Nanofiltration on:
[Wikipedia]
[Google]
[Amazon]
Nanofiltration is a membrane filtration process used most often to soften and disinfect water.
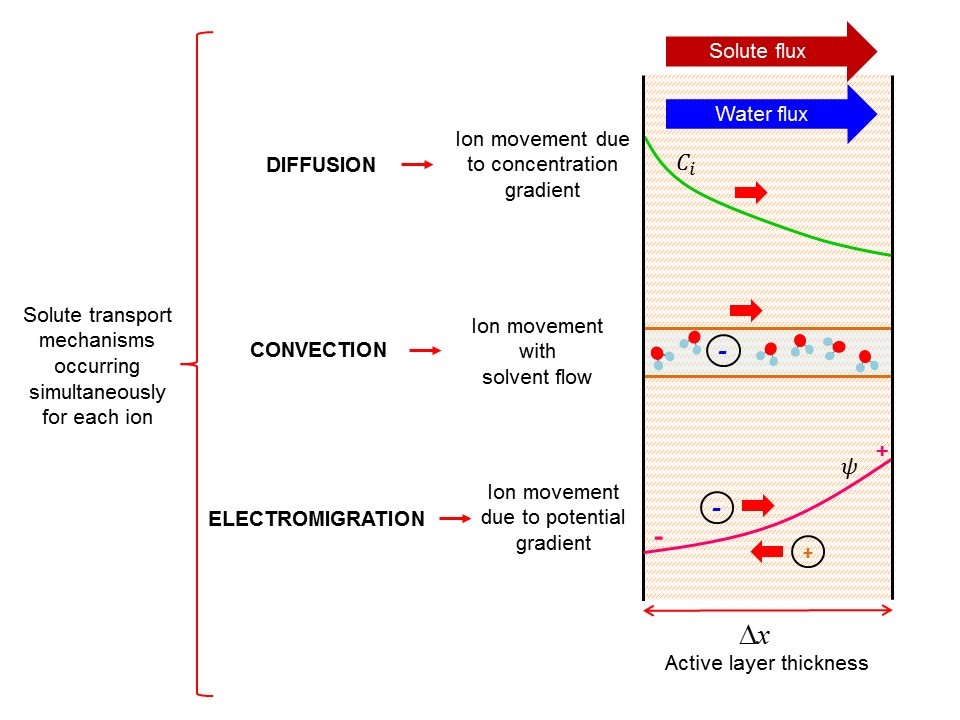 Unlike membranes with larger and smaller pore sizes, passage of solutes through nanofiltration is significantly more complex.
Because of the pore sizes, there are three modes of transport of solutes through the membrane. These include 1) diffusion (molecule travel due to concentration potential gradients, as seen through reverse osmosis membranes), 2) convection (travel with flow, like in larger pore size filtration such as microfiltration), and 3) electromigration (attraction or repulsion from charges within and near the membrane).
Additionally, the exclusion mechanisms in nanofiltration are more complex than in other forms of filtration. Most filtration systems operate solely by size (steric) exclusion, but at small length scales seen in nanofiltration, important effects include surface charge and hydration (
Unlike membranes with larger and smaller pore sizes, passage of solutes through nanofiltration is significantly more complex.
Because of the pore sizes, there are three modes of transport of solutes through the membrane. These include 1) diffusion (molecule travel due to concentration potential gradients, as seen through reverse osmosis membranes), 2) convection (travel with flow, like in larger pore size filtration such as microfiltration), and 3) electromigration (attraction or repulsion from charges within and near the membrane).
Additionally, the exclusion mechanisms in nanofiltration are more complex than in other forms of filtration. Most filtration systems operate solely by size (steric) exclusion, but at small length scales seen in nanofiltration, important effects include surface charge and hydration ( The transport and exclusion mechanisms are heavily influenced by membrane pore size, solvent viscosity, membrane thickness, solute diffusivity, solution temperature, solution pH, and membrane dielectric constant. The pore size distribution is also important. Modeling rejection accurately for NF is very challenging. It can be done with applications of the
The transport and exclusion mechanisms are heavily influenced by membrane pore size, solvent viscosity, membrane thickness, solute diffusivity, solution temperature, solution pH, and membrane dielectric constant. The pore size distribution is also important. Modeling rejection accurately for NF is very challenging. It can be done with applications of the
 Because NF permeate is rarely clean enough to be used as the final product for drinking water and other water purification, is it commonly used as a pre treatment step for
Because NF permeate is rarely clean enough to be used as the final product for drinking water and other water purification, is it commonly used as a pre treatment step for
Project ETAP-ERN, that uses renewable energies for desalinization
{{in lang, es
- Hawk's Perch Technical Writing, LLC Nanotechnology Water treatment Filters Water desalination Membrane technology
Overview
Nanofiltration is amembrane filtration
Membrane technology encompasses the scientific processes used in the construction and application of membranes. Membranes are used to facilitate the transport or rejection of substances between mediums, and the mechanical separation of gas and li ...
-based method that uses nanometer
330px, Different lengths as in respect to the molecular scale.
The nanometre (international spelling as used by the International Bureau of Weights and Measures; SI symbol: nm) or nanometer (American and British English spelling differences#-re ...
sized pores through which particles smaller than 10 nanometers pass through the membrane. Nanofiltration membranes have pore sizes from 1-10 nanometers, smaller than that used in microfiltration
Microfiltration is a type of physical filtration process where a contaminated fluid is passed through a special pore-sized membrane filter to separate microorganisms and suspended particles from process liquid. It is commonly used in conjunction ...
and ultrafiltration
Ultrafiltration (UF) is a variety of membrane filtration in which forces such as pressure or concentration gradients lead to a separation through a semipermeable membrane. Suspended solids and solutes of high molecular weight are retained in the s ...
, but a little bit bigger than that in reverse osmosis
Reverse osmosis (RO) is a water purification process that uses a partially permeable membrane to separate ions, unwanted molecules and larger particles from drinking water. In reverse osmosis, an applied pressure is used to overcome osmotic pre ...
. Membranes used are predominantly created from polymer thin films. Materials that are commonly used include polyethylene terephthalate
Polyethylene terephthalate (or poly(ethylene terephthalate), PET, PETE, or the obsolete PETP or PET-P), is the most common thermoplastic polymer resin of the polyester family and is used in fibres for clothing, containers for liquids and foods ...
or metals such as aluminum
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. It has ...
. Pore dimensions are controlled by pH, temperature and time during development with pore densities ranging from 1 to 106 pores per cm2.
Membranes made from polyethylene terephthalate and other similar materials, are referred to as "track-etch" membranes, named after the way the pores on the membranes are made. "Tracking" involves bombarding the polymer thin film with high energy particles. This results in making tracks that are chemically developed into the membrane, or "etched" into the membrane, which are the pores.
Membranes created from metal such as alumina membranes, are made by electrochemically growing a thin layer of aluminum oxide from aluminum metal in an acidic medium.
Range of applications
Historically, nanofiltration and other membrane technology used for molecular separation was applied entirely onaqueous
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, or sodium chloride (NaCl), in water would be rep ...
systems. The original uses for nanofiltration were water treatment and in particular water softening
Water softening is the removal of calcium, magnesium, and certain other metal cations in hard water. The resulting soft water requires less soap for the same cleaning effort, as soap is not wasted bonding with calcium ions. Soft water also exten ...
. Nanofilters "soften" water by retaining scale-forming divalent ions (e.g. Ca2+, Mg2+).
Nanofiltration has been extended into other industries such as milk and juice production as well as pharmaceuticals
A medication (also called medicament, medicine, pharmaceutical drug, medicinal drug or simply drug) is a drug used to diagnose, cure, treat, or prevent disease. Drug therapy (pharmacotherapy) is an important part of the medical field and rel ...
, fine chemicals, and flavour and fragrance industries.
Advantages and disadvantages
One of the main advantages of nanofiltration as a method of softening water is that during the process of retainingcalcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to ...
and magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic ta ...
ions while passing smaller hydrated monovalent ions, filtration is performed without adding extra sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable iso ...
ions, as used in ion exchangers. Many separation processes do not operate at room temperature (e.g. distillation
Distillation, or classical distillation, is the process of separation process, separating the components or substances from a liquid mixture by using selective boiling and condensation, usually inside an apparatus known as a still. Dry distilla ...
), which greatly increases the cost of the process when continuous heating or cooling is applied. Performing gentle molecular separation is linked with nanofiltration that is often not included with other forms of separation processes (centrifugation
Centrifugation is a mechanical process which involves the use of the centrifugal force to separate particles from a solution according to their size, shape, density, medium viscosity and rotor speed. The denser components of the mixture migrate ...
). These are two of the main benefits that are associated with nanofiltration.
Nanofiltration has a very favorable benefit of being able to process large volumes and continuously produce streams of products. Still, Nanofiltration is the least used method of membrane filtration in industry as the membrane pores sizes are limited to only a few nanometers. Anything smaller, reverse osmosis is used and anything larger is used for ultrafiltration. Ultrafiltration can also be used in cases where nanofiltration can be used, due to it being more conventional.
A main disadvantage associated with nanotechnology, as with all membrane filter technology, is the cost and maintenance of the membranes used. Nanofiltration membranes are an expensive part of the process. Repairs and replacement of membranes is dependent on total dissolved solids, flow rate and components of the feed. With nanofiltration being used across various industries, only an estimation of replacement frequency can be used. This causes nanofilters to be replaced a short time before or after their prime usage is complete.
Design and operation
Industrial applications of membranes require hundreds to thousands of square meters of membranes and therefore an efficient way to reduce the footprint by packing them is required. Membranes first became commercially viable when low cost methods of housing in 'modules' were achieved. Membranes are not self-supporting. They need to be stayed by a porous support that can withstand the pressures required to operate the NF membrane without hindering the performance of the membrane. To do this effectively, the module needs to provide a channel to remove the membranepermeation
In physics and engineering, permeation (also called imbuing) is the penetration of a permeate (a fluid such as a liquid, gas, or vapor) through a solid. It is directly related to the concentration gradient of the permeate, a material's intrinsic ...
and provide appropriate flow condition that reduces the phenomena of concentration polarisation. A good design minimises pressure losses on both the feed side and permeate side and thus energy requirements.
Concentration polarisation
Concentration polarization describes the accumulation of the species being retained close to the surface of the membrane which reduces separation capabilities. It occurs because the particles are convected towards the membrane with the solvent and its magnitude is the balance between this convection caused by solventflux
Flux describes any effect that appears to pass or travel (whether it actually moves or not) through a surface or substance. Flux is a concept in applied mathematics and vector calculus which has many applications to physics. For transport ph ...
and the particle transport away from the membrane due to the concentration gradient (predominantly caused by diffusion
Diffusion is the net movement of anything (for example, atoms, ions, molecules, energy) generally from a region of higher concentration to a region of lower concentration. Diffusion is driven by a gradient in Gibbs free energy or chemical p ...
.) Although concentration polarization is easily reversible, it can lead to fouling
Fouling is the accumulation of unwanted material on solid surfaces. The fouling materials can consist of either living organisms (biofouling) or a non-living substance (inorganic or organic). Fouling is usually distinguished from other surf ...
of the membrane.
Spiral wound module
Spiral wound modules are the most commonly used style of module and are 'standardized' design, available in a range of standard diameters (2.5", 4" and 8") to fit standardpressure vessel
A pressure vessel is a container designed to hold gases or liquids at a pressure substantially different from the ambient pressure.
Construction methods and materials may be chosen to suit the pressure application, and will depend on the size o ...
that can hold several modules in series connected by O-rings. The module uses flat sheets wrapped around a central tube. The membranes are glued along three edges over a permeate spacer to form 'leaves'. The permeate spacer supports the membrane and conducts the permeate to the central permeate tube. Between each leaf, a mesh like feed spacer is inserted. The reason for the mesh like dimension of the spacer is to provide a hydrodynamic
In physics and engineering, fluid dynamics is a subdiscipline of fluid mechanics that describes the flow of fluids—liquids and gases. It has several subdisciplines, including ''aerodynamics'' (the study of air and other gases in motion) and ...
environment near the surface of the membrane that discourages concentration polarisation. Once the leaves have been wound around the central tube, the module is wrapped in a casing layer and caps placed on the end of the cylinder to prevent 'telescoping' that can occur in high flow rate and pressure conditions.
Tubular module
Tubular modules look similar toshell and tube heat exchanger
A shell and tube heat exchanger is a class of heat exchanger designs. It is the most common type of heat exchanger in oil refineries and other large chemical processes, and is suited for higher-pressure applications. As its name implies, this ty ...
s with bundles of tubes with the active surface of the membrane on the inside. Flow through the tubes is normally turbulent
In fluid dynamics, turbulence or turbulent flow is fluid motion characterized by chaotic changes in pressure and flow velocity. It is in contrast to a laminar flow, which occurs when a fluid flows in parallel layers, with no disruption between t ...
, ensuring low concentration polarisation but also increasing energy costs. The tubes can either be self-supporting or supported by insertion into perforated metal tubes. This module design is limited for nanofiltration by the pressure they can withstand before bursting, limiting the maximum flux possible. Due to both the high energy operating costs of turbulent flow and the limiting burst pressure, tubular modules are more suited to 'dirty' applications where feeds have particulates such as filtering raw water to gain potable water in the Fyne process. The membranes can be easily cleaned through a 'pigging
In pipeline transportation, pigging is the practice of using pipeline inspection gauges or gadgets, devices generally referred to as pigs or scrapers, to perform various maintenance operations. This is done without stopping the flow of the pr ...
' technique with foam balls are squeezed through the tubes, scouring the caked deposits.
Flux enhancing strategies
These strategies work to reduce the magnitude of concentration polarisation and fouling. There is a range of techniques available however the most common is feed channel spacers as described in spiral wound modules. All of the strategies work by increasingeddies
In fluid dynamics, an eddy is the swirling of a fluid and the reverse current created when the fluid is in a turbulent flow regime. The moving fluid creates a space devoid of downstream-flowing fluid on the downstream side of the object. Fluid beh ...
and generating a high shear
Shear may refer to:
Textile production
*Animal shearing, the collection of wool from various species
**Sheep shearing
*The removal of nap during wool cloth production
Science and technology Engineering
*Shear strength (soil), the shear strength ...
in the flow near the membrane surface. Some of these strategies include vibrating the membrane, rotating the membrane, having a rotor disk above the membrane, pulsing the feed flow rate and introducing gas bubbling close to the surface of the membrane.
Characterisation
Performance parameters
Retention of both charged and unchargedsolutes
In chemistry, a solution is a special type of homogeneous mixture composed of two or more substances. In such a mixture, a solute is a substance dissolved in another substance, known as a solvent. If the attractive forces between the solvent ...
and permeation
In physics and engineering, permeation (also called imbuing) is the penetration of a permeate (a fluid such as a liquid, gas, or vapor) through a solid. It is directly related to the concentration gradient of the permeate, a material's intrinsic ...
measurements can be categorised into performance parameters since the performance under natural conditions of a membrane is based on the ratio of solute retained/ permeated through the membrane.
For charged solutes, the ionic distribution of salts near the membrane-solution interface plays an important role in determining the retention characteristic of a membrane. If the charge of the membrane and the composition and concentration of the solution to be filtered is known, the distribution of various salts can be found. This in turn can be combined with the known charge of the membrane and the Gibbs–Donnan effect
The Gibbs–Donnan effect (also known as the Donnan's effect, Donnan law, Donnan equilibrium, or Gibbs–Donnan equilibrium) is a name for the behaviour of charged particles near a semi-permeable membrane that sometimes fail to distribute evenly ...
to predict the retention characteristics for that membrane.
Uncharged solutes cannot be characterised simply by Molecular Weight Cut Off (MWCO,) although in general an increase in molecular weight or solute size leads to an increase in retention. The charge and structure, pH of the solute, influence the retention characteristics.
Morphology parameters
The morphology of a membrane is usually established by microscopy.Atomic force microscopy
Atomic force microscopy (AFM) or scanning force microscopy (SFM) is a very-high-resolution type of scanning probe microscopy (SPM), with demonstrated resolution on the order of fractions of a nanometer, more than 1000 times better than the op ...
(AFM) is one method used to characterise the surface roughness of a membrane by passing a small sharp tip (<100 Ă) across the surface of a membrane and measuring the resulting Van der Waals force
In molecular physics, the van der Waals force is a distance-dependent interaction between atoms or molecules. Unlike ionic or covalent bonds, these attractions do not result from a chemical electronic bond; they are comparatively weak and th ...
between the atoms in the end of the tip and the surface. This is useful as a direct correlation between surface roughness and colloidal fouling has been developed. Correlations also exist between fouling and other morphology parameters, such as hydrophobe
In chemistry, hydrophobicity is the physical property of a molecule that is seemingly repelled from a mass of water (known as a hydrophobe). In contrast, hydrophiles are attracted to water.
Hydrophobic molecules tend to be nonpolar and, th ...
, showing that the more hydrophobic a membrane is, the less prone to fouling it is. See membrane fouling
Membrane fouling is a process whereby a solution or a particle is deposited on a membrane surface or in membrane pores in a processes such as in a membrane bioreactor, reverse osmosis, forward osmosis, membrane distillation, ultrafiltration, microf ...
for more information.
Methods to determine the porosity
Porosity or void fraction is a measure of the void (i.e. "empty") spaces in a material, and is a fraction of the volume of voids over the total volume, between 0 and 1, or as a percentage between 0% and 100%. Strictly speaking, some tests measure ...
of porous membranes have also been found via permporometry, making use of differing vapour pressure
Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases ...
s to characterise the pore size and pore size distribution within the membrane. Initially all pores in the membrane are completely filled with a liquid and as such no permeation of a gas occurs, but after reducing the relative vapour pressure some gaps will start to form within the pores as dictated by the Kelvin equation
The Kelvin equation describes the change in vapour pressure due to a curved liquid–vapor interface, such as the surface of a droplet. The vapor pressure at a convex curved surface is higher than that at a flat surface. The Kelvin equation is de ...
. Polymeric (non-porous) membranes cannot be subjected to this methodology as the condensable vapour should have a negligible interaction within the membrane.
Solute transport and rejection
 Unlike membranes with larger and smaller pore sizes, passage of solutes through nanofiltration is significantly more complex.
Because of the pore sizes, there are three modes of transport of solutes through the membrane. These include 1) diffusion (molecule travel due to concentration potential gradients, as seen through reverse osmosis membranes), 2) convection (travel with flow, like in larger pore size filtration such as microfiltration), and 3) electromigration (attraction or repulsion from charges within and near the membrane).
Additionally, the exclusion mechanisms in nanofiltration are more complex than in other forms of filtration. Most filtration systems operate solely by size (steric) exclusion, but at small length scales seen in nanofiltration, important effects include surface charge and hydration (
Unlike membranes with larger and smaller pore sizes, passage of solutes through nanofiltration is significantly more complex.
Because of the pore sizes, there are three modes of transport of solutes through the membrane. These include 1) diffusion (molecule travel due to concentration potential gradients, as seen through reverse osmosis membranes), 2) convection (travel with flow, like in larger pore size filtration such as microfiltration), and 3) electromigration (attraction or repulsion from charges within and near the membrane).
Additionally, the exclusion mechanisms in nanofiltration are more complex than in other forms of filtration. Most filtration systems operate solely by size (steric) exclusion, but at small length scales seen in nanofiltration, important effects include surface charge and hydration (solvation shell
A solvation shell or solvation sheath is the solvent interface of any chemical compound or biomolecule that constitutes the solute. When the solvent is water it is called a hydration shell or hydration sphere. The number of solvent molecules sur ...
). The exclusion due to hydration is referred to as dielectric exclusion, a reference to the dielectric constants (energy) associated with a particles precense in solution versus within a membrane substrate. Solution pH strongly impacts surface charge, providing a method to understand and better control rejection.
 The transport and exclusion mechanisms are heavily influenced by membrane pore size, solvent viscosity, membrane thickness, solute diffusivity, solution temperature, solution pH, and membrane dielectric constant. The pore size distribution is also important. Modeling rejection accurately for NF is very challenging. It can be done with applications of the
The transport and exclusion mechanisms are heavily influenced by membrane pore size, solvent viscosity, membrane thickness, solute diffusivity, solution temperature, solution pH, and membrane dielectric constant. The pore size distribution is also important. Modeling rejection accurately for NF is very challenging. It can be done with applications of the Nernst–Planck equation
The Nernst–Planck equation is a conservation of mass equation used to describe the motion of a charged chemical species in a fluid medium. It extends Fick's law of diffusion for the case where the diffusing particles are also moved with respect t ...
, although a heavy reliance on fitting parameters to experimental data is usually requried.
In general, charged solutes are much more effectively rejected in NF than uncharged solutes, and multivalent solutes like (valence of 2) experience very high rejection.
Typical figures for industrial applications
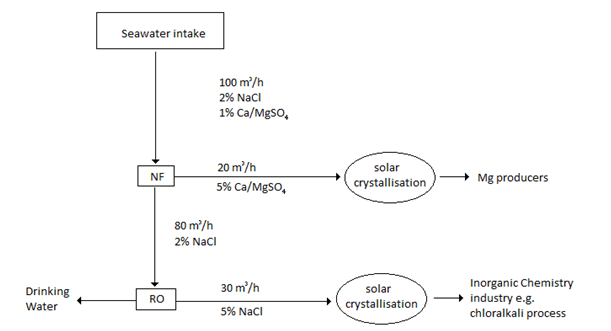
Keeping in mind that NF is usually part of a composite system for purification, a single unit is chosen based on the design specifications for the NF unit. For drinking water purification many commercial membranes exist, coming from chemical families having diverse structures, chemical tolerances and salt rejections. NF units in drinking water purification range from extremely low salt rejection (<5% in 1001A membranes) to almost complete rejection (99% in 8040-TS80-TSA membranes.) Flow rates range from 25–60 m3/day for each unit, so commercial filtration requires multiple NF units in parallel to process large quantities of feed water. The pressures required in these units are generally between 4.5-7.5 bar. For seawaterdesalination
Desalination is a process that takes away mineral components from saline water. More generally, desalination refers to the removal of salts and minerals from a target substance, as in Soil salinity control, soil desalination, which is an issue f ...
using a NF-RO system a typical process is shown below.
 Because NF permeate is rarely clean enough to be used as the final product for drinking water and other water purification, is it commonly used as a pre treatment step for
Because NF permeate is rarely clean enough to be used as the final product for drinking water and other water purification, is it commonly used as a pre treatment step for reverse osmosis
Reverse osmosis (RO) is a water purification process that uses a partially permeable membrane to separate ions, unwanted molecules and larger particles from drinking water. In reverse osmosis, an applied pressure is used to overcome osmotic pre ...
(RO) as is shown above.
Post-treatment
As with other membrane based separations such asultrafiltration
Ultrafiltration (UF) is a variety of membrane filtration in which forces such as pressure or concentration gradients lead to a separation through a semipermeable membrane. Suspended solids and solutes of high molecular weight are retained in the s ...
, microfiltration
Microfiltration is a type of physical filtration process where a contaminated fluid is passed through a special pore-sized membrane filter to separate microorganisms and suspended particles from process liquid. It is commonly used in conjunction ...
and reverse osmosis
Reverse osmosis (RO) is a water purification process that uses a partially permeable membrane to separate ions, unwanted molecules and larger particles from drinking water. In reverse osmosis, an applied pressure is used to overcome osmotic pre ...
, post-treatment of eitherpermeate or retentate flow streams (depending on the application) – is a necessary stage in industrial NF separation prior to commercial distribution of the product. The choice and order of unit operations employed in post-treatment is dependent on water quality regulations and the design of the NF system. Typical NF water purification post-treatment stages include aeration and disinfection & stabilisation.
Aeration
A Polyvinyl chloride (PVC) orfibre-reinforced plastic
Fibre-reinforced plastic (FRP; also called fibre-reinforced polymer, or in American English ''fiber'') is a composite material made of a polymer matrix reinforced with fibres. The fibres are usually glass (in fibreglass), carbon (in carbon-fib ...
(FRP) degasifier is used to remove dissolved gases such as carbon dioxide and hydrogen sulfide from the permeate stream. This is achieved by blowing air in a countercurrent direction to the water falling through packing material in the degasifier. The air effectively strips the unwanted gases from the water.
Disinfection and stabilisation
The permeate water from a NF separation is demineralised and may be disposed to large changes in pH, thus providing a substantial risk of corrosion in piping and other equipment components. To increase the stability of the water, chemical addition of alkaline solutions such as lime and caustic soda is employed. Furthermore, disinfectants such as chlorine or chloroamine are added to the permeate, as well as phosphate or fluoride corrosion inhibitors in some cases.Research trends
Challenges in nanofiltration (NF) technology include minimising membrane fouling and reducing energy requirements. Thin film composite membranes (TFC), which consist of a number of extremely thin selective layers interfacially polymerized over a microporous substrate, have had commercial success in industrial membrane applications. Electrospunnanofibrous membrane layers (ENMs) enhances permeate flux. Energy-efficient alternatives to the commonly used spiral wound arrangement are hollow fibre membranes, which require less pre-treatment.Titanium Dioxide
Titanium dioxide, also known as titanium(IV) oxide or titania , is the inorganic compound with the chemical formula . When used as a pigment, it is called titanium white, Pigment White 6 (PW6), or CI 77891. It is a white solid that is insolubl ...
nanoparticles have been used to minimize for membrane fouling.
See also
* * * * *References
External links
Project ETAP-ERN, that uses renewable energies for desalinization
{{in lang, es
- Hawk's Perch Technical Writing, LLC Nanotechnology Water treatment Filters Water desalination Membrane technology