Michael Faraday on:
[Wikipedia]
[Google]
[Amazon]
File:M Faraday Lab H Moore.jpg, Michael Faraday in his laboratory, c. 1850s.
File:Royal Institution - Michael Faraday's study.jpg , Michael Faraday's study at the Royal Institution.
File:Michael Faradays Flat at Royal Institution.jpg , Michael Faraday's flat at the Royal Institution.
File:Harriett Moore small.jpg, Artist Harriet Jane Moore who documented Faraday's life in watercolours.
 Faraday's books, with the exception of ''Chemical Manipulation'', were collections of scientific papers or transcriptions of lectures. Hamilton, p. 220 Since his death, Faraday's diary has been published, as have several large volumes of his letters and Faraday's journal from his travels with Davy in 1813–1815.
*
Faraday's books, with the exception of ''Chemical Manipulation'', were collections of scientific papers or transcriptions of lectures. Hamilton, p. 220 Since his death, Faraday's diary has been published, as have several large volumes of his letters and Faraday's journal from his travels with Davy in 1813–1815.
*
2nd ed. 18303rd ed. 1842
* ; vol. iii. Richard Taylor and William Francis, 1855 * * * * – published in eight volumes; see also th
2009 publication
of Faraday's diary * * – volume 2, 1993; volume 3, 1996; volume 4, 1999 *
Course of six lectures on the various forces of matter, and their relations to each other
London; Glasgow: R. Griffin, 1860. * The Liquefaction of Gases, Edinburgh: W.F. Clay, 1896.
The letters of Faraday and Schoenbein 1836–1862. With notes, comments and references to contemporary letters
London: Williams & Norgate 1899.
Digital edition
by the University and State Library Düsseldorf)
File:Faraday-1.jpg, Volumes 1-3 of Michael Faraday's "Experimental researches in electricity," from 1839, 1844, and 1855, respectively
File:Faraday-2.jpg, Title page of Volume 1 of Michael Faraday's "Experimental researches in electricity," 1839
File:Faraday-7.jpg, First page of Volume 1 of Michael Faraday's "Experimental researches in electricity," 1839
Biography at The Royal Institution of Great Britain
Faraday as a Discoverer by John Tyndall, Project Gutenberg
(downloads)
The Life and Discoveries of Michael Faraday
by J. A. Crowther, London: Society for Promoting Christian Knowledge, 1920
Complete Correspondence of Michael Faraday
Searchable full texts of all letters to and from Faraday, based on the standard edition by Frank James
Video Podcast
with Sir John Cadogan talking about Benzene since Faraday
The letters of Faraday and Schoenbein 1836–1862. With notes, comments and references to contemporary letters (1899)
full downloa
PDF
Faraday School, located on Trinity Buoy Wharf
at the New Model School Company Limited's website * , Chemical Heritage Foundation {{DEFAULTSORT:Faraday, Michael 1791 births 1867 deaths 18th-century English people 19th-century English scientists 19th-century British physicists 19th-century British chemists Experimental physicists Optical physicists English chemists English inventors English physicists Fellows of the American Academy of Arts and Sciences Fellows of the Royal Society Foreign associates of the National Academy of Sciences Honorary members of the Saint Petersburg Academy of Sciences People from Elephant and Castle People associated with electricity Burials at Highgate Cemetery English Protestants Members of the French Academy of Sciences Members of the Royal Netherlands Academy of Arts and Sciences Members of the Royal Swedish Academy of Sciences Recipients of the Copley Medal Recipients of the Pour le Mérite (civil class) Royal Medal winners Glasites Writers about religion and science Magneticians
Michael Faraday (; 22 September 1791 – 25 August 1867) was an English scientist who contributed to the study of electromagnetism and electrochemistry. His main discoveries include the principles underlying electromagnetic induction,
_Volume_1">James_Clerk_Maxwell">The_Scientific_Papers_of_James_Clerk_Maxwell
_Volume_1p._360;_Courier_Dover_2003,__The_International_System_of_Units.html" ;"title="James_Clerk_Maxwell
_Volume_1.html" ;"title="James Clerk Maxwell">The Scientific Papers of James Clerk Maxwell
Volume 1">James Clerk Maxwell">The Scientific Papers of James Clerk Maxwell
Volume 1p. 360; Courier Dover 2003, The International System of Units">SI unit of capacitance is named in his honour: the farad. Albert Einstein kept a picture of Faraday on his study wall, alongside pictures of Arthur Schopenhauer and James Clerk Maxwell. Physicist Ernest Rutherford stated, "When we consider the magnitude and extent of his discoveries and their influence on the progress of science and of industry, there is no honour too great to pay to the memory of Faraday, one of the greatest scientific discoverers of all time." C. N. R. Rao, Rao, C.N.R. (2000). ''Understanding Chemistry''. Universities Press. . p. 281.
 In 1812, at the age of 20 and at the end of his apprenticeship, Faraday attended lectures by the eminent English chemist Humphry Davy of the Royal Institution and the Royal Society, and John Tatum, founder of the City Philosophical Society. Many of the tickets for these lectures were given to Faraday by William Dance, who was one of the founders of the
In 1812, at the age of 20 and at the end of his apprenticeship, Faraday attended lectures by the eminent English chemist Humphry Davy of the Royal Institution and the Royal Society, and John Tatum, founder of the City Philosophical Society. Many of the tickets for these lectures were given to Faraday by William Dance, who was one of the founders of the
 In June 1832, the
In June 1832, the  Faraday suffered a nervous breakdown in 1839 but eventually returned to his investigations into electromagnetism. In 1848, as a result of representations by the Prince Consort, Faraday was awarded a
Faraday suffered a nervous breakdown in 1839 but eventually returned to his investigations into electromagnetism. In 1848, as a result of representations by the Prince Consort, Faraday was awarded a
 Faraday's earliest chemical work was as an assistant to Humphry Davy. Faraday was involved in the study of
Faraday's earliest chemical work was as an assistant to Humphry Davy. Faraday was involved in the study of
 In 1821, soon after the Danish physicist and chemist Hans Christian Ørsted discovered the phenomenon of electromagnetism, Davy and William Hyde Wollaston tried, but failed, to design an
In 1821, soon after the Danish physicist and chemist Hans Christian Ørsted discovered the phenomenon of electromagnetism, Davy and William Hyde Wollaston tried, but failed, to design an 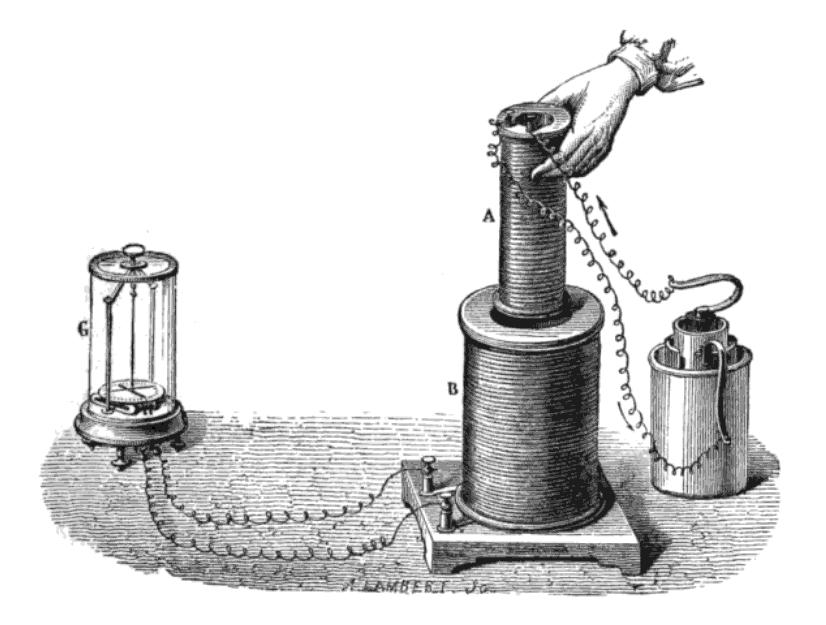 From his initial discovery in 1821, Faraday continued his laboratory work, exploring electromagnetic properties of materials and developing requisite experience. In 1824, Faraday briefly set up a circuit to study whether a magnetic field could regulate the flow of a current in an adjacent wire, but he found no such relationship. This experiment followed similar work conducted with light and magnets three years earlier that yielded identical results. During the next seven years, Faraday spent much of his time perfecting his recipe for optical quality (heavy) glass, borosilicate of lead, which he used in his future studies connecting light with magnetism. In his spare time, Faraday continued publishing his experimental work on optics and electromagnetism; he conducted correspondence with scientists whom he had met on his journeys across Europe with Davy, and who were also working on electromagnetism. Two years after the death of Davy, in 1831, he began his great series of experiments in which he discovered electromagnetic induction, recording in his laboratory diary on 28 October 1831 he was; "making many experiments with the great magnet of the Royal Society".
From his initial discovery in 1821, Faraday continued his laboratory work, exploring electromagnetic properties of materials and developing requisite experience. In 1824, Faraday briefly set up a circuit to study whether a magnetic field could regulate the flow of a current in an adjacent wire, but he found no such relationship. This experiment followed similar work conducted with light and magnets three years earlier that yielded identical results. During the next seven years, Faraday spent much of his time perfecting his recipe for optical quality (heavy) glass, borosilicate of lead, which he used in his future studies connecting light with magnetism. In his spare time, Faraday continued publishing his experimental work on optics and electromagnetism; he conducted correspondence with scientists whom he had met on his journeys across Europe with Davy, and who were also working on electromagnetism. Two years after the death of Davy, in 1831, he began his great series of experiments in which he discovered electromagnetic induction, recording in his laboratory diary on 28 October 1831 he was; "making many experiments with the great magnet of the Royal Society".

 Faraday's breakthrough came when he wrapped two insulated coils of wire around an iron ring, and found that, upon passing a current through one coil, a momentary current was induced in the other coil. This phenomenon is now known as mutual induction. The iron ring-coil apparatus is still on display at the Royal Institution. In subsequent experiments, he found that if he moved a magnet through a loop of wire an electric current flowed in that wire. The current also flowed if the loop was moved over a stationary magnet. His demonstrations established that a changing magnetic field produces an electric field; this relation was modelled mathematically by
Faraday's breakthrough came when he wrapped two insulated coils of wire around an iron ring, and found that, upon passing a current through one coil, a momentary current was induced in the other coil. This phenomenon is now known as mutual induction. The iron ring-coil apparatus is still on display at the Royal Institution. In subsequent experiments, he found that if he moved a magnet through a loop of wire an electric current flowed in that wire. The current also flowed if the loop was moved over a stationary magnet. His demonstrations established that a changing magnetic field produces an electric field; this relation was modelled mathematically by
 In 1845, Faraday discovered that many materials exhibit a weak repulsion from a magnetic field: a phenomenon he termed
In 1845, Faraday discovered that many materials exhibit a weak repulsion from a magnetic field: a phenomenon he termed
 Faraday had a long association with the
Faraday had a long association with the  As a respected scientist in a nation with strong maritime interests, Faraday spent extensive amounts of time on projects such as the construction and operation of lighthouses and protecting the bottoms of ships from
As a respected scientist in a nation with strong maritime interests, Faraday spent extensive amounts of time on projects such as the construction and operation of lighthouses and protecting the bottoms of ships from  Faraday assisted with the planning and judging of exhibits for the Great Exhibition of 1851 in London. He also advised the
Faraday assisted with the planning and judging of exhibits for the Great Exhibition of 1851 in London. He also advised the  Before his famous Christmas lectures, Faraday delivered chemistry lectures for the City Philosophical Society from 1816 to 1818 in order to refine the quality of his lectures.
Between 1827 and 1860 at the Royal Institution in London, Faraday gave a series of nineteen Christmas lectures for young people, a series which continues today. The objective of the lectures was to present science to the general public in the hopes of inspiring them and generating revenue for the Royal Institution. They were notable events on the social calendar among London's gentry. Over the course of several letters to his close friend Benjamin Abbott, Faraday outlined his recommendations on the art of lecturing, writing "a flame should be lighted at the commencement and kept alive with unremitting splendour to the end". His lectures were joyful and juvenile, he delighted in filling soap bubbles with various gasses (in order to determine whether or not they are magnetic), but the lectures were also deeply philosophical. In his lectures he urged his audiences to consider the mechanics of his experiments: "you know very well that ice floats upon water ... Why does the ice float? Think of that, and philosophise". The subjects in his lectures consisted of Chemistry and Electricity, and included: 1841: ''The Rudiments of Chemistry'', 1843: ''First Principles of Electricity'', 1848: ''
Before his famous Christmas lectures, Faraday delivered chemistry lectures for the City Philosophical Society from 1816 to 1818 in order to refine the quality of his lectures.
Between 1827 and 1860 at the Royal Institution in London, Faraday gave a series of nineteen Christmas lectures for young people, a series which continues today. The objective of the lectures was to present science to the general public in the hopes of inspiring them and generating revenue for the Royal Institution. They were notable events on the social calendar among London's gentry. Over the course of several letters to his close friend Benjamin Abbott, Faraday outlined his recommendations on the art of lecturing, writing "a flame should be lighted at the commencement and kept alive with unremitting splendour to the end". His lectures were joyful and juvenile, he delighted in filling soap bubbles with various gasses (in order to determine whether or not they are magnetic), but the lectures were also deeply philosophical. In his lectures he urged his audiences to consider the mechanics of his experiments: "you know very well that ice floats upon water ... Why does the ice float? Think of that, and philosophise". The subjects in his lectures consisted of Chemistry and Electricity, and included: 1841: ''The Rudiments of Chemistry'', 1843: ''First Principles of Electricity'', 1848: ''
 A statue of Faraday stands in Savoy Place, London, outside the
A statue of Faraday stands in Savoy Place, London, outside the  A Royal Society of Arts blue plaque, unveiled in 1876, commemorates Faraday at 48 Blandford Street in London's Marylebone district. From 1991 until 2001, Faraday's picture featured on the reverse of Series E £20
A Royal Society of Arts blue plaque, unveiled in 1876, commemorates Faraday at 48 Blandford Street in London's Marylebone district. From 1991 until 2001, Faraday's picture featured on the reverse of Series E £20
's list of the 100 Greatest Britons following a UK-wide vote.
Faraday has been commemorated on postage stamps issued by the Royal Mail. In 1991, as a pioneer of electricity he featured in their Scientific Achievements issue along with pioneers in three other fields ( Charles Babbage (computing), diamagnetism
Diamagnetic materials are repelled by a magnetic field; an applied magnetic field creates an induced magnetic field in them in the opposite direction, causing a repulsive force. In contrast, paramagnetic and ferromagnetic materials are attracte ...
and electrolysis.
Although Faraday received little formal education, he was one of the most influential scientists in history. It was by his research on the magnetic field around a conductor carrying a direct current
Direct current (DC) is one-directional flow of electric charge. An electrochemical cell is a prime example of DC power. Direct current may flow through a conductor such as a wire, but can also flow through semiconductors, insulators, or eve ...
that Faraday established the concept of the electromagnetic field in physics. Faraday also established that magnetism could affect rays of light and that there was an underlying relationship between the two phenomena.. the 1911 Encyclopædia Britannica. He similarly discovered the principles of electromagnetic induction, diamagnetism, and the laws of electrolysis
Faraday's laws of electrolysis are quantitative relationships based on the electrochemical research published by Michael Faraday in 1833.
First law
Michael Faraday reported that the mass (m) of elements deposited at an electrode is directly prop ...
. His inventions of electromagnetic rotary devices formed the foundation of electric motor technology, and it was largely due to his efforts that electricity
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as describ ...
became practical for use in technology.
As a chemist, Faraday discovered benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms ...
, investigated the clathrate hydrate of chlorine, invented an early form of the Bunsen burner
A Bunsen burner, named after Robert Bunsen, is a kind of ambient air gas burner used as laboratory equipment; it produces a single open gas flame, and is used for heating, sterilization, and combustion.
The gas can be natural gas (which is ma ...
and the system of oxidation numbers, and popularised terminology such as "anode
An anode is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, an electrode of the device through which conventional current leaves the device. A common mnemonic ...
", " cathode", " electrode" and " ion". Faraday ultimately became the first and foremost Fullerian Professor of Chemistry
The Fullerian Chairs at the Royal Institution in London, England, were established by John 'Mad Jack' Fuller.
Fullerian Professors of Chemistry
* 1833 Michael Faraday
* 1868 William Odling
* 1874 John Hall Gladstone
* 1877 James Dewar
* 1923 Wi ...
at the Royal Institution, a lifetime position.
Faraday was an excellent experimentalist who conveyed his ideas in clear and simple language; his mathematical abilities, however, did not extend as far as trigonometry and were limited to the simplest algebra. James Clerk Maxwell
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish mathematician and scientist responsible for the classical theory of electromagnetic radiation, which was the first theory to describe electricity, magnetism and li ...
took the work of Faraday and others and summarized it in a set of equations which is accepted as the basis of all modern theories of electromagnetic phenomena. On Faraday's uses of lines of force
A line of force in Faraday's extended sense is synonymous with Maxwell's line of induction. According to J.J. Thomson, Faraday usually discusses ''lines of force'' as chains of polarized particles in a dielectric, yet sometimes Faraday discusses ...
, Maxwell wrote that they show Faraday "to have been in reality a mathematician of a very high order – one from whom the mathematicians of the future may derive valuable and fertile methods."The_Scientific_Papers_of_ The_Scientific_Papers_of_James_Clerk_Maxwell_Volume_1">James_Clerk_Maxwell">The_Scientific_Papers_of_James_Clerk_Maxwell
_Volume_1p._360;_Courier_Dover_2003,__The_International_System_of_Units.html" ;"title="James_Clerk_Maxwell
_Volume_1.html" ;"title="James Clerk Maxwell">The Scientific Papers of James Clerk Maxwell
Volume 1">James Clerk Maxwell">The Scientific Papers of James Clerk Maxwell
Volume 1p. 360; Courier Dover 2003, The International System of Units">SI unit of capacitance is named in his honour: the farad. Albert Einstein kept a picture of Faraday on his study wall, alongside pictures of Arthur Schopenhauer and James Clerk Maxwell. Physicist Ernest Rutherford stated, "When we consider the magnitude and extent of his discoveries and their influence on the progress of science and of industry, there is no honour too great to pay to the memory of Faraday, one of the greatest scientific discoverers of all time." C. N. R. Rao, Rao, C.N.R. (2000). ''Understanding Chemistry''. Universities Press. . p. 281.
Personal life
Early life
Michael Faraday was born on 22 September 1791 in Newington Butts, Surrey (which is now part of theLondon Borough of Southwark
The London Borough of Southwark ( ) in South London forms part of Inner London and is connected by bridges across the River Thames to the City of London and London Borough of Tower Hamlets. It was created in 1965 when three smaller council areas ...
). His family was not well off. His father, James, was a member of the Glasite
The Glasites or Glassites were a small Christian church founded in about 1730 in Scotland by John Glas.John Glas preached supremacy of God's word (Bible) over allegiance to Church and state to his congregation in Tealing near Dundee in July 172 ...
sect of Christianity. James Faraday moved his wife, Margaret (née Hastwell), and two children to London during the winter of 1790 from Outhgill
Outhgill is a hamlet in Mallerstang, Cumbria, England. It lies about south of Kirkby Stephen.
It is the main hamlet in the dale of Mallerstang (a civil parish) which retains the Norse pattern of its original settlement: a series of small ...
in Westmorland, where he had been an apprentice to the village blacksmith. Michael was born in the autumn of that year. The young Michael Faraday, who was the third of four children, having only the most basic school education, had to educate himself.
At the age of 14 he became an apprentice to George Riebau, a local bookbinder and bookseller in Blandford Street. During his seven-year apprenticeship Faraday read many books, including Isaac Watts
Isaac Watts (17 July 1674 – 25 November 1748) was an English Congregational minister, hymn writer, theologian, and logician. He was a prolific and popular hymn writer and is credited with some 750 hymns. His works include "When I Survey the ...
's ''The Improvement of the Mind'', and he enthusiastically implemented the principles and suggestions contained therein. He also developed an interest in science, especially in electricity. Faraday was particularly inspired by the book ''Conversations on Chemistry'' by Jane Marcet
Jane Marcet (née Haldimand) (1 January 1769 – 28 June 1858) was an English salonnière of Swiss origin, and an innovative writer of popular, explanatory science books. She also broke ground with ''Conversations on Political Economy'' (1816 ...
.
Adult life
 In 1812, at the age of 20 and at the end of his apprenticeship, Faraday attended lectures by the eminent English chemist Humphry Davy of the Royal Institution and the Royal Society, and John Tatum, founder of the City Philosophical Society. Many of the tickets for these lectures were given to Faraday by William Dance, who was one of the founders of the
In 1812, at the age of 20 and at the end of his apprenticeship, Faraday attended lectures by the eminent English chemist Humphry Davy of the Royal Institution and the Royal Society, and John Tatum, founder of the City Philosophical Society. Many of the tickets for these lectures were given to Faraday by William Dance, who was one of the founders of the Royal Philharmonic Society
The Royal Philharmonic Society (RPS) is a British music society, formed in 1813. Its original purpose was to promote performances of instrumental music in London. Many composers and performers have taken part in its concerts. It is now a memb ...
. Faraday subsequently sent Davy a 300-page book based on notes that he had taken during these lectures. Davy's reply was immediate, kind, and favourable. In 1813, when Davy damaged his eyesight in an accident with nitrogen trichloride, he decided to employ Faraday as an assistant. Coincidentally one of the Royal Institution's assistants, John Payne, was sacked and Sir Humphry Davy had been asked to find a replacement; thus he appointed Faraday as Chemical Assistant at the Royal Institution on 1 March 1813. Very soon Davy entrusted Faraday with the preparation of nitrogen trichloride samples, and they both were injured in an explosion of this very sensitive substance.
Faraday married Sarah Barnard (1800–1879) on 12 June 1821. They met through their families at the Sandemanian
The Glasites or Glassites were a small Christian church founded in about 1730 in Scotland by John Glas.John Glas preached supremacy of God's word (Bible) over allegiance to Church and state to his congregation in Tealing near Dundee in July 172 ...
church, and he confessed his faith to the Sandemanian congregation the month after they were married. They had no children.
Faraday was a devout Christian; his Sandemanian denomination was an offshoot of the Church of Scotland
The Church of Scotland ( sco, The Kirk o Scotland; gd, Eaglais na h-Alba) is the national church in Scotland.
The Church of Scotland was principally shaped by John Knox, in the Reformation of 1560, when it split from the Catholic Church ...
. Well after his marriage, he served as deacon
A deacon is a member of the diaconate, an office in Christian churches that is generally associated with service of some kind, but which varies among theological and denominational traditions. Major Christian churches, such as the Catholic Chur ...
and for two terms as an elder in the meeting house of his youth. His church was located at Paul's Alley in the Barbican. This meeting house relocated in 1862 to Barnsbury
Barnsbury is an area of north London in the London Borough of Islington, within the N1 and N7 postal districts.
The name is a syncopated form of ''Bernersbury'' (1274), being so called after the Berners family: powerful medieval manorial ...
Grove, Islington; this North London location was where Faraday served the final two years of his second term as elder prior to his resignation from that post. Biographers have noted that "a strong sense of the unity of God and nature pervaded Faraday's life and work."
Later life
 In June 1832, the
In June 1832, the University of Oxford
, mottoeng = The Lord is my light
, established =
, endowment = £6.1 billion (including colleges) (2019)
, budget = £2.145 billion (2019–20)
, chancellor ...
granted Faraday an honorary Doctor of Civil Law
Doctor of Civil Law (DCL; la, Legis Civilis Doctor or Juris Civilis Doctor) is a degree offered by some universities, such as the University of Oxford, instead of the more common Doctor of Laws (LLD) degrees.
At Oxford, the degree is a higher ...
degree. During his lifetime, he was offered a knighthood
A knight is a person granted an honorary title of knighthood by a head of state (including the Pope) or representative for service to the monarch, the church or the country, especially in a military capacity. Knighthood finds origins in the ...
in recognition for his services to science, which he turned down on religious grounds, believing that it was against the word of the Bible to accumulate riches and pursue worldly reward, and stating that he preferred to remain "plain Mr Faraday to the end". Elected a Fellow of the Royal Society in 1824, he twice refused to become President. He became the first Fullerian Professor of Chemistry
The Fullerian Chairs at the Royal Institution in London, England, were established by John 'Mad Jack' Fuller.
Fullerian Professors of Chemistry
* 1833 Michael Faraday
* 1868 William Odling
* 1874 John Hall Gladstone
* 1877 James Dewar
* 1923 Wi ...
at the Royal Institution in 1833.
In 1832, Faraday was elected a Foreign Honorary Member of the American Academy of Arts and Sciences
The American Academy of Arts and Sciences (abbreviation: AAA&S) is one of the oldest learned societies in the United States. It was founded in 1780 during the American Revolution by John Adams, John Hancock, James Bowdoin, Andrew Oliver, a ...
. He was elected a foreign member of the Royal Swedish Academy of Sciences in 1838. In 1840, he was elected to the American Philosophical Society
The American Philosophical Society (APS), founded in 1743 in Philadelphia, is a scholarly organization that promotes knowledge in the sciences and humanities through research, professional meetings, publications, library resources, and communit ...
. He was one of eight foreign members elected to the French Academy of Sciences in 1844. In 1849 he was elected as associated member to the Royal Institute of the Netherlands, which two years later became the Royal Netherlands Academy of Arts and Sciences and he was subsequently made foreign member.
 Faraday suffered a nervous breakdown in 1839 but eventually returned to his investigations into electromagnetism. In 1848, as a result of representations by the Prince Consort, Faraday was awarded a
Faraday suffered a nervous breakdown in 1839 but eventually returned to his investigations into electromagnetism. In 1848, as a result of representations by the Prince Consort, Faraday was awarded a grace and favour
''Grace & Favour'' (American title: ''Are You Being Served? Again!'') is a British sitcom and a spin-off of '' Are You Being Served?'' that aired on BBC1 for two series from 1992 to 1993. It was written by ''Are You Being Served?'' creators and ...
house in Hampton Court
Hampton Court Palace is a Grade I listed royal palace in the London Borough of Richmond upon Thames, southwest and upstream of central London on the River Thames. The building of the palace began in 1514 for Cardinal Thomas Wolsey, the chie ...
in Middlesex, free of all expenses and upkeep. This was the Master Mason's House, later called Faraday House, and now No. 37 Hampton Court Road. In 1858 Faraday retired to live there.
Having provided a number of various service projects for the British government, when asked by the government to advise on the production of chemical weapons for use in the Crimean War
The Crimean War, , was fought from October 1853 to February 1856 between Russia and an ultimately victorious alliance of the Ottoman Empire, France, the United Kingdom and Piedmont-Sardinia.
Geopolitical causes of the war included the ...
(1853–1856), Faraday refused to participate, citing ethical reasons.
Faraday died at his house at Hampton Court
Hampton Court Palace is a Grade I listed royal palace in the London Borough of Richmond upon Thames, southwest and upstream of central London on the River Thames. The building of the palace began in 1514 for Cardinal Thomas Wolsey, the chie ...
on 25 August 1867, aged 75. He had some years before turned down an offer of burial in Westminster Abbey
Westminster Abbey, formally titled the Collegiate Church of Saint Peter at Westminster, is an historic, mainly Gothic church in the City of Westminster, London, England, just to the west of the Palace of Westminster. It is one of the Unite ...
upon his death, but he has a memorial plaque there, near Isaac Newton's tomb. Faraday was interred in the dissenters' (non- Anglican) section of Highgate Cemetery.
Scientific achievements
Chemistry
 Faraday's earliest chemical work was as an assistant to Humphry Davy. Faraday was involved in the study of
Faraday's earliest chemical work was as an assistant to Humphry Davy. Faraday was involved in the study of chlorine
Chlorine is a chemical element with the symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine i ...
; he discovered two new compounds of chlorine and carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon mak ...
. He also conducted the first rough experiments on the diffusion of gases, a phenomenon that was first pointed out by John Dalton. The physical importance of this phenomenon was more fully revealed by Thomas Graham and Joseph Loschmidt
Johann Josef Loschmidt (15 March 1821 – 8 July 1895), who referred to himself mostly as Josef Loschmidt (omitting his first name), was a notable Austrian scientist who performed ground-breaking work in chemistry, physics (thermodynamics, optics, ...
. Faraday succeeded in liquefying several gases, investigated the alloys of steel, and produced several new kinds of glass intended for optical purposes. A specimen of one of these heavy glasses subsequently became historically important; when the glass was placed in a magnetic field Faraday determined the rotation of the plane of polarisation of light. This specimen was also the first substance found to be repelled by the poles of a magnet.
Faraday invented an early form of what was to become the Bunsen burner
A Bunsen burner, named after Robert Bunsen, is a kind of ambient air gas burner used as laboratory equipment; it produces a single open gas flame, and is used for heating, sterilization, and combustion.
The gas can be natural gas (which is ma ...
, which is still in practical use in science laboratories around the world as a convenient source of heat.
Faraday worked extensively in the field of chemistry, discovering chemical substances such as benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms ...
(which he called bicarburet of hydrogen) and liquefying gases such as chlorine. The liquefying of gases helped to establish that gases are the vapours of liquids possessing a very low boiling point and gave a more solid basis to the concept of molecular aggregation. In 1820 Faraday reported the first synthesis of compounds made from carbon and chlorine, C2Cl6 and C2Cl4, and published his results the following year. Faraday also determined the composition of the chlorine clathrate hydrate, which had been discovered by Humphry Davy in 1810. Faraday is also responsible for discovering the laws of electrolysis
Faraday's laws of electrolysis are quantitative relationships based on the electrochemical research published by Michael Faraday in 1833.
First law
Michael Faraday reported that the mass (m) of elements deposited at an electrode is directly prop ...
, and for popularizing terminology such as anode
An anode is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, an electrode of the device through which conventional current leaves the device. A common mnemonic ...
, cathode, electrode, and ion, terms proposed in large part by William Whewell.
Faraday was the first to report what later came to be called metallic nanoparticles. In 1847 he discovered that the optical properties of gold colloids differed from those of the corresponding bulk metal. This was probably the first reported observation of the effects of quantum size, and might be considered to be the birth of nanoscience
The nanoscopic scale (or nanoscale) usually refers to structures with a length scale applicable to nanotechnology, usually cited as 1–100 nanometers (nm). A nanometer is a billionth of a meter. The nanoscopic scale is (roughly speaking) a lo ...
.
Electricity and magnetism
Faraday is best known for his work on electricity and magnetism. His first recorded experiment was the construction of avoltaic pile
upright=1.2, Schematic diagram of a copper–zinc voltaic pile. The copper and zinc discs were separated by cardboard or felt spacers soaked in salt water (the electrolyte). Volta's original piles contained an additional zinc disk at the bottom, ...
with seven British halfpenny coins, stacked together with seven discs of sheet zinc, and six pieces of paper moistened with salt water. With this pile he decomposed sulfate of magnesia (first letter to Abbott, 12 July 1812).
 In 1821, soon after the Danish physicist and chemist Hans Christian Ørsted discovered the phenomenon of electromagnetism, Davy and William Hyde Wollaston tried, but failed, to design an
In 1821, soon after the Danish physicist and chemist Hans Christian Ørsted discovered the phenomenon of electromagnetism, Davy and William Hyde Wollaston tried, but failed, to design an electric motor
An electric motor is an electrical machine that converts electrical energy into mechanical energy. Most electric motors operate through the interaction between the motor's magnetic field and electric current in a wire winding to generate for ...
. Faraday, having discussed the problem with the two men, went on to build two devices to produce what he called "electromagnetic rotations". One of these, now known as the homopolar motor, caused a continuous circular motion that was engendered by the circular magnetic force around a wire that extended into a pool of mercury wherein was placed a magnet; the wire would then rotate around the magnet if supplied with current from a chemical battery. These experiments and inventions, first carried out in the basement of the Royal Institution on 3 September 1821, formed the foundation of modern electromagnetic technology. Faraday published the results of his discovery in the '' Quarterly Journal of Science'', and sent copies of his paper along with pocket-sized models of his device to colleagues around the world so they could also witness the phenomenon of electromagnetic rotations. In his excitement, Faraday hastily released the paper which had not acknowledged his work with either Wollaston or Davy. The resulting controversy within the Royal Society strained his mentor relationship with Davy and may well have contributed to Faraday's assignment to other activities, which consequently prevented his involvement in electromagnetic research for several years.
 From his initial discovery in 1821, Faraday continued his laboratory work, exploring electromagnetic properties of materials and developing requisite experience. In 1824, Faraday briefly set up a circuit to study whether a magnetic field could regulate the flow of a current in an adjacent wire, but he found no such relationship. This experiment followed similar work conducted with light and magnets three years earlier that yielded identical results. During the next seven years, Faraday spent much of his time perfecting his recipe for optical quality (heavy) glass, borosilicate of lead, which he used in his future studies connecting light with magnetism. In his spare time, Faraday continued publishing his experimental work on optics and electromagnetism; he conducted correspondence with scientists whom he had met on his journeys across Europe with Davy, and who were also working on electromagnetism. Two years after the death of Davy, in 1831, he began his great series of experiments in which he discovered electromagnetic induction, recording in his laboratory diary on 28 October 1831 he was; "making many experiments with the great magnet of the Royal Society".
From his initial discovery in 1821, Faraday continued his laboratory work, exploring electromagnetic properties of materials and developing requisite experience. In 1824, Faraday briefly set up a circuit to study whether a magnetic field could regulate the flow of a current in an adjacent wire, but he found no such relationship. This experiment followed similar work conducted with light and magnets three years earlier that yielded identical results. During the next seven years, Faraday spent much of his time perfecting his recipe for optical quality (heavy) glass, borosilicate of lead, which he used in his future studies connecting light with magnetism. In his spare time, Faraday continued publishing his experimental work on optics and electromagnetism; he conducted correspondence with scientists whom he had met on his journeys across Europe with Davy, and who were also working on electromagnetism. Two years after the death of Davy, in 1831, he began his great series of experiments in which he discovered electromagnetic induction, recording in his laboratory diary on 28 October 1831 he was; "making many experiments with the great magnet of the Royal Society".
 Faraday's breakthrough came when he wrapped two insulated coils of wire around an iron ring, and found that, upon passing a current through one coil, a momentary current was induced in the other coil. This phenomenon is now known as mutual induction. The iron ring-coil apparatus is still on display at the Royal Institution. In subsequent experiments, he found that if he moved a magnet through a loop of wire an electric current flowed in that wire. The current also flowed if the loop was moved over a stationary magnet. His demonstrations established that a changing magnetic field produces an electric field; this relation was modelled mathematically by
Faraday's breakthrough came when he wrapped two insulated coils of wire around an iron ring, and found that, upon passing a current through one coil, a momentary current was induced in the other coil. This phenomenon is now known as mutual induction. The iron ring-coil apparatus is still on display at the Royal Institution. In subsequent experiments, he found that if he moved a magnet through a loop of wire an electric current flowed in that wire. The current also flowed if the loop was moved over a stationary magnet. His demonstrations established that a changing magnetic field produces an electric field; this relation was modelled mathematically by James Clerk Maxwell
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish mathematician and scientist responsible for the classical theory of electromagnetic radiation, which was the first theory to describe electricity, magnetism and li ...
as Faraday's law, which subsequently became one of the four Maxwell equations, and which have in turn evolved into the generalization known today as field theory. Faraday would later use the principles he had discovered to construct the electric dynamo
"Dynamo Electric Machine" (end view, partly section, )
A dynamo is an electrical generator that creates direct current using a commutator. Dynamos were the first electrical generators capable of delivering power for industry, and the foundati ...
, the ancestor of modern power generators and the electric motor.
In 1832, he completed a series of experiments aimed at investigating the fundamental nature of electricity; Faraday used "static
Static may refer to:
Places
*Static Nunatak, a nunatak in Antarctica
United States
* Static, Kentucky and Tennessee
*Static Peak, a mountain in Wyoming
**Static Peak Divide, a mountain pass near the peak
Science and technology Physics
*Static el ...
", batteries, and " animal electricity" to produce the phenomena of electrostatic attraction, electrolysis, magnetism, etc. He concluded that, contrary to the scientific opinion of the time, the divisions between the various "kinds" of electricity were illusory. Faraday instead proposed that only a single "electricity" exists, and the changing values of quantity and intensity (current and voltage) would produce different groups of phenomena.
Near the end of his career, Faraday proposed that electromagnetic forces extended into the empty space around the conductor. This idea was rejected by his fellow scientists, and Faraday did not live to see the eventual acceptance of his proposition by the scientific community. Faraday's concept of lines of flux emanating from charged bodies and magnets provided a way to visualize electric and magnetic fields; that conceptual model was crucial for the successful development of the electromechanical devices that dominated engineering and industry for the remainder of the 19th century.
Diamagnetism
 In 1845, Faraday discovered that many materials exhibit a weak repulsion from a magnetic field: a phenomenon he termed
In 1845, Faraday discovered that many materials exhibit a weak repulsion from a magnetic field: a phenomenon he termed diamagnetism
Diamagnetic materials are repelled by a magnetic field; an applied magnetic field creates an induced magnetic field in them in the opposite direction, causing a repulsive force. In contrast, paramagnetic and ferromagnetic materials are attracte ...
.
Faraday also discovered that the plane of polarization of linearly polarized light can be rotated by the application of an external magnetic field aligned with the direction in which the light is moving. This is now termed the Faraday effect
The Faraday effect or Faraday rotation, sometimes referred to as the magneto-optic Faraday effect (MOFE), is a physical magneto-optical phenomenon. The Faraday effect causes a polarization rotation which is proportional to the projection of the ...
. In Sept 1845 he wrote in his notebook, "I have at last succeeded in ''illuminating a magnetic curve'' or '' line of force'' and in ''magnetising a ray of light
''Ray of Light'' is the seventh studio album by American singer-songwriter Madonna, released in early 1998 by Maverick Records. A stylistic and aesthetical departure from her previous work, ''Ray of Light'' is an electronica and techno-pop reco ...
''".
Later on in his life, in 1862, Faraday used a spectroscope to search for a different alteration of light, the change of spectral lines by an applied magnetic field. The equipment available to him was, however, insufficient for a definite determination of spectral change. Pieter Zeeman later used an improved apparatus to study the same phenomenon, publishing his results in 1897 and receiving the 1902 Nobel Prize in Physics for his success. In both his 1897 paper and his Nobel acceptance speech, Zeeman made reference to Faraday's work.
Faraday cage
In his work on static electricity,Faraday's ice pail experiment
Faraday's ice pail experiment is a simple electrostatics experiment performed in 1843 by British scientist Michael Faraday that demonstrates the effect of electrostatic induction on a conducting container. For a container, Faraday used a metal p ...
demonstrated that the charge resided only on the exterior of a charged conductor, and exterior charge had no influence on anything enclosed within a conductor. This is because the exterior charges redistribute such that the interior fields emanating from them cancel one another. This shielding effect is used in what is now known as a Faraday cage. In January 1836, Faraday had put a wooden frame, 12ft square, on four glass supports and added paper walls and wire mesh. He then stepped inside and electrified it. When he stepped out of his electrified cage, Faraday had shown that electricity was a force, not an imponderable fluid as was believed at the time.
Royal Institution and public service
 Faraday had a long association with the
Faraday had a long association with the Royal Institution of Great Britain
The Royal Institution of Great Britain (often the Royal Institution, Ri or RI) is an organisation for scientific education and research, based in the City of Westminster. It was founded in 1799 by the leading British scientists of the age, inc ...
. He was appointed Assistant Superintendent of the House of the Royal Institution in 1821. He was elected a Fellow of the Royal Society in 1824. In 1825, he became Director of the Laboratory of the Royal Institution. Six years later, in 1833, Faraday became the first Fullerian Professor of Chemistry
The Fullerian Chairs at the Royal Institution in London, England, were established by John 'Mad Jack' Fuller.
Fullerian Professors of Chemistry
* 1833 Michael Faraday
* 1868 William Odling
* 1874 John Hall Gladstone
* 1877 James Dewar
* 1923 Wi ...
at the Royal Institution of Great Britain
The Royal Institution of Great Britain (often the Royal Institution, Ri or RI) is an organisation for scientific education and research, based in the City of Westminster. It was founded in 1799 by the leading British scientists of the age, inc ...
, a position to which he was appointed for life without the obligation to deliver lectures. His sponsor and mentor was John 'Mad Jack' Fuller, who created the position at the Royal Institution for Faraday.
Beyond his scientific research into areas such as chemistry, electricity, and magnetism at the Royal Institution, Faraday undertook numerous, and often time-consuming, service projects for private enterprise and the British government. This work included investigations of explosions in coal mines, being an expert witness
An expert witness, particularly in common law countries such as the United Kingdom, Australia, and the United States, is a person whose opinion by virtue of education, training, certification, skills or experience, is accepted by the judge as ...
in court, and along with two engineers from Chance Brothers
Chance Brothers and Company was a glassworks originally based in Spon Lane, Smethwick, West Midlands (formerly in Staffordshire), in England. It was a leading glass manufacturer and a pioneer of British glassmaking technology.
The Chance fam ...
c.1853, the preparation of high-quality optical glass, which was required by Chance for its lighthouses. In 1846, together with Charles Lyell, he produced a lengthy and detailed report on a serious explosion in the colliery at Haswell, County Durham
Haswell is a village in County Durham, in England. It is situated east of the city of Durham, south of the city of Sunderland and north-west of the town of Peterlee.
Haswell was the birthplace of world champion road racing cyclist Tom Simpson, ...
, which killed 95 miners. Their report was a meticulous forensic investigation
Forensic science, also known as criminalistics, is the application of science to criminal and civil laws, mainly—on the criminal side—during criminal investigation, as governed by the legal standards of admissible evidence and criminal p ...
and indicated that coal dust Coal dust is a fine powdered form of which is created by the crushing, grinding, or pulverizing of coal. Because of the brittle nature of coal, coal dust can be created during mining, transportation, or by mechanically handling coal. It is a form ...
contributed to the severity of the explosion. The first-time explosions had been linked to dust, Faraday gave a demonstration during a lecture on how ventilation could prevent it. The report should have warned coal owners of the hazard of coal dust explosions, but the risk was ignored for over 60 years until the 1913 Senghenydd Colliery Disaster
The Senghenydd colliery disaster, also known as the Senghenydd explosion ( cy, Tanchwa Senghennydd), occurred at the Universal Colliery in Senghenydd, near Caerphilly, Glamorgan, Wales, on 14 October 1913. The explosion, which killed 439 mine ...
.
 As a respected scientist in a nation with strong maritime interests, Faraday spent extensive amounts of time on projects such as the construction and operation of lighthouses and protecting the bottoms of ships from
As a respected scientist in a nation with strong maritime interests, Faraday spent extensive amounts of time on projects such as the construction and operation of lighthouses and protecting the bottoms of ships from corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engi ...
. His workshop still stands at Trinity Buoy Wharf
Trinity Buoy Wharf is the site of a lighthouse, by the confluence of the River Thames and Bow Creek on the Leamouth Peninsula, Poplar. It lies within the London Borough of Tower Hamlets. The lighthouse no longer functions, but is the home of ...
above the Chain and Buoy Store, next to London's only lighthouse where he carried out the first experiments in electric lighting for lighthouses.
Faraday was also active in what would now be called environmental science, or engineering. He investigated industrial pollution at Swansea and was consulted on air pollution at the Royal Mint. In July 1855, Faraday wrote a letter to ''The Times
''The Times'' is a British daily national newspaper based in London. It began in 1785 under the title ''The Daily Universal Register'', adopting its current name on 1 January 1788. ''The Times'' and its sister paper '' The Sunday Times'' (f ...
'' on the subject of the foul condition of the River Thames
The River Thames ( ), known alternatively in parts as the River Isis, is a river that flows through southern England including London. At , it is the longest river entirely in England and the second-longest in the United Kingdom, after the R ...
, which resulted in an often-reprinted cartoon in '' Punch''. (See also The Great Stink).
 Faraday assisted with the planning and judging of exhibits for the Great Exhibition of 1851 in London. He also advised the
Faraday assisted with the planning and judging of exhibits for the Great Exhibition of 1851 in London. He also advised the National Gallery
The National Gallery is an art museum in Trafalgar Square in the City of Westminster, in Central London, England. Founded in 1824, it houses a collection of over 2,300 paintings dating from the mid-13th century to 1900. The current Director ...
on the cleaning and protection of its art collection, and served on the National Gallery Site Commission in 1857. Education was another of Faraday's areas of service; he lectured on the topic in 1854 at the Royal Institution, and, in 1862, he appeared before a Public Schools Commission to give his views on education in Great Britain. Faraday also weighed in negatively on the public's fascination with table-turning
Table-turning (also known as table-tapping, table-tipping or table-tilting) is a type of séance in which participants sit around a table, place their hands on it, and wait for rotations. The table was purportedly made to serve as a means of comm ...
, mesmerism, and seances, and in so doing chastised both the public and the nation's educational system.
 Before his famous Christmas lectures, Faraday delivered chemistry lectures for the City Philosophical Society from 1816 to 1818 in order to refine the quality of his lectures.
Between 1827 and 1860 at the Royal Institution in London, Faraday gave a series of nineteen Christmas lectures for young people, a series which continues today. The objective of the lectures was to present science to the general public in the hopes of inspiring them and generating revenue for the Royal Institution. They were notable events on the social calendar among London's gentry. Over the course of several letters to his close friend Benjamin Abbott, Faraday outlined his recommendations on the art of lecturing, writing "a flame should be lighted at the commencement and kept alive with unremitting splendour to the end". His lectures were joyful and juvenile, he delighted in filling soap bubbles with various gasses (in order to determine whether or not they are magnetic), but the lectures were also deeply philosophical. In his lectures he urged his audiences to consider the mechanics of his experiments: "you know very well that ice floats upon water ... Why does the ice float? Think of that, and philosophise". The subjects in his lectures consisted of Chemistry and Electricity, and included: 1841: ''The Rudiments of Chemistry'', 1843: ''First Principles of Electricity'', 1848: ''
Before his famous Christmas lectures, Faraday delivered chemistry lectures for the City Philosophical Society from 1816 to 1818 in order to refine the quality of his lectures.
Between 1827 and 1860 at the Royal Institution in London, Faraday gave a series of nineteen Christmas lectures for young people, a series which continues today. The objective of the lectures was to present science to the general public in the hopes of inspiring them and generating revenue for the Royal Institution. They were notable events on the social calendar among London's gentry. Over the course of several letters to his close friend Benjamin Abbott, Faraday outlined his recommendations on the art of lecturing, writing "a flame should be lighted at the commencement and kept alive with unremitting splendour to the end". His lectures were joyful and juvenile, he delighted in filling soap bubbles with various gasses (in order to determine whether or not they are magnetic), but the lectures were also deeply philosophical. In his lectures he urged his audiences to consider the mechanics of his experiments: "you know very well that ice floats upon water ... Why does the ice float? Think of that, and philosophise". The subjects in his lectures consisted of Chemistry and Electricity, and included: 1841: ''The Rudiments of Chemistry'', 1843: ''First Principles of Electricity'', 1848: ''The Chemical History of a Candle
''The Chemical History of a Candle'' was the title of a series of six lectures on the chemistry and physics of flames given by Michael Faraday at the Royal Institution in 1848, as part of the series of Christmas lectures for young people found ...
'', 1851: ''Attractive Forces'', 1853: ''Voltaic Electricity'', 1854: ''The Chemistry of Combustion'', 1855: ''The Distinctive Properties of the Common Metals'', 1857: ''Static Electricity'', 1858: ''The Metallic Properties'', 1859: ''The Various Forces of Matter and their Relations to Each Other''.
Commemorations
 A statue of Faraday stands in Savoy Place, London, outside the
A statue of Faraday stands in Savoy Place, London, outside the Institution of Engineering and Technology
The Institution of Engineering and Technology (IET) is a multidisciplinary professional engineering institution. The IET was formed in 2006 from two separate institutions: the Institution of Electrical Engineers (IEE), dating back to 1871, and ...
. The Michael Faraday Memorial, designed by brutalist architect Rodney Gordon
Rodney H Gordon (2 February 1933 – 30 May 2008) was an English architect. He was the primary architect of the Tricorn Centre, Portsmouth, and Trinity Square, Gateshead. Architecturally, his works were primarily in concrete; he was said to be ...
and completed in 1961, is at the Elephant & Castle
The Elephant and Castle is an area around a major road junction in London, England, in the London Borough of Southwark. The name also informally refers to much of Walworth and Newington, due to the proximity of the London Underground stati ...
gyratory system, near Faraday's birthplace at Newington Butts, London. Faraday School is located on Trinity Buoy Wharf
Trinity Buoy Wharf is the site of a lighthouse, by the confluence of the River Thames and Bow Creek on the Leamouth Peninsula, Poplar. It lies within the London Borough of Tower Hamlets. The lighthouse no longer functions, but is the home of ...
where his workshop still stands above the Chain and Buoy Store, next to London's only lighthouse. Faraday Gardens is a small park in Walworth, London, not far from his birthplace at Newington Butts. It lies within the local council ward of Faraday in the London Borough of Southwark
The London Borough of Southwark ( ) in South London forms part of Inner London and is connected by bridges across the River Thames to the City of London and London Borough of Tower Hamlets. It was created in 1965 when three smaller council areas ...
. Michael Faraday Primary school is situated on the Aylesbury Estate
The Aylesbury Estate is a large housing estate located in Walworth, South East London.
The Aylesbury Estate contains 2,704 dwellings, spread over a number of different blocks and buildings, and was built between 1963 and 1977. There are approx ...
in Walworth.
A building at London South Bank University
London South Bank University (LSBU) is a public university in Elephant and Castle, London. It is based in the London Borough of Southwark, near the South Bank of the River Thames, from which it takes its name. Founded in 1892 as the Borough ...
, which houses the institute's electrical engineering departments is named the Faraday Wing, due to its proximity to Faraday's birthplace in Newington Butts. A hall at Loughborough University was named after Faraday in 1960. Near the entrance to its dining hall is a bronze casting, which depicts the symbol of an electrical transformer
A transformer is a passive component that transfers electrical energy from one electrical circuit to another circuit, or multiple circuits. A varying current in any coil of the transformer produces a varying magnetic flux in the transformer' ...
, and inside there hangs a portrait, both in Faraday's honour. An eight-story building at the University of Edinburgh
The University of Edinburgh ( sco, University o Edinburgh, gd, Oilthigh Dhùn Èideann; abbreviated as ''Edin.'' in post-nominals) is a public research university based in Edinburgh, Scotland. Granted a royal charter by King James VI in 15 ...
's science & engineering campus is named for Faraday, as is a recently built hall of accommodation at Brunel University, the main engineering building at Swansea University
Swansea University ( cy, Prifysgol Abertawe) is a public university, public research university located in Swansea, Wales, United Kingdom. It was chartered as University College of Swansea in 1920, as the fourth college of the University of Wales. ...
, and the instructional and experimental physics building at Northern Illinois University
Northern Illinois University (NIU) is a public research university in DeKalb, Illinois. It was founded as Northern Illinois State Normal School on May 22, 1895, by Illinois Governor John P. Altgeld as part of an expansion of the state's system ...
. The former UK Faraday Station in Antarctica
Antarctica () is Earth's southernmost and least-populated continent. Situated almost entirely south of the Antarctic Circle and surrounded by the Southern Ocean, it contains the geographic South Pole. Antarctica is the fifth-largest cont ...
was named after him.
Streets named for Faraday can be found in many British cities (e.g., London, Fife, Swindon, Basingstoke, Nottingham
Nottingham ( , locally ) is a city and unitary authority area in Nottinghamshire, East Midlands, England. It is located north-west of London, south-east of Sheffield and north-east of Birmingham. Nottingham has links to the legend of Robi ...
, Whitby
Whitby is a seaside town, port and civil parish in the Scarborough borough of North Yorkshire, England. Situated on the east coast of Yorkshire at the mouth of the River Esk, Whitby has a maritime, mineral and tourist heritage. Its East Clif ...
, Kirkby, Crawley, Newbury, Swansea, Aylesbury and Stevenage) as well as in France (Paris), Germany (Berlin
Berlin ( , ) is the capital and List of cities in Germany by population, largest city of Germany by both area and population. Its 3.7 million inhabitants make it the European Union's List of cities in the European Union by population within ci ...
- Dahlem, Hermsdorf), Canada (Quebec City
Quebec City ( or ; french: Ville de Québec), officially Québec (), is the capital city of the Canadian province of Quebec. As of July 2021, the city had a population of 549,459, and the metropolitan area had a population of 839,311. It is t ...
, Quebec; Deep River, Ontario; Ottawa, Ontario), the United States ( Reston, Virginia), and New Zealand (Hawke's Bay
Hawke's Bay ( mi, Te Matau-a-Māui) is a local government region on the east coast of New Zealand's North Island. The region's name derives from Hawke Bay, which was named by Captain James Cook in honour of Admiral Edward Hawke. The region i ...
).
 A Royal Society of Arts blue plaque, unveiled in 1876, commemorates Faraday at 48 Blandford Street in London's Marylebone district. From 1991 until 2001, Faraday's picture featured on the reverse of Series E £20
A Royal Society of Arts blue plaque, unveiled in 1876, commemorates Faraday at 48 Blandford Street in London's Marylebone district. From 1991 until 2001, Faraday's picture featured on the reverse of Series E £20 banknotes
A banknote—also called a bill (North American English), paper money, or simply a note—is a type of negotiable promissory note, made by a bank or other licensed authority, payable to the bearer on demand.
Banknotes were originally issued ...
issued by the Bank of England. He was portrayed conducting a lecture at the Royal Institution with the magneto-electric spark apparatus. In 2002, Faraday was ranked number 22 in the BBC #REDIRECT BBC #REDIRECT BBC
Here i going to introduce about the best teacher of my life b BALAJI sir. He is the precious gift that I got befor 2yrs . How has helped and thought all the concept and made my success in the 10th board exam. ...
...Frank Whittle
Air Commodore Sir Frank Whittle, (1 June 1907 – 8 August 1996) was an English engineer, inventor and Royal Air Force (RAF) air officer. He is credited with inventing the turbojet engine. A patent was submitted by Maxime Guillaume in 1921 fo ...
(jet engine) and Robert Watson-Watt
Sir Robert Alexander Watson Watt (13 April 1892 – 5 December 1973) was a Scottish pioneer of radio direction finding and radar technology.
Watt began his career in radio physics with a job at the Met Office, where he began looking for accura ...
(radar)). In 1999, under the title "Faraday's Electricity", he featured in their World Changers issue along with Charles Darwin
Charles Robert Darwin ( ; 12 February 1809 – 19 April 1882) was an English naturalist, geologist, and biologist, widely known for his contributions to evolutionary biology. His proposition that all species of life have descended ...
, Edward Jenner
Edward Jenner, (17 May 1749 – 26 January 1823) was a British physician and scientist who pioneered the concept of vaccines, and created the smallpox vaccine, the world's first vaccine. The terms ''vaccine'' and ''vaccination'' are derived f ...
and Alan Turing.
The Faraday Institute for Science and Religion
The Faraday Institute for Science and Religion is an interdisciplinary academic research institute based in Cambridge, England. It is named after the 19th-century English scientist Michael Faraday, the pioneer of electromagnetic induction.
It wa ...
derives its name from the scientist, who saw his faith as integral to his scientific research. The logo of the institute is also based on Faraday's discoveries. It was created in 2006 by a $2,000,000 grant
Grant or Grants may refer to:
Places
*Grant County (disambiguation)
Australia
* Grant, Queensland, a locality in the Barcaldine Region, Queensland, Australia
United Kingdom
* Castle Grant
United States
* Grant, Alabama
* Grant, Inyo County, ...
from the John Templeton Foundation
The John Templeton Foundation (Templeton Foundation) is a philanthropic organization that reflects the ideas of its founder, John Templeton, who became wealthy via a career as a contrarian investor, and wanted to support progress in religious a ...
to carry out academic research, to foster understanding of the interaction between science and religion, and to engage public understanding in both these subject areas.
The Faraday Institution, an independent energy storage research institute established in 2017, also derives its name from Michael Faraday. The organisation serves as the UK's primary research programme to advance battery science and technology, education, public engagement and market research.
Faraday's life and contributions to electromagnetics was the principal topic of the tenth episode, titled " The Electric Boy", of the 2014 American science documentary series, '' Cosmos: A Spacetime Odyssey'', which was broadcast on Fox and the National Geographic Channel.
Aldous Huxley
Aldous Leonard Huxley (26 July 1894 – 22 November 1963) was an English writer and philosopher. He wrote nearly 50 books, both novels and non-fiction works, as well as wide-ranging essays, narratives, and poems.
Born into the prominent Huxle ...
wrote about Faraday in an essay entitled, ''A Night in Pietramala'': “He is always the natural philosopher. To discover truth is his sole aim and interest…even if I could be Shakespeare, I think I should still choose to be Faraday.” Calling Faraday her "hero", in a speech to the Royal Society, Margaret Thatcher
Margaret Hilda Thatcher, Baroness Thatcher (; 13 October 19258 April 2013) was Prime Minister of the United Kingdom from 1979 to 1990 and Leader of the Conservative Party from 1975 to 1990. She was the first female British prime ...
declared: “The value of his work must be higher than the capitalisation of all the shares on the Stock Exchange!”. She borrowed his bust from the Royal Institution and had it placed in the hall of 10 Downing Street.
Awards named in Faraday's honour
In honor and remembrance of his great scientific contributions, several institutions have created prizes and awards in his name. This include: * TheIET __NOTOC__
IET can refer to:
Organizations
* Institute of Educational Technology, part of the Open University
* Institution of Engineering and Technology, a UK-based professional engineering institution
** Institute of Engineers and Technicians, wh ...
Faraday Medal
* The Royal Society of London Michael Faraday Prize
The Royal Society of London Michael Faraday Prize is awarded for "excellence in communicating science to UK audiences". Named after Michael Faraday, the medal itself is made of silver gilt, and is accompanied by a purse of £2500.
Background
Th ...
* The Institute of Physics Michael Faraday Medal and Prize
* The Royal Society of Chemistry Faraday Lectureship Prize
Gallery
Bibliography
 Faraday's books, with the exception of ''Chemical Manipulation'', were collections of scientific papers or transcriptions of lectures. Hamilton, p. 220 Since his death, Faraday's diary has been published, as have several large volumes of his letters and Faraday's journal from his travels with Davy in 1813–1815.
*
Faraday's books, with the exception of ''Chemical Manipulation'', were collections of scientific papers or transcriptions of lectures. Hamilton, p. 220 Since his death, Faraday's diary has been published, as have several large volumes of his letters and Faraday's journal from his travels with Davy in 1813–1815.
* 2nd ed. 1830
* ; vol. iii. Richard Taylor and William Francis, 1855 * * * * – published in eight volumes; see also th
2009 publication
of Faraday's diary * * – volume 2, 1993; volume 3, 1996; volume 4, 1999 *
Course of six lectures on the various forces of matter, and their relations to each other
London; Glasgow: R. Griffin, 1860. * The Liquefaction of Gases, Edinburgh: W.F. Clay, 1896.
The letters of Faraday and Schoenbein 1836–1862. With notes, comments and references to contemporary letters
London: Williams & Norgate 1899.
Digital edition
by the University and State Library Düsseldorf)
See also
*Faraday
Michael Faraday (; 22 September 1791 – 25 August 1867) was an English scientist who contributed to the study of electromagnetism and electrochemistry. His main discoveries include the principles underlying electromagnetic induction, ...
(Unit of electrical charge)
* Forensic engineering
Forensic engineering has been defined as ''"the investigation of failures - ranging from serviceability to catastrophic - which may lead to legal activity, including both civil and criminal".'' It includes the investigation of materials, produc ...
* Nikola Tesla
* Tetrachloroethylene
Tetrachloroethylene, also known under the systematic name tetrachloroethene, or perchloroethylene, and many other names (and abbreviations such as "perc" or "PERC", and "PCE"), is a chlorocarbon with the formula Cl2C=CCl2 . It is a colorless li ...
* Timeline of hydrogen technologies
* Timeline of low-temperature technology
* Zeeman effect
References
Sources
* * * *Further reading
Biographies
* * * * The British Electrical and Allied Manufacturers Association (1931). ''Faraday''. Edinburgh: R. & R. Clark, Ltd. * * * * * * * * * *External links
Biographies
Biography at The Royal Institution of Great Britain
Faraday as a Discoverer by John Tyndall, Project Gutenberg
(downloads)
The Life and Discoveries of Michael Faraday
by J. A. Crowther, London: Society for Promoting Christian Knowledge, 1920
Others
* * *Complete Correspondence of Michael Faraday
Searchable full texts of all letters to and from Faraday, based on the standard edition by Frank James
Video Podcast
with Sir John Cadogan talking about Benzene since Faraday
The letters of Faraday and Schoenbein 1836–1862. With notes, comments and references to contemporary letters (1899)
full downloa
Faraday School, located on Trinity Buoy Wharf
at the New Model School Company Limited's website * , Chemical Heritage Foundation {{DEFAULTSORT:Faraday, Michael 1791 births 1867 deaths 18th-century English people 19th-century English scientists 19th-century British physicists 19th-century British chemists Experimental physicists Optical physicists English chemists English inventors English physicists Fellows of the American Academy of Arts and Sciences Fellows of the Royal Society Foreign associates of the National Academy of Sciences Honorary members of the Saint Petersburg Academy of Sciences People from Elephant and Castle People associated with electricity Burials at Highgate Cemetery English Protestants Members of the French Academy of Sciences Members of the Royal Netherlands Academy of Arts and Sciences Members of the Royal Swedish Academy of Sciences Recipients of the Copley Medal Recipients of the Pour le Mérite (civil class) Royal Medal winners Glasites Writers about religion and science Magneticians