. The origin of infectiousbacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one Cell (biology), biological cell. They constitute a large domain (biology), domain of prokaryotic microorganisms. Typically a few micrometr ...
and some other pathogen
In biology, a pathogen ( el, πάθος, "suffering", "passion" and , "producer of") in the oldest and broadest sense, is any organism or agent that can produce disease. A pathogen may also be referred to as an infectious agent, or simply a germ ...
s is often the maternal
]
A mother is the female parent of a child. A woman may be considered a mother by virtue of having given birth, by raising a child who may or may not be her biological offspring, or by supplying her ovum for fertilisation in the case of gestat ...
gastrointestinal tract, gastrointestinal and genitourinary tract
The genitourinary system, or urogenital system, are the organs of the reproductive system and the urinary system. These are grouped together because of their proximity to each other, their common embryological origin and the use of common pathw ...
. Many of the maternal infections with these organisms are asymptomatic in the mother. Other maternal infections that may be transmitted to the infant in utero or during birth are bacterial and viral sexually transmitted infections. The infant's ability to resist infection is limited by its immature immune system
The immune system is a network of biological processes that protects an organism from diseases. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splint ...
. The causative agents of neonatal infection are bacteria, viruses, and fungi
A fungus ( : fungi or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and molds, as well as the more familiar mushrooms. These organisms are classified as a kingdom, separately from ...
. In addition, the immune system of the neonate may respond in ways that can create problems that complicate treatment, such as the release of inflammatory chemicals. Congenital defects of the immune system also affect the infants ability to fight off the infection.
Bacteria
 Group B streptococcus are typically identified as the cause of the majority of early-onset infections in the neonate.
This pathogen is vertically transmitted (transmitted directly from the mother) to the infant.
Group B streptococcus are typically identified as the cause of the majority of early-onset infections in the neonate.
This pathogen is vertically transmitted (transmitted directly from the mother) to the infant. Enteric
The gastrointestinal tract (GI tract, digestive tract, alimentary canal) is the tract or passageway of the digestive system that leads from the mouth to the anus. The GI tract contains all the major organs of the digestive system, in humans and ...
bacilli
Bacilli is a taxonomic class of bacteria that includes two orders, Bacillales and Lactobacillales, which contain several well-known pathogens such as ''Bacillus anthracis'' (the cause of anthrax). ''Bacilli'' are almost exclusively gram-positi ...
that originate from the digestive system of the mother have become as prevalent as the group B streptococcus pathogens and are currently as likely to cause infection. With the advances in preventing group B streptococcus infections, β-lactam-resistant ''Escherichia coli
''Escherichia coli'' (),Wells, J. C. (2000) Longman Pronunciation Dictionary. Harlow ngland Pearson Education Ltd. also known as ''E. coli'' (), is a Gram-negative, facultative anaerobic, rod-shaped, coliform bacterium of the genus '' Esc ...
'' infections have increased in causing neonatal deaths in very low birthweight and premature infants. Infections with '' Staphylococcus aureus'' are also diagnosed, but not as frequently as group B streptococcus infections.
''Listeria monocytogenes
''Listeria monocytogenes'' is the species of pathogenic bacteria that causes the infection listeriosis. It is a facultative anaerobic bacterium, capable of surviving in the presence or absence of oxygen. It can grow and reproduce inside the host ...
'' can also cause infection acquired from tainted food and present in the mother. The presence of this pathogen can sometimes be determined by the symptoms that appear as a gastrointestinal illness in the mother. The mother acquires infection from ingesting food that contains animal product
An animal product is any material derived from the body of an animal. Examples are fat, flesh, blood, milk, eggs, and lesser known products, such as isinglass and rennet.
Animal by-products, as defined by the USDA, are products harvested or m ...
s such as hot dog
A hot dog (uncommonly spelled hotdog) is a food consisting of a grilled or steamed sausage served in the slit of a partially sliced bun. The term hot dog can refer to the sausage itself. The sausage used is a wiener ( Vienna sausage) or a f ...
s, unpasteurized milk
Milk is a white liquid food produced by the mammary glands of mammals. It is the primary source of nutrition for young mammals (including breastfed human infants) before they are able to digest solid food. Immune factors and immune-modula ...
, delicatessen meats, and cheese.
Neonatal infection can also occur in term and post-term infants. Infections that develop one month after the birth of the infant are more likely due to Gram-positive bacteria and coagulase positive staphylococci.
Acquired maternal infection and subsequent inflammation from '' Ureaplasma urealyticum'' is accompanied by a strong immune response and is correlated with the need for prolonged mechanical ventilation
Mechanical ventilation, assisted ventilation or intermittent mandatory ventilation (IMV), is the medical term for using a machine called a ventilator to fully or partially provide artificial ventilation. Mechanical ventilation helps move a ...
.
''Clostridium tetani
''Clostridium tetani'' is a common soil bacterium and the causative agent of tetanus. Vegetative cells of ''Clostridium tetani'' are usually rod-shaped and up to 2.5 μm long, but they become enlarged and tennis racket- or drumstick-shaped wh ...
'' can cause a generalised form of tetanus
Tetanus, also known as lockjaw, is a bacterial infection caused by ''Clostridium tetani'', and is characterized by muscle spasms. In the most common type, the spasms begin in the jaw and then progress to the rest of the body. Each spasm usually ...
in the neonate. This usually occurs when the mother has not been vaccinated against tetanus and the baby has not acquired passive immunity. The umbilical cord region is the most susceptible.
Other bacterial pathogens include ''Streptococcus agalactiae
''Streptococcus agalactiae'' (also known as group B streptococcus or GBS) is a gram-positive coccus (round bacterium) with a tendency to form chains (as reflected by the genus name ''Streptococcus''). It is a beta- hemolytic, catalase-negative, ...
'', ''Streptococcus pyogenes
''Streptococcus pyogenes'' is a species of Gram-positive, aerotolerant bacteria in the genus '' Streptococcus''. These bacteria are extracellular, and made up of non-motile and non-sporing cocci (round cells) that tend to link in chains. They ...
'', ''Viridans streptococci
The viridans streptococci are a large group of commensal streptococcal Gram-positive bacteria species that are α-hemolytic, producing a green coloration on blood agar plates (hence the name "viridans", from Latin "vĭrĭdis", green), although ...
'', ''Streptococcus pneumoniae
''Streptococcus pneumoniae'', or pneumococcus, is a Gram-positive, spherical bacteria, alpha-hemolytic (under aerobic conditions) or beta-hemolytic (under anaerobic conditions), aerotolerant anaerobic member of the genus Streptococcus. They ar ...
'', ''Haemophilus influenzae
''Haemophilus influenzae'' (formerly called Pfeiffer's bacillus or ''Bacillus influenzae'') is a Gram-negative, non-motile, coccobacillary, facultatively anaerobic, capnophilic pathogenic bacterium of the family Pasteurellaceae. The bacter ...
'', and ''Pseudomonas aeruginosa
''Pseudomonas aeruginosa'' is a common encapsulated, gram-negative, aerobic–facultatively anaerobic, rod-shaped bacterium that can cause disease in plants and animals, including humans. A species of considerable medical importance, ''P. aerug ...
''.
Viruses
HIV
Human immunodeficiency virus
The human immunodeficiency viruses (HIV) are two species of ''Lentivirus'' (a subgroup of retrovirus) that infect humans. Over time, they cause acquired immunodeficiency syndrome (AIDS), a condition in which progressive failure of the immun ...
type I (HIV) infection can occur during labor and delivery, in utero through mother-to-child transmission or postnatally by way of breastfeeding. Transmission can occur during pregnancy, delivery or breastfeeding. Most transmission occurs during delivery. In women with low detectable levels of the virus, the incidence of transmission is lower. Transmission risk can be reduced by:
* providing antiretroviral therapy during pregnancy and immediately after birth
* delivery by caesarean section
* not breastfeeding
* antiretroviral prophylaxis in infants born to mothers with HIV.
A low number of women whose HIV status are unknown until after the birth, do not benefit from interventions that could help lower the risk of mother-to-child HIV transmission.
Cytomegalovirus
Sixty percent of mothers of preterm infants are infected with cytomegalovirus (CMV). Infection is asymptomatic in most instances but 9% to 12% of postnatally infected low birth weight, preterm infants have severe, sepsis-like infection. CMV infection duration can be long and result inpneumonitis
Pneumonitis describes general inflammation of lung tissue. Possible causative agents include radiation therapy of the chest, exposure to medications used during chemo-therapy, the inhalation of debris (e.g., animal dander), aspiration, herbicide ...
in association with fibrosis
Fibrosis, also known as fibrotic scarring, is a pathological wound healing in which connective tissue replaces normal parenchymal tissue to the extent that it goes unchecked, leading to considerable tissue remodelling and the formation of perma ...
. CMV infection in infants has an unexpected effect on the white blood cells
White blood cells, also called leukocytes or leucocytes, are the cells of the immune system that are involved in protecting the body against both infectious disease and foreign invaders. All white blood cells are produced and derived from mult ...
of the immune system
The immune system is a network of biological processes that protects an organism from diseases. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splint ...
causing them to prematurely age. This leads to a reduced immune response
An immune response is a reaction which occurs within an organism for the purpose of defending against foreign invaders. These invaders include a wide variety of different microorganisms including viruses, bacteria, parasites, and fungi which could ...
similar to that found in the elderly.
HSV
Herpes simplex virus (HSV) can infect the infant during birth. Most women with HVSgenital herpes
Genital herpes is an infection by the herpes simplex virus (HSV) of the genitals. Most people either have no or mild symptoms and thus do not know they are infected. When symptoms do occur, they typically include small blisters that break open ...
develop asymptomatic infection during pregnancy. HVS inoculation from mother to fetus has a high likelihood of occurring. Mothers who are treated with antiviral prophylaxis are less prone to have an active, symptomatic case at the time of birth. Mothers who have received prophylactic antiviral medication
Antiviral drugs are a class of medication used for treating viral infections. Most antivirals target specific viruses, while a broad-spectrum antiviral is effective against a wide range of viruses. Unlike most antibiotics, antiviral drugs do ...
have been shown to be less likely to require a cesarean section. At delivery, mothers treated with antiviral medication are less likely to have a viral shedding at the time of birth.
Zika
Zika fever
Zika fever, also known as Zika virus disease or simply Zika, is an infectious disease caused by the Zika virus. Most cases have no symptoms, but when present they are usually mild and can resemble dengue fever. Symptoms may include fever, red ...
is caused by a virus that is acquired by the mother and then transmitted to the infant in utero. The CDC is concerned with the potential that this viral infection may cause microcephaly
Microcephaly (from New Latin ''microcephalia'', from Ancient Greek μικρός ''mikrós'' "small" and κεφαλή ''kephalé'' "head") is a medical condition involving a smaller-than-normal head. Microcephaly may be present at birth or it ...
in newborns.
Rubella
Congential rubella is still a risk with higher risk among immigrant women from countries without adequate vaccination programs.Hepatitis
There are five liver specific viruses (hepatitis A, B, C, D, E) that could potentially harm the mother and child. Acute hepatitis A virus or acute hepatitis E virus infection present the greatest risk to maternal and fetal health and increased risk of adverse pregnancy outcomes. Hepatitis B, C and D virus present a risk of mother to child transmission but are dependent on the severity of the underlying disease in the mother. However, hepatitis B virus is the major cause of neonatal infection. *Hepatitis A
Hepatitis A is an infectious disease of the liver caused by ''Hepatovirus A'' (HAV); it is a type of viral hepatitis. Many cases have few or no symptoms, especially in the young. The time between infection and symptoms, in those who develop them ...
is a non-enveloped, single-stranded RNA virus that is spread through the fecal-oral route with the main modes of transmission being close personal contact or ingestion of contaminated food or water. During pregnancy, hepatitis A can cause placental abruption, premature rupture of membranes, and increased rates of preterm labor.
* Hepatitis B
Hepatitis B is an infectious disease caused by the '' Hepatitis B virus'' (HBV) that affects the liver; it is a type of viral hepatitis. It can cause both acute and chronic infection.
Many people have no symptoms during an initial infection. ...
is an enveloped, double stranded DNA virus that is spread by exposure to blood, with the main modes of transmission are blood, sexual transmission, or perinatal. During pregnancy, acute hepatitis B infection can result in increased risk of preterm delivery, low birth weight and increased risk of gestational diabetes mellitus. Chronic hepatitis B infection is the largest concern globally. Chronic hepatitis B infection can lead to acute liver failure and increased alanine aminotransferase flares. There is also an increased risk of mother to child transmission and occurs during the delivery of the neonate from an infected mother. Some infected neonates will develop acute hepatitis B and symptomatically will develop abdominal distension, jaundice, clay- colored stools and failure to thrive. However, most infected neonates will be asymptomatic but will chronically have persistent hepatitis B surface antigens in the blood and elevated transaminase levels.
* Hepatitis C is an enveloped, single stranded RNA virus that is spread by exposure to blood, with the main modes of transmission are blood, sexual transmission, or perinatal. Chronic infection with hepatitis C virus may influence pregnancy outcomes, such as increased rates of small for gestational age, intrauterine death, low birthweight, and preterm delivery, but no clear association between these adverse outcomes and hepatitis C infection have been observed. There is also an increased risk of mother to child transmission and is largely attributable to events during the birth process.
* Hepatitis D
Hepatitis D is a type of viral hepatitis caused by the hepatitis delta virus (HDV). HDV is one of five known hepatitis viruses: A, B, C, D, and E. HDV is considered to be a satellite (a type of subviral agent) because it can propagate only in ...
is a single stranded RNA virus that is spread by exposure to blood, with the main modes of transmission are blood, sexual transmission, or perinatal. There is limited research on the effects of hepatitis D infection on fetal or infant outcomes, but the effects are thought to be similar to those with hepatitis B infection.
* Hepatitis E
Hepatitis E is inflammation of the liver caused by infection with the hepatitis E virus (HEV); it is a type of viral hepatitis. Hepatitis E has mainly a fecal-oral transmission route that is similar to hepatitis A, although the viruses are unrel ...
is a non-enveloped, single stranded RNA virus that is spread through the fecal-oral route with the main modes of transmission being close personal contact or ingestion of contaminated food or water. During pregnancy, acute hepatitis E infections result in an increase in adverse pregnancy outcomes such as increased maternal and fetal morbidity and mortality, acute hepatic failure, and associated complications with preterm birth.
Other
Other viral infections such asrespiratory syncytial virus
Respiratory syncytial virus (RSV), also called human respiratory syncytial virus (hRSV) and human orthopneumovirus, is a common, contagious virus that causes infections of the respiratory tract. It is a negative-sense, single-stranded RNA virus. ...
(RSV), metapneumovirus (hMPV), rhinovirus
The rhinovirus (from the grc, ῥίς, rhis "nose", , romanized: "of the nose", and the la, vīrus) is the most common viral infectious agent in humans and is the predominant cause of the common cold. Rhinovirus infection proliferates in tem ...
, parainfluenza
Human parainfluenza viruses (HPIVs) are the viruses that cause human parainfluenza. HPIVs are a paraphyletic group of four distinct single-stranded RNA viruses belonging to the '' Paramyxoviridae'' family. These viruses are closely associated wit ...
(PIV), and human coronavirus in the neonatal period are associated with recurrent wheezing
A wheeze is a continuous, coarse, whistling sound produced in the respiratory airways during breathing. For wheezes to occur, some part of the respiratory tree must be narrowed or obstructed (for example narrowing of the lower respiratory tract ...
in later childhood. RSV infections can be prolonged. Premature infants born at less than 32 weeks gestation have more days of cough and wheeze at 1 year of age than those uninfected with RSV.
Fungi
In very low birth weight infants (VLBWI), systemic fungus infection is ahospital-acquired infection
A hospital-acquired infection, also known as a nosocomial infection (from the Greek , meaning "hospital"), is an infection that is acquired in a hospital or other health care facility. To emphasize both hospital and nonhospital settings, it is so ...
with serious consequences. The pathogens are usually ''Candida albicans
''Candida albicans'' is an opportunistic pathogenic yeast that is a common member of the human gut flora. It can also survive outside the human body. It is detected in the gastrointestinal tract and mouth in 40–60% of healthy adults. It is usu ...
'' and ''Candida parapsilosis
''Candida parapsilosis'' is a fungal species of yeast that has become a significant cause of sepsis and of wound and tissue infections in immunocompromised people. Unlike ''Candida albicans'' and '' Candida tropicalis'', ''C. parapsilosis'' is no ...
''. A small percentage of fungal infections are caused by ''Aspergillus
' () is a genus consisting of several hundred mold species found in various climates worldwide.
''Aspergillus'' was first catalogued in 1729 by the Italian priest and biologist Pier Antonio Micheli. Viewing the fungi under a microscope, Mic ...
'', ''Zygomycetes
Zygomycota, or zygote fungi, is a former phylum, division or phylum of the kingdom Fungi. The members are now part of two Phylum, phyla: the Mucoromycota and Zoopagomycotina, Zoopagomycota. Approximately 1060 species are known. They are mostly t ...
'', ''Malassezia
''Malassezia'' (formerly known as ''Pityrosporum'') is a genus of fungi. It is the sole genus in family Malasseziaceae, which is the only family in order Malasseziales, itself the single member of class Malasseziomycetes. ''Malassezia'' species a ...
'', and '' Trichosporon''. Infection is usually late-onset. Up to 9% of VLBWI with birth weights of <1,000 g develop these fungus infections leading to sepsis or meningitis. As many as one-third of these infants can die. Candidiasis is associated with retinopathy, prematurity and negative neurodevelopmental consequences. Candida can colonize the gastrointestinal tract of low birthweight infants (LBI). This gastrointestinal colonization is often a precursor to a more serious invasive infection. The risk of serious candida infection increases when multiple factors are present. These are: thrombocytopenia
Thrombocytopenia is a condition characterized by abnormally low levels of platelets, also known as thrombocytes, in the blood. It is the most common coagulation disorder among intensive care patients and is seen in a fifth of medical patients a ...
, the presence of candidal dermatitis, the use of systemic steroids, birth weights of <1,000 g, presence of a central catheter, postponing enteral feeding, vaginal delivery, and the amount of time broad-spectrum antibiotics were given.
Protozoans
Infants born withmalaria
Malaria is a mosquito-borne infectious disease that affects humans and other animals. Malaria causes symptoms that typically include fever, tiredness, vomiting, and headaches. In severe cases, it can cause jaundice, seizures, coma, or death. S ...
can be infected with a variety of species
In biology, a species is the basic unit of classification and a taxonomic rank of an organism, as well as a unit of biodiversity. A species is often defined as the largest group of organisms in which any two individuals of the appropriate s ...
; ''Plasmodium vivax
''Plasmodium vivax'' is a protozoal parasite and a human pathogen. This parasite is the most frequent and widely distributed cause of recurring malaria. Although it is less virulent than ''Plasmodium falciparum'', the deadliest of the five huma ...
'', ''Plasmodium malariae
''Plasmodium malariae'' is a parasitic protozoan that causes malaria in humans. It is one of several species of ''Plasmodium'' parasites that infect other organisms as pathogens, also including ''Plasmodium falciparum'' and ''Plasmodium vivax'' ...
'', ''Plasmodium ovale
''Plasmodium ovale'' is a species of parasitic protozoon that causes tertian malaria in humans. It is one of several species of ''Plasmodium'' parasites that infect humans, including ''Plasmodium falciparum'' and ''Plasmodium vivax'' which are ...
'', and '' Plasmodium falciparum''. In most instances of congenital malaria is caused by ''P. vivax'' and ''P. falciparum''. Women living in areas where malaria is prevalent and common are repeatedly exposed to malaria. In response to maternal infection, mothers develop antimalarial antibodies. It is probable that the antibodies present in the mother offers protection for the baby. Bacterial infection can develop with malaria.
Infants that are infected by the protozoan ''Toxoplasma gondii
''Toxoplasma gondii'' () is an obligate intracellular parasitic protozoan (specifically an apicomplexan) that causes toxoplasmosis. Found worldwide, ''T. gondii'' is capable of infecting virtually all warm-blooded animals, but felids, such as d ...
'' in utero can be born with chorioretinitis
Chorioretinitis is an inflammation of the choroid (thin pigmented vascular coat of the eye) and retina of the eye. It is a form of posterior uveitis. If only the choroid is inflamed, not the retina, the condition is termed choroiditis. The ophtha ...
or ocular toxoplasmosis. Globally, it is the most common cause of infections of the back of the eye. (posterior segment). The most common sign is decreased vision in one eye. Other signs and symptoms may appear after the neonatal period and include: chorioretinitis development later in life, intracranial calcification hydrocephalus or central nervous system abnormalities.
Risk factors
Risk factors are those conditions which increase the likelihood that an infant will be born with or develop an infection. The risk for developing catheter-related infections is offset by the increased survival rate of premature infants that have early onset sepsis. Intravenous administration of prophylactic immunoglobin enhances immunity of the immature infant and is used for treatment.Mechanism
 Inflammation accompanies infection and is likely to complicate treatment and recovery. Inflammation is linked to reduced growth of the lungs of the premature baby.
Inflammation accompanies infection and is likely to complicate treatment and recovery. Inflammation is linked to reduced growth of the lungs of the premature baby.
Pathogenesis
The recent identification of the presence ofmicroorganism
A microorganism, or microbe,, ''mikros'', "small") and ''organism'' from the el, ὀργανισμός, ''organismós'', "organism"). It is usually written as a single word but is sometimes hyphenated (''micro-organism''), especially in olde ...
s in maternal-infant body fluids that were previously thought to be sterile has provided one explanation for the presence of the inflammatory response in both the mother and infant. Sixty-one percent of pregnant women with chorioamnionitis, or inflammation of the amniotic fluid, were found to be infected by microorganisms. Often, more than one pathogen was present. In fifteen percent of pregnant women inflammation was still evident even though there was no evidence of pathogens. This may indicate that there are other causes. A high percentage, 51% to 62%, of pregnant women who had chorioamnionitis also had inflammation of the placenta
The placenta is a temporary embryonic and later fetal organ that begins developing from the blastocyst shortly after implantation. It plays critical roles in facilitating nutrient, gas and waste exchange between the physically separate mate ...
.
Diagnosis
Diagnosis
Diagnosis is the identification of the nature and cause of a certain phenomenon. Diagnosis is used in many different disciplines, with variations in the use of logic, analytics, and experience, to determine " cause and effect". In systems engin ...
of infection is based upon the recovery of the pathogen or pathogens from the typically sterile sites in the mother or the baby. Unfortunately, as many half of pregnant women are asymptomatic with a gonorrhea infection and other sexually transmitted infections. Access provided by the University of Pittsburgh. Samples are obtained from urine
Urine is a liquid by-product of metabolism in humans and in many other animals. Urine flows from the kidneys through the ureters to the urinary bladder. Urination results in urine being excreted from the body through the urethra.
Cellular ...
, blood
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells. Blood in the cir ...
or cerebrospinal fluid
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found within the tissue that surrounds the brain and spinal cord of all vertebrates.
CSF is produced by specialised ependymal cells in the choroid plexus of the ventricles of the ...
. Diagnosis of infection can also be aided by the use of more nonspecific tests such as determining the total white blood cell count, cytokine
Cytokines are a broad and loose category of small proteins (~5–25 kDa) important in cell signaling. Cytokines are peptides and cannot cross the lipid bilayer of cells to enter the cytoplasm. Cytokines have been shown to be involved in autocrin ...
levels and other blood tests and signs.
Viral infection
Symptoms and the isolation of the virus pathogen the upper respiratory tract is diagnostic. Virus identification is specific immunologic methods and PCR. The presence of the virus can be rapidly confirmed by the detection of the virus antigen. The methods and materials used for identifying the RSV virus has a specificity and sensitivity approaching 85% to 95%. Not all studies confirm this sensitivity. Antigen detection has comparatively lower sensitivity rates that approach 65% to 75%.Protozoan infection
Congential malaria has its own set of signs:Neonatal sepsis
Neonatal sepsis of the newborn is an infection that has spread through the entire body. The inflammatory response to this systematic infection can be as serious as the infection itself. In infants that weigh under 1500 g,sepsis
Sepsis, formerly known as septicemia (septicaemia in British English) or blood poisoning, is a life-threatening condition that arises when the body's response to infection causes injury to its own tissues and organs. This initial stage is follo ...
is the most common cause of death. Three to four percent of infants per 1000 births contract sepsis. The mortality rate from sepsis is near 25%. Infected sepsis in an infant can be identified by culturing the blood and spinal fluid and if suspected, intravenous antibiotics are usually started. Lumbar puncture is controversial because in some cases it has found not to be necessary while concurrently, without it estimates of missing up to one third of infants with meningitis is predicted.
Prevention
To reduce neonatal infection, screening of pregnant women for HIV, hepatitis B, and syphilis, is available in the UK and the United States. Treatment with an vaginal antibiotic wash prior to birth does not prevent infection with group B streptococcus bacteria (GBS). Treatment with vaginal chlorhexidine prior to birth does not prevent neonatal infections. Because GBS bacteria can colonize the lower reproductive tract of 30% of women, typically pregnant women are tested for this pathogen from 35 to 37 weeks of pregnancy. Before delivery treatment of the mother with antibiotics reduces the rate of neonatal infection. Prevention of the infection of the baby is done by treating the mother with penicillin. Since the adoption of this prophylactic treatment, infant mortality from GBS infection has decreased by 80%. Mothers with symptomaticgenital herpes
Genital herpes is an infection by the herpes simplex virus (HSV) of the genitals. Most people either have no or mild symptoms and thus do not know they are infected. When symptoms do occur, they typically include small blisters that break open ...
and who are treated with antiviral prophylaxis are less prone to have an active, symptomatic case at the time of birth and it may be able to reduce the risk of passing on HSV during birth. Cesarean delivery reduces the risk of infection of the infant.
Breastfeeding
Breastfeeding, or nursing, is the process by which human breast milk is fed to a child. Breast milk may be from the breast, or may be expressed by hand or pumped and fed to the infant. The World Health Organization (WHO) recommends that br ...
has been shown to protect the neonate from some infections. . Breast milk protects against necrotizing enterocolitis
Enterocolitis is an inflammation of the digestive tract, involving enteritis of the small intestine and colitis of the colon. It may be caused by various infections, with bacteria, viruses, fungi, parasites, or other causes. Common clinical manif ...
.
Hepatitis A, B, and D are preventable via vaccination. Hepatitis A can be prevented by immunization against HAV, while Hepatitis B and D can both be prevented by immunization against HBV.
The Hepatitis B vaccination is among the first two injections a newborn receives. The other is vitamin K to prevent vitamin K deficiency bleeding. In cases where the pregnant woman is positive for hepatitis B, the baby will receive an additional dose of hepatitis B immunoglobulin in addition to the hepatitis B vaccine to prevent transmission of the disease from mother to baby. Prevention of hepatitis B is crucial as there is no currently no cure.
In general, in an unvaccinated individual, the viral hepatitis family causes liver damage due to a cell-mediated response via cytotoxic lymphocytes. Acute viral hepatitis can lead to jaundice, elevated bilirubin, elevated liver enzymes (aspartate transaminase, alanine transaminase, and alkaline phosphatase), and flu-like symptoms. Importantly, acute neonatal HBV infections progress to chronic Hepatitis B about 90% of the time. For both Hepatitis B and Hepatitis D, the most significant long-term complications are progression to cirrhosis and hepatocellular carcinoma, which carries high morbidity and mortality.
Treatment
Neonatal infection treatment is typically started before the diagnosis of the cause can be confirmed. Neonatal infection can be prophylactically treated with antibiotics. Maternal treatment with antibiotics is primarily used to protect against group B streptococcus. Women with a history ofgenital herpes
Genital herpes is an infection by the herpes simplex virus (HSV) of the genitals. Most people either have no or mild symptoms and thus do not know they are infected. When symptoms do occur, they typically include small blisters that break open ...
, can be treated with antiviral drugs to prevent symptomatic lesions and viral shedding that could infect the infant at birth. The antiviral medications used include acyclovir, penciclovir, valacyclovir, and famciclovir. Only very small amounts of the drug can be detected in the fetus. There are no increases in drug-related abnormalities in the infant that could be attributed to acyclovir. Long-term effects of antiviral medications have not been evaluated for their effects after growth and development of the child occurs. Neutropenia can be a complication of acyclovir treatment of neonatal HSV infection, but is usually transient. Treatment with immunoglobulin therapy has not been proven to be effective and is not recommended.
Epidemiology
Up to 3.3 million newborns die each year and 23.4% of these die of neonatal infection. About half of the deaths caused by sepsis or pneumonia happen in the first week postpartum. In industrialized countries, prophylactic antibiotic treatment of the mothers identified with group B streptococcus, early identification of sepsis in the newborn, and administration of antibiotics to the newborn has reduced mortality. Neonatal herpes in North America is estimated to be from 5 – 80 per 100,000 live births. HSV has a lower prevalence in mothers outside the United States. In the United Kingdom the incidence is much lower and estimated to be 1.6 per 100,000 live births. Approximately 70% to 80% of infected infants are born to mothers with no reported history of HSV infection. Regions with low neonatal mortality include Europe, the Western Pacific, and the Americas, which have sepsis rates that account for 9.1% to 15.3% of the total neonatal deaths worldwide. This is in contrast with the 22.5 to 27.2% percentage of total deaths in resource-poor countries such as Nigeria, the Democratic Republic of the Congo, India, Pakistan, and China. In the UK, the proportions of pregnant women who are newly screened positive for hepatitis B, syphilis, and HIV have remained constant since 2010 at about 0.4%, 0.14% and 0.15%, respectively. Estimated prevalence levels among pregnant women for hepatitis B and HIV, including previous diagnoses, were higher at 0.67% and 0.27%. Pregnant women evaluated as susceptible to rubella due to low antibody levels have increased by over 60%, to about 7.2%. However, this increase is probably due to changes in testing methods and evaluation criteria. In North America, prior to the 1950s, group A β-hemolytic streptococcus (GAS) was the most common pathogen associated with neonatal sepsis prior to the 1960s. In the past twenty years, the most common pathogen causing sepsis is coagulase-negative staphylococci that exist as biofilms associated with infected central venous or arterial catheters. Infections can be fatal and contribute to long-term morbidity and disability among the infants who survive into childhood. Neonatal sepsis effects 128 cases per 1000 live births. Meningitis can occur in the septic infant. Expectant mothers with HSV have a 75% chance of at least one flare-up during their pregnancy. In limited studies it was found that infants in Africa born to mothers with malaria have a 7% of acquiring congenital malaria.Early-onset infections
Early onset sepsis can occur in the first week of life. It usually is apparent on the first day after birth. This type of infection is usually acquired before the birth of the infant. Premature rupture of membranes and other obstetrical complications can add to the risk of early-onset sepsis. If the amniotic membrane has been ruptured greater than 18 hours before delivery the infant may be at more risk for this complication. Prematurity, low birth weight, chorioamnionitis, maternal urinary tract infection and/or maternal fever are complications that increase the risk for early-onset sepsis. Early onset sepsis is indicated by serious respiratory symptoms. The infant usually develops pneumonia, hypothermia, or shock. The mortality rate is 30 to 50%.Late onset infections
Infections that occur after the first week of life but before the age of 30 days are considered late onset infections. Obstetrical and maternal complications are not typically the cause of these late onset infections; they are usually acquired by the infant in the hospital neonatal intensive care unit. The widespread use of broad-spectrum antibiotics in the nursery intensive care unit can cause a higher prevalence of invasive antibiotic resistant bacteria. Meconium aspiration syndrome has a mortality rate just over 4%. This accounts for 2% for all neonatal deaths.Research
The susceptibility to risk of infection and immune deficiencies are active areas of research. Studies regarding the role of viruses in neonatal infections are lacking. Research also continues into the role and protective effect of gut, skin and other human microbiomes and the colonization during the neonatal period. The comparison between both resource rich and poor countries makes it difficult to compare the diagnosis success; as industrialized regions are able to confirm the diagnosis and presence of pathogens in the clinical laboratory. Clinical testing may not be available in all settings and clinicians must rely on the signs of infection in the newborn. Research data from Africa and Southeast Asia is scarce. The result of some research has been the identification of diagnostic tools and procedures that could identify mothers with group B streptococcus infection in resource-poor regions. These procedures would be easy and inexpensive to use. Those mothers who are identified as being infected could then be prophylactly treated prior to the birth of the baby. Probiotic administration of Lactobacillus species has shown some success. A GBS vaccine is currently being tested but not currently available. Vaccination is estimated to being able to prevent 4% of GBS infections for preterm births and 60–70% for neonatal GBS infections in the US. The projected benefits of maternal vaccination is the prevention of 899 cases of GBS disease and 35 deaths among infants. The cost savings in the prevention of GBS may be over 43 million dollars. Vaccination may be especially beneficial in low to middle income countries where screening and prophylactic treatment is not possible. Analysts project that GBS vaccination would prevent 30–54% of infant GBS cases. Screening, prophylactic antibiotics and vaccine would prevent 48% of infection.See also
*Preterm birth
Preterm birth, also known as premature birth, is the birth of a baby at fewer than 37 weeks gestational age, as opposed to full-term delivery at approximately 40 weeks. Extreme preterm is less than 28 weeks, very early preterm birth is between 2 ...
References
Further reading
* *External links
* {{Diseases of maternal transmission Infections specific to the perinatal period Neonatology Infectious diseases Streptococcal infections Viral diseases Human pregnancy HIV/AIDS Sexually transmitted diseases and infections