MLH1 on:
[Wikipedia]
[Google]
[Amazon]
DNA mismatch repair protein Mlh1 or MutL protein homolog 1 is a
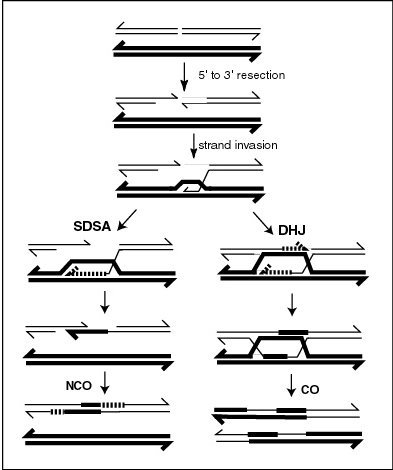 MLH1 protein appears to localize to sites of crossing over in meiotic chromosomes. Recombination during meiosis is often initiated by a DNA double-strand break (DSB) as illustrated in the accompanying diagram. During recombination, sections of DNA at the 5' ends of the break are cut away in a process called ''resection''. In the ''strand invasion'' step that follows, an overhanging 3' end of the broken DNA molecule then "invades" the DNA of an homologous chromosome that is not broken forming a displacement loop (
MLH1 protein appears to localize to sites of crossing over in meiotic chromosomes. Recombination during meiosis is often initiated by a DNA double-strand break (DSB) as illustrated in the accompanying diagram. During recombination, sections of DNA at the 5' ends of the break are cut away in a process called ''resection''. In the ''strand invasion'' step that follows, an overhanging 3' end of the broken DNA molecule then "invades" the DNA of an homologous chromosome that is not broken forming a displacement loop (
FAQs on HNPCC
from the
GeneReviews/NCBI/NIH/UW entry on Lynch syndrome
* * {{DNA repair
protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, respo ...
that in humans is encoded by the MLH1 gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a ba ...
located on chromosome 3
Chromosome 3 is one of the 23 pairs of chromosomes in humans. People normally have two copies of this chromosome. Chromosome 3 spans almost 200 million base pairs (the building material of DNA) and represents about 6.5 percent of the total DNA in ...
. It is a gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a ba ...
commonly associated with hereditary nonpolyposis colorectal cancer
Hereditary nonpolyposis colorectal cancer (HNPCC) or Lynch syndrome is an autosomal dominant genetic condition that is associated with a high risk of colon cancer as well as other cancers including endometrial cancer (second most common), ovary, ...
. Orthologs
Sequence homology is the biological homology between DNA, RNA, or protein sequences, defined in terms of shared ancestry in the evolutionary history of life. Two segments of DNA can have shared ancestry because of three phenomena: either a spec ...
of human MLH1 have also been studied in other organisms including mouse and the budding yeast ''Saccharomyces cerevisiae
''Saccharomyces cerevisiae'' () (brewer's yeast or baker's yeast) is a species of yeast (single-celled fungus microorganisms). The species has been instrumental in winemaking, baking, and brewing since ancient times. It is believed to have been o ...
''.
Function
This gene was identified as a locus frequently mutated in hereditary nonpolyposis colon cancer. It is a humanhomolog
In biology, homology is similarity due to shared ancestry between a pair of structures or genes in different taxa. A common example of homologous structures is the forelimbs of vertebrates, where the wings of bats and birds, the arms of prima ...
of the ''E. coli'' DNA mismatch repair gene, mutL, which mediates protein-protein interactions during mismatch recognition, strand discrimination, and strand removal. Defects in MLH1 are associated with the microsatellite instability
Microsatellite instability (MSI) is the condition of genetic hypermutability (predisposition to mutation) that results from impaired DNA mismatch repair (MMR). The presence of MSI represents phenotypic evidence that MMR is not functioning normal ...
observed in hereditary nonpolyposis colon cancer. Alternatively spliced transcript variants encoding different isoforms have been described, but their full-length natures have not been determined.
Role in DNA mismatch repair
MLH1 protein is one component of a system of sevenDNA mismatch repair
DNA mismatch repair (MMR) is a system for recognizing and repairing erroneous insertion, deletion, and mis-incorporation of nucleobase, bases that can arise during DNA replication and Genetic recombination, recombination, as well as DNA repair, r ...
proteins that work coordinately in sequential steps to initiate repair of DNA mismatches in humans. Defects in mismatch repair, found in about 13% of colorectal cancers, are much more frequently due to deficiency of MLH1 than deficiencies of other DNA mismatch repair proteins. The seven DNA mismatch repair proteins in humans are MLH1, MLH3
DNA mismatch repair protein Mlh3 is a protein that in humans is encoded by the ''MLH3'' gene.
Function
This gene is a member of the MutL-homolog (MLH) family of DNA mismatch repair (MMR) genes. MLH genes are implicated in maintaining genomic in ...
, MSH2
DNA mismatch repair protein Msh2 also known as MutS homolog 2 or MSH2 is a protein that in humans is encoded by the ''MSH2'' gene, which is located on chromosome 2. MSH2 is a tumor suppressor gene and more specifically a caretaker gene that codes ...
, MSH3
DNA mismatch repair protein, MutS Homolog 3 (MSH3) is a human homologue of the bacterial mismatch repair protein MutS that participates in the mismatch repair (MMR) system. MSH3 typically forms the heterodimer MutSβ with MSH2 in order to correc ...
, MSH6
MSH6 or mutS homolog 6 is a gene that codes for DNA mismatch repair protein Msh6 in the budding yeast ''Saccharomyces cerevisiae''. It is the homologue of the human "G/T binding protein," (GTBP) also called p160 or hMSH6 (human MSH6). The MSH6 prot ...
, PMS1
PMS1 protein homolog 1 is a protein that in humans is encoded by the ''PMS1'' gene.
Function
The protein encoded by this gene was identified by its homology to a yeast protein involved in DNA mismatch repair. A role for this protein in mismatch ...
and PMS2
Mismatch repair endonuclease PMS2 is an enzyme that in humans is encoded by the ''PMS2'' gene.
Function
This gene is one of the PMS2 gene family members which are found in clusters on chromosome 7. Human PMS2 related genes are located at bands ...
. In addition, there are Exo1
Exonuclease 1 is an enzyme that in humans is encoded by the ''EXO1'' gene.
This gene encodes a protein with 5' to 3' exonuclease activity as well as RNase activity (endonuclease activity cleaving RNA on DNA/RNA hybrid). It is similar to the Saccha ...
-dependent and Exo1-independent DNA mismatch repair subpathways.
DNA mismatches occur where one base is improperly paired with another base, or where there is a short addition or deletion in one strand of DNA that is not matched in the other strand. Mismatches commonly occur as a result of DNA replication errors or during genetic recombination. Recognizing those mismatches and repairing them is important for cells because failure to do so results in microsatellite instability] and an elevated spontaneous mutation rate
In genetics, the mutation rate is the frequency of new mutations in a single gene or organism over time. Mutation rates are not constant and are not limited to a single type of mutation; there are many different types of mutations. Mutation rates ...
(mutator phenotype). Among 20 cancers evaluated, microsatellite instable colon cancer (mismatch repair deficient) had the second highest frequency of mutations (after melanoma).
A heterodimer between MSH2 and MSH6 first recognizes the mismatch, although a heterodimer between MSH2 and MSH3 also can start the process. The formation of the MSH2-MSH6 heterodimer accommodates a second heterodimer of MLH1 and PMS2, although a heterodimer between MLH1 and either PMS3 or MLH3 can substitute for PMS2. This protein complex formed between the 2 sets of heterodimers enables initiation of repair of the mismatch defect.
Other gene products involved in mismatch repair (subsequent to initiation by DNA mismatch repair genes) include DNA polymerase delta
DNA polymerase delta (DNA Pol δ) is an enzyme complex found in eukaryotes that is involved in DNA replication and repair. The DNA polymerase delta complex consists of 4 subunits: POLD1, POLD2, POLD3, and POLD4. DNA Pol δ is an enzyme used for ...
, PCNA
Proliferating cell nuclear antigen (PCNA) is a DNA clamp that acts as a processivity factor for DNA polymerase δ in eukaryotic cells and is essential for replication. PCNA is a homotrimer and achieves its processivity by encircling the DNA, wh ...
, RPA, HMGB1
High mobility group box 1 protein, also known as high-mobility group protein 1 (HMG-1) and amphoterin, is a protein that in humans is encoded by the ''HMGB1'' gene.
HMG-1 belongs to the high mobility group and contains a HMG-box domain.
Functi ...
, RFC
RFC may refer to:
Computing
* Request for Comments, a memorandum on Internet standards
* Request for change, change management
* Remote Function Call, in SAP computer systems
* Rhye's and Fall of Civilization, a modification for Sid Meier's Civ ...
and DNA ligase I
DNA ligase 1 is an enzyme that in humans is encoded by the ''LIG1'' gene. DNA ligase I is the only known eukaryotic DNA ligase involved in both DNA replication and repair, making it the most studied of the ligases.
Discovery
It was known that ...
, plus histone
In biology, histones are highly basic proteins abundant in lysine and arginine residues that are found in eukaryotic cell nuclei. They act as spools around which DNA winds to create structural units called nucleosomes. Nucleosomes in turn a ...
and chromatin
Chromatin is a complex of DNA and protein found in eukaryotic cells. The primary function is to package long DNA molecules into more compact, denser structures. This prevents the strands from becoming tangled and also plays important roles in r ...
modifying factors.
Deficient expression in cancer
Epigenetic repression
Only a minority of sporadic cancers with a DNA repair deficiency have a mutation in a DNA repair gene. However, a majority of sporadic cancers with a DNA repair deficiency do have one or moreepigenetic
In biology, epigenetics is the study of stable phenotypic changes (known as ''marks'') that do not involve alterations in the DNA sequence. The Greek prefix '' epi-'' ( "over, outside of, around") in ''epigenetics'' implies features that are "o ...
alterations that reduce or silence DNA repair gene expression. In the table above, the majority of deficiencies of MLH1 were due to methylation of the promoter region of the ''MLH1'' gene. Another epigenetic mechanism reducing MLH1 expression is over-expression of miR-155
MiR-155 is a microRNA that in humans is encoded by the ''MIR155'' host gene or ''MIR155HG''. MiR-155 plays a role in various physiological and pathological processes. Exogenous molecular control ''in vivo'' of miR-155 expression may inhibit mali ...
. MiR-155 targets MLH1 and MSH2 and an inverse correlation between the expression of miR-155 and the expression of MLH1 or MSH2 proteins was found in human colorectal cancer.
Deficiency in field defects
A field defect is an area or "field" of epithelium that has been preconditioned by epigenetic changes and/or mutations so as to predispose it towards development of cancer. As pointed out by Rubin, "The vast majority of studies in cancer research has been done on well-defined tumors ''in vivo,'' or on discrete neoplastic foci ''in vitro.'' Yet there is evidence that more than 80% of the somatic mutations found in mutator phenotype human colorectal tumors occur before the onset of terminal clonal expansion." Similarly, Vogelstein et al. point out that more than half of somatic mutations identified in tumors occurred in a pre-neoplastic phase (in a field defect), during growth of apparently normal cells. In the Table above, MLH1 deficiencies were noted in the field defects (histologically normal tissues) surrounding most of the cancers. If MLH1 is epigenetically reduced or silenced, it would not likely confer a selective advantage upon a stem cell. However, reduced or absent expression of MLH1 would cause increased rates of mutation, and one or more of the mutated genes may provide the cell with a selective advantage. The expression-deficient ''MLH1'' gene could then be carried along as a selectively neutral or only slightly deleterious passenger (hitch-hiker) gene when the mutated stem cell generates an expanded clone. The continued presence of a clone with an epigenetically repressed ''MLH1'' would continue to generate further mutations, some of which could produce a tumor.Repression in coordination with other DNA repair genes
In a cancer, multiple DNA repair genes are often found to be simultaneously repressed. In one example, involving ''MLH1'', Jiang et al. conducted a study where they evaluated the mRNA expression of 27 DNA repair genes in 40astrocytoma
Astrocytomas are a type of brain tumor. They originate in a particular kind of glial cells, star-shaped brain cells in the cerebrum called astrocytes. This type of tumor does not usually spread outside the brain and spinal cord and it does not usu ...
s compared to normal brain tissues from non-astrocytoma individuals. Among the 27 DNA repair genes evaluated, 13 DNA repair genes, ''MLH1'', ''MLH3
DNA mismatch repair protein Mlh3 is a protein that in humans is encoded by the ''MLH3'' gene.
Function
This gene is a member of the MutL-homolog (MLH) family of DNA mismatch repair (MMR) genes. MLH genes are implicated in maintaining genomic in ...
'', ''MGMT
MGMT () is an American indie rock band formed in 2002 in Middletown, Connecticut. It was founded by multi-instrumentalists Andrew VanWyngarden and Ben Goldwasser. Alongside VanWyngarden and Goldwasser, MGMT's live lineup currently consists of ...
,'' ''NTHL1
Endonuclease III-like protein 1 is an enzyme that in humans is encoded by the ''NTHL1'' gene.
As reviewed by Li et al., NTHL1 is a bifunctional DNA glycosylase that has an associated beta-elimination activity. NTHL1 is usually involved in re ...
,'' '' OGG1,'' ''SMUG1
Single-strand selective monofunctional uracil DNA glycosylase is an enzyme that in humans is encoded by the ''SMUG1'' gene. SMUG1 is a glycosylase that removes uracil from single- and double-stranded DNA in nuclear chromatin, thus contributing to ...
,'' ''ERCC1
DNA excision repair protein ERCC-1 is a protein that in humans is encoded by the ''ERCC1'' gene. Together with ERCC4, ERCC1 forms the ERCC1-XPF enzyme complex that participates in DNA repair and DNA recombination.
Many aspects of these two gene ...
,'' ''ERCC2
__NOTOC__
ERCC2, or XPD is a protein involved in transcription-coupled nucleotide excision repair.
The XPD (ERCC2) gene encodes for a 2.3-kb mRNA containing 22 exons and 21 introns. The XPD protein contains 760 amino acids and is a polypeptide ...
'', ''ERCC3
XPB (xeroderma pigmentosum type B) is an ATP-dependent DNA helicase in humans that is a part of the TFIIH transcription factor complex.
Structure
The 3D-structure of the archaeal homolog of XPB has been solved by X-ray crystallography by Dr. Jo ...
'', ''ERCC4
ERCC4 is a protein designated as DNA repair endonuclease XPF that in humans is encoded by the ''ERCC4'' gene. Together with ERCC1, ERCC4 forms the ERCC1-XPF enzyme complex that participates in DNA repair and DNA recombination.
The nuclease enz ...
'', ''RAD50
DNA repair protein RAD50, also known as RAD50, is a protein that in humans is encoded by the ''RAD50'' gene.
Function
The protein encoded by this gene is highly similar to ''Saccharomyces cerevisiae'' Rad50, a protein involved in DNA double- ...
'', ''XRCC4
DNA repair protein XRCC4 also known as X-ray repair cross-complementing protein 4 or XRCC4 is a protein that in humans is encoded by the XRCC4 gene. In addition to humans, the XRCC4 protein is also expressed in many other metazoans, fungi and in ...
'' and ''XRCC5
Ku80 is a protein that, in humans, is encoded by the ''XRCC5'' gene. Together, Ku70 and Ku80 make up the Ku heterodimer, which binds to DNA double-strand break ends and is required for the non-homologous end joining (NHEJ) pathway of DNA repai ...
'' were all significantly down-regulated in all three grades (II, III and IV) of astrocytomas. The repression of these 13 genes in lower grade as well as in higher grade astrocytomas suggested that they may be important in early as well as in later stages of astrocytoma. In another example, Kitajima et al. found that immunoreactivity for MLH1 and MGMT
MGMT () is an American indie rock band formed in 2002 in Middletown, Connecticut. It was founded by multi-instrumentalists Andrew VanWyngarden and Ben Goldwasser. Alongside VanWyngarden and Goldwasser, MGMT's live lineup currently consists of ...
expression was closely correlated in 135 specimens of gastric cancer and loss of MLH1 and MGMTappeared to be synchronously accelerated during tumor progression.
Deficient expression of multiple DNA repair genes are often found in cancers, and may contribute to the thousands of mutations usually found in cancers (see Mutation frequencies in cancers).
Meiosis
In addition to its role in DNA mismatch repair, MLH1 protein is also involved inmeiotic
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately res ...
crossing over. MLH1 forms a heterodimer with MLH3
DNA mismatch repair protein Mlh3 is a protein that in humans is encoded by the ''MLH3'' gene.
Function
This gene is a member of the MutL-homolog (MLH) family of DNA mismatch repair (MMR) genes. MLH genes are implicated in maintaining genomic in ...
that appears to be necessary for oocyte
An oocyte (, ), oöcyte, or ovocyte is a female gametocyte or germ cell involved in reproduction. In other words, it is an immature ovum, or egg cell. An oocyte is produced in a female fetus in the ovary during female gametogenesis. The female ...
s to progress through metaphase II of meiosis
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately resu ...
. Female and male ''MLH1''(-/-) mutant mice are infertile, and sterility is associated with a reduced level of chiasmata. During spermatogenesis
Spermatogenesis is the process by which haploid spermatozoa develop from germ cells in the seminiferous tubules of the testis. This process starts with the mitotic division of the stem cells located close to the basement membrane of the tubule ...
in ''MLH1''(-/-) mutant mice chromosomes often separate prematurely and there is frequent arrest in the first division of meiosis. In humans, a common variant of the ''MLH1'' gene is associated with increased risk of sperm damage and male infertility.
 MLH1 protein appears to localize to sites of crossing over in meiotic chromosomes. Recombination during meiosis is often initiated by a DNA double-strand break (DSB) as illustrated in the accompanying diagram. During recombination, sections of DNA at the 5' ends of the break are cut away in a process called ''resection''. In the ''strand invasion'' step that follows, an overhanging 3' end of the broken DNA molecule then "invades" the DNA of an homologous chromosome that is not broken forming a displacement loop (
MLH1 protein appears to localize to sites of crossing over in meiotic chromosomes. Recombination during meiosis is often initiated by a DNA double-strand break (DSB) as illustrated in the accompanying diagram. During recombination, sections of DNA at the 5' ends of the break are cut away in a process called ''resection''. In the ''strand invasion'' step that follows, an overhanging 3' end of the broken DNA molecule then "invades" the DNA of an homologous chromosome that is not broken forming a displacement loop (D-loop
In molecular biology, a displacement loop or D-loop is a DNA structure where the two strands of a double-stranded DNA molecule are separated for a stretch and held apart by a third strand of DNA. An R-loop is similar to a D-loop, but in this ca ...
). After strand invasion, the further sequence of events may follow either of two main pathways leading to a crossover (CO) or a non-crossover (NCO) recombinant (see Genetic recombination
Genetic recombination (also known as genetic reshuffling) is the exchange of genetic material between different organisms which leads to production of offspring with combinations of traits that differ from those found in either parent. In eukaryo ...
). The pathway leading to a CO involves a double Holliday junction
A Holliday junction is a branched nucleic acid structure that contains four double-stranded arms joined. These arms may adopt one of several conformations depending on buffer salt concentrations and the sequence of nucleobases closest to the ju ...
(DHJ) intermediate. Holliday junctions need to be resolved for CO recombination to be completed.
In the budding yeast ''Saccharomyces cerevisiae
''Saccharomyces cerevisiae'' () (brewer's yeast or baker's yeast) is a species of yeast (single-celled fungus microorganisms). The species has been instrumental in winemaking, baking, and brewing since ancient times. It is believed to have been o ...
'', as in the mouse, MLH1 forms a heterodimer with MLH3. Meiotic CO requires resolution of Holliday junction
A Holliday junction is a branched nucleic acid structure that contains four double-stranded arms joined. These arms may adopt one of several conformations depending on buffer salt concentrations and the sequence of nucleobases closest to the ju ...
s through actions of the MLH1-MLH3 heterodimer
In biochemistry, a protein dimer is a macromolecular complex formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ''dimer'' has ...
. The MLH1-MLH3 heterodimer
In biochemistry, a protein dimer is a macromolecular complex formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ''dimer'' has ...
is an endonuclease
Endonucleases are enzymes that cleave the phosphodiester bond within a polynucleotide chain. Some, such as deoxyribonuclease I, cut DNA relatively nonspecifically (without regard to sequence), while many, typically called restriction endonucleases ...
that makes single-strand breaks in supercoiled double-stranded DNA. MLH1-MLH3 binds specifically to Holliday junctions and may act as part of a larger complex to process Holliday junctions during meiosis
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately resu ...
. MLH1-MLH3 heterodimer (MutL gamma) together with EXO1
Exonuclease 1 is an enzyme that in humans is encoded by the ''EXO1'' gene.
This gene encodes a protein with 5' to 3' exonuclease activity as well as RNase activity (endonuclease activity cleaving RNA on DNA/RNA hybrid). It is similar to the Saccha ...
and Sgs1
Sgs1, also known as slow growth suppressor 1, is a DNA helicase protein found in ''Saccharomyces cerevisiae''. It is a homolog of the bacterial RecQ helicase. Like the other members of the RecQ helicase family, Sgs1 is important for DNA repair ...
(ortholog of Bloom syndrome helicase) define a joint molecule resolution pathway that produces the majority of crossovers in budding yeast and, by inference, in mammals.
Clinical significance
It can also be associated withTurcot syndrome
Mismatch repair cancer syndrome (MMRCS) is a cancer syndrome associated with biallelic DNA mismatch repair mutations. It is also known as Turcot syndrome (after Jacques Turcot, who described the condition in 1959) and by several other names.
In M ...
.
Interactions
MLH1 has been shown tointeract
Advocates for Informed Choice, dba interACT or interACT Advocates for Intersex Youth, is a 501(c)(3) nonprofit organization using innovative strategies to advocate for the legal and human rights of children with intersex traits. The organizati ...
with:
* Bloom syndrome protein
Bloom syndrome protein is a protein that in humans is encoded by the ''BLM'' gene and is not expressed in Bloom syndrome.
The Bloom syndrome gene product is related to the RecQ subset of DExH box-containing DNA helicases and has both DNA-stimula ...
* Exonuclease 1
Exonuclease 1 is an enzyme that in humans is encoded by the ''EXO1'' gene.
This gene encodes a protein with 5' to 3' exonuclease activity as well as RNase activity (endonuclease activity cleaving RNA on DNA/RNA hybrid). It is similar to the Sacch ...
,
* MBD4
Methyl-CpG-binding domain protein 4 is a protein that in humans is encoded by the ''MBD4'' gene.
Structure
Human MBD4 protein has 580 amino acids with a methyl-CpG-binding domain at amino acids 82–147 and a C-terminal DNA glycosylase domain at ...
,
* MSH4,
* Myc
''Myc'' is a family of regulator genes and proto-oncogenes that code for transcription factors. The ''Myc'' family consists of three related human genes: ''c-myc'' (MYC), ''l-myc'' ( MYCL), and ''n-myc'' (MYCN). ''c-myc'' (also sometimes refe ...
, and
* PMS2
Mismatch repair endonuclease PMS2 is an enzyme that in humans is encoded by the ''PMS2'' gene.
Function
This gene is one of the PMS2 gene family members which are found in clusters on chromosome 7. Human PMS2 related genes are located at bands ...
.
See also
* Mismatch repair#MutH: an endonuclease present in E. coli and SalmonellaReferences
Further reading
* * * * * * *External links
FAQs on HNPCC
from the
National Institute of Health
The National Institutes of Health, commonly referred to as NIH (with each letter pronounced individually), is the primary agency of the United States government responsible for biomedical and public health research. It was founded in the late 1 ...
GeneReviews/NCBI/NIH/UW entry on Lynch syndrome
* * {{DNA repair