Heparin on:
[Wikipedia]
[Google]
[Amazon]
Heparin, also known as unfractionated heparin (UFH), is a medication and naturally occurring
 Heparin acts as an anticoagulant, preventing the formation of clots and extension of existing clots within the blood. While heparin itself does not break down clots that have already formed (unlike tissue plasminogen activator), it allows the body's natural clot lysis mechanisms to work normally to break down clots that have formed. Heparin is generally used for anticoagulation for the following conditions:
* Acute coronary syndrome, e.g., NSTEMI
* Atrial fibrillation
* Deep-vein thrombosis and pulmonary embolism (both prevention and treatment)
* Other thrombotic states and conditions
*
Heparin acts as an anticoagulant, preventing the formation of clots and extension of existing clots within the blood. While heparin itself does not break down clots that have already formed (unlike tissue plasminogen activator), it allows the body's natural clot lysis mechanisms to work normally to break down clots that have formed. Heparin is generally used for anticoagulation for the following conditions:
* Acute coronary syndrome, e.g., NSTEMI
* Atrial fibrillation
* Deep-vein thrombosis and pulmonary embolism (both prevention and treatment)
* Other thrombotic states and conditions
*
 Heparin is given
Heparin is given
 Native heparin is a polymer with a
Native heparin is a polymer with a
File:IdoA(2S)-GlcNS(6S).png,
File:IdoA(2S)-GlcNS.png,
File:IdoA-GlcNS(6S).png,
File:GlcA-GlcNAc.png,
File:GlcA-GlcNS.png,
File:IdoA-GlcNS.png,
GlcA = β--
Jmol viewer
* B = van der Waals radius space filling model of ''A'' * C = 1HPN (all IdoA(2S) residues in 1C4 conformation
Jmol viewer
* D = van der Waals radius space filling model of ''C'' In these models, heparin adopts a helical conformation, the rotation of which places clusters of sulfate groups at regular intervals of about 17
 The lyases cleave heparin/HS by a beta elimination mechanism. This action generates an unsaturated double bond between C4 and C5 of the uronate residue. The C4-C5 unsaturated uronate is termed ΔUA or UA. It is a sensitive UV chromophore (max absorption at 232 nm) and allows the rate of an enzyme digest to be followed, as well as providing a convenient method for detecting the fragments produced by enzyme digestion.
The lyases cleave heparin/HS by a beta elimination mechanism. This action generates an unsaturated double bond between C4 and C5 of the uronate residue. The C4-C5 unsaturated uronate is termed ΔUA or UA. It is a sensitive UV chromophore (max absorption at 232 nm) and allows the rate of an enzyme digest to be followed, as well as providing a convenient method for detecting the fragments produced by enzyme digestion.
 At both 'high' (4) and 'low' (1.5) pH, deaminative cleavage occurs between GlcNS-GlcA and GlcNS-IdoA, albeit at a slower rate at the higher pH. The deamination reaction, and therefore chain cleavage, is regardless of O-sulfation carried by either monosaccharide unit.
At low pH, deaminative cleavage results in the release of inorganic SO4, and the conversion of GlcNS into anhydromannose (aMan). Low-pH nitrous acid treatment is an excellent method to distinguish N-sulfated polysaccharides such as heparin and HS from non N-sulfated polysaccharides such as
At both 'high' (4) and 'low' (1.5) pH, deaminative cleavage occurs between GlcNS-GlcA and GlcNS-IdoA, albeit at a slower rate at the higher pH. The deamination reaction, and therefore chain cleavage, is regardless of O-sulfation carried by either monosaccharide unit.
At low pH, deaminative cleavage results in the release of inorganic SO4, and the conversion of GlcNS into anhydromannose (aMan). Low-pH nitrous acid treatment is an excellent method to distinguish N-sulfated polysaccharides such as heparin and HS from non N-sulfated polysaccharides such as
History of heparin
{{Authority control Chemical substances for emergency medicine Glycosaminoglycans Heparins World Health Organization essential medicines Wikipedia medicine articles ready to translate Aldols Ophthalmology drugs
glycosaminoglycan
Glycosaminoglycans (GAGs) or mucopolysaccharides are long, linear polysaccharides consisting of repeating disaccharide units (i.e. two-sugar units). The repeating two-sugar unit consists of a uronic sugar and an amino sugar, except in the case ...
. Since heparins depend on the activity of antithrombin, they are considered anticoagulants. Specifically it is also used in the treatment of heart attacks
A myocardial infarction (MI), commonly known as a heart attack, occurs when blood flow decreases or stops to the coronary artery of the heart, causing damage to the heart muscle. The most common symptom is chest pain or discomfort which may ...
and unstable angina. It is given intravenously or by injection under the skin. Other uses for its anticoagulant properties include inside blood specimen test tubes and kidney dialysis machines.
Common side effects include bleeding, pain at the injection site, and low blood platelets. Serious side effects include heparin-induced thrombocytopenia
Heparin-induced thrombocytopenia (HIT) is the development of thrombocytopenia (a low platelet count), due to the administration of various forms of heparin, an anticoagulant. HIT predisposes to thrombosis (the abnormal formation of blood clots ...
. Greater care is needed in those with poor kidney function.
Heparin is contraindicated for suspected cases of vaccine-induced pro-thrombotic immune thrombocytopenia (VIPIT) secondary to SARS-CoV-2
Severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) is a strain of coronavirus that causes COVID-19 (coronavirus disease 2019), the respiratory illness responsible for the ongoing COVID-19 pandemic. The virus previously had a ...
vaccination, as heparin may further increase the risk of bleeding in an anti-PF4/heparin complex autoimmune manner, in favor of alternative anticoagulant medications (such as argatroban or danaparoid).
Heparin appears to be relatively safe for use during pregnancy
Pregnancy is the time during which one or more offspring develops ( gestates) inside a woman's uterus (womb). A multiple pregnancy involves more than one offspring, such as with twins.
Pregnancy usually occurs by sexual intercourse, but ...
and breastfeeding
Breastfeeding, or nursing, is the process by which human breast milk is fed to a child. Breast milk may be from the breast, or may be expressed by hand or pumped and fed to the infant. The World Health Organization (WHO) recommends that bre ...
. Heparin is produced by basophils and mast cells in all mammals.
The discovery of heparin was announced in 1916. It is on the World Health Organization's List of Essential Medicines. A fractionated
Fractionation is a separation process in which a certain quantity of a mixture (of gases, solids, liquids, enzymes, or isotopes, or a suspension) is divided during a phase transition, into a number of smaller quantities (fractions) in whi ...
version of heparin, known as low molecular weight heparin, is also available.
History
Heparin was discovered byJay McLean
Jay McLean (1890 – November 14, 1957) was an American surgeon. He is most notable for his major contribution to the discovery of heparin.
Early life
Born in San Francisco in 1890, he was the son of a physician, John T. McLean. McLean's fa ...
and William Henry Howell in 1916, although it did not enter clinical trials until 1935. It was originally isolated from dog liver
The liver is a major organ only found in vertebrates which performs many essential biological functions such as detoxification of the organism, and the synthesis of proteins and biochemicals necessary for digestion and growth. In humans, it i ...
cells, hence its name (''hepar'' or "ήπαρ" is Greek for "liver"; ''hepar'' + '' -in'').
McLean was a second-year medical student at Johns Hopkins University
Johns Hopkins University (Johns Hopkins, Hopkins, or JHU) is a private research university in Baltimore, Maryland. Founded in 1876, Johns Hopkins is the oldest research university in the United States and in the western hemisphere. It consi ...
, and was working under the guidance of Howell investigating pro-coagulant preparations, when he isolated a fat-soluble phosphatide anticoagulant in canine liver tissue. In 1918, Howell coined the term 'heparin' for this type of fat-soluble anticoagulant. In the early 1920s, Howell isolated a water-soluble polysaccharide anticoagulant, which he also termed 'heparin', although it was different from the previously discovered phosphatide preparations. McLean's work as a surgeon probably changed the focus of the Howell group to look for anticoagulants, which eventually led to the polysaccharide discovery.
In the 1930s, several researchers were investigating heparin. Erik Jorpes
Johan Erik Jorpes (born Johansson, 15 July 1894 – 10 July 1973) was a Finnish-born Swedish physician and biochemist. He identified the chemical structure of heparin and developed its clinical applications. Jorpes was the professor of medical c ...
at Karolinska Institutet published his research on the structure of heparin in 1935, which made it possible for the Swedish company Vitrum AB to launch the first heparin product for intravenous use in 1936. Between 1933 and 1936, Connaught Medical Research Laboratories
Sanofi S.A. is a French multinational pharmaceutical and healthcare company headquartered in Paris, France. Originally, the corporation was established in 1973 and merged with Synthélabo in 1999 to form Sanofi-Synthélabo. In 2004, Sanofi-Syn ...
, then a part of the University of Toronto, perfected a technique for producing safe, nontoxic heparin that could be administered to patients, in a saline solution. The first human trials of heparin began in May 1935, and, by 1937, it was clear that Connaught's heparin was safe, easily available, and effective as a blood anticoagulant. Prior to 1933, heparin was available in small amounts, was extremely expensive and toxic, and, as a consequence, of no medical value.
Heparin production experienced a break in the 1990s. Until then, heparin was mainly obtained from cattle tissue, which was a by-product of the meat industry, especially in North America. With the rapid spread of BSE, more and more manufacturers abandoned this source of supply. As a result, global heparin production became increasingly concentrated in China, where the substance was now procured from the expanding industry of breeding and slaughtering hog. The dependence of medical care on the meat industry assumed threatening proportions in the wake of the COVID-19 pandemic
The COVID-19 pandemic, also known as the coronavirus pandemic, is an ongoing global pandemic of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The novel virus was first identi ...
. In 2020, several studies demonstrated the efficacy of heparin in mitigating severe disease progression, as its anticoagulant effect counteracted the formation of immunothrombosis. However, the availability of heparin on the world market was decreased, because concurrently a renewed swine flu epidemic had reduced significant portions of the Chinese hog population. The situation was further exacerbated by the fact that mass slaughterhouses around the world became corona hotspots themselves and were forced to close temporarily. In less affluent countries, the resulting heparin shortage also led to worsened health care beyond the treatment of covid, for example through the cancellation of cardiac surgeries.
Medical use
 Heparin acts as an anticoagulant, preventing the formation of clots and extension of existing clots within the blood. While heparin itself does not break down clots that have already formed (unlike tissue plasminogen activator), it allows the body's natural clot lysis mechanisms to work normally to break down clots that have formed. Heparin is generally used for anticoagulation for the following conditions:
* Acute coronary syndrome, e.g., NSTEMI
* Atrial fibrillation
* Deep-vein thrombosis and pulmonary embolism (both prevention and treatment)
* Other thrombotic states and conditions
*
Heparin acts as an anticoagulant, preventing the formation of clots and extension of existing clots within the blood. While heparin itself does not break down clots that have already formed (unlike tissue plasminogen activator), it allows the body's natural clot lysis mechanisms to work normally to break down clots that have formed. Heparin is generally used for anticoagulation for the following conditions:
* Acute coronary syndrome, e.g., NSTEMI
* Atrial fibrillation
* Deep-vein thrombosis and pulmonary embolism (both prevention and treatment)
* Other thrombotic states and conditions
* Cardiopulmonary bypass
Cardiopulmonary bypass (CPB) is a technique in which a machine temporarily takes over the function of the heart and lungs during surgery, maintaining the circulation of blood and oxygen to the body. The CPB pump itself is often referred to as a ...
for heart surgery
* ECMO circuit for extracorporeal life support
* Hemofiltration
Hemofiltration, also haemofiltration, is a renal replacement therapy which is used in the intensive care setting. It is usually used to treat acute kidney injury (AKI), but may be of benefit in multiple organ dysfunction syndrome or sepsis. During ...
* Indwelling central or peripheral venous catheters
Heparin and its low-molecular-weight derivatives (e.g., enoxaparin, dalteparin, tinzaparin
Tinzaparin is an antithrombotic drug in the heparin group. It is a low molecular weight heparin (LMWH) marketed as Innohep worldwide. It has been approved by the U.S. Food and Drug Administration (FDA) for once daily treatment and prophylaxis of ...
) are effective in preventing deep vein thromboses and pulmonary emboli in people at risk, but no evidence indicates any one is more effective than the other in preventing mortality.
In angiography, 2 to 5 units/mL of unfractionated heparin saline flush is used to prevent the clotting of blood in guidewires, sheaths, and catheters, thus preventing thrombus from dislodging from these devices into the circulatory system .
Unfractionated heparin is used in hemodialysis. Comparing to low-molecular-weight heparin
Low-molecular-weight heparin (LMWH) is a class of anticoagulant medications. They are used in the prevention of blood clots and treatment of venous thromboembolism (deep vein thrombosis and pulmonary embolism) and in the treatment of myocardial in ...
, unfractionated heparin does not have prolonged anticoagulation action after dialysis, and low cost. However, the short duration of action for heparin would require it to maintain continuous infusion to maintain its action. Meanwhile, unfractionated heparin has higher risk of heparin-induced thrombocytopenia
Heparin-induced thrombocytopenia (HIT) is the development of thrombocytopenia (a low platelet count), due to the administration of various forms of heparin, an anticoagulant. HIT predisposes to thrombosis (the abnormal formation of blood clots ...
.
Adverse effects
A serious side-effect of heparin isheparin-induced thrombocytopenia
Heparin-induced thrombocytopenia (HIT) is the development of thrombocytopenia (a low platelet count), due to the administration of various forms of heparin, an anticoagulant. HIT predisposes to thrombosis (the abnormal formation of blood clots ...
(HIT), caused by an immunological reaction that makes platelets a target of immunological response, resulting in the degradation of platelets, which causes thrombocytopenia. This condition is usually reversed on discontinuation, and in general can be avoided with the use of synthetic heparins. Not all patients with heparin antibodies will develop thrombocytopenia. Also, a benign form of thrombocytopenia is associated with early heparin use, which resolves without stopping heparin. Approximately one-third of patients with diagnosed heparin-induced thrombocytopenia will ultimately develop thrombotic complications.
Two non-hemorrhagic side-effects of heparin treatment are known. The first is elevation of serum aminotransferase
Transaminases or aminotransferases are enzymes that catalyze a transamination reaction between an amino acid and an α-keto acid. They are important in the synthesis of amino acids, which form proteins.
Function and mechanism
An amino acid c ...
levels, which has been reported in as many as 80% of patients receiving heparin. This abnormality is not associated with liver dysfunction, and it disappears after the drug is discontinued. The other complication is hyperkalemia
Hyperkalemia is an elevated level of potassium (K+) in the blood. Normal potassium levels are between 3.5 and 5.0mmol/L (3.5 and 5.0mEq/L) with levels above 5.5mmol/L defined as hyperkalemia. Typically hyperkalemia does not cause symptoms. Occa ...
, which occurs in 5 to 10% of patients receiving heparin, and is the result of heparin-induced aldosterone suppression. The hyperkalemia can appear within a few days after the onset of heparin therapy. More rarely, the side-effects alopecia and osteoporosis can occur with chronic use.
As with many drugs, overdoses of heparin can be fatal. In September 2006, heparin received worldwide publicity when three prematurely born infants died after they were mistakenly given overdoses of heparin at an Indianapolis hospital.
Contraindications
Heparin is contraindicated in those with risk of bleeding (especially in people with uncontrolled blood pressure, liver disease, and stroke), severe liver disease, or severe hypertension.Antidote to heparin
Protamine sulfate
Protamine sulfate is a medication that is used to reverse the effects of heparin. It is specifically used in heparin overdose, in low molecular weight heparin overdose, and to reverse the effects of heparin during delivery and heart surgery. I ...
has been given to counteract the anticoagulant effect of heparin (1 mg per 100 units of heparin that had been given over the past 6 hours). It may be used in those who overdose on heparin or to reverse heparin's effect when it is no longer needed.
Physiological function
Heparin's normal role in the body is unclear. Heparin is usually stored within the secretory granules of mast cells and released only into the vasculature at sites of tissue injury. It has been proposed that, rather than anticoagulation, the main purpose of heparin is defense at such sites against invading bacteria and other foreign materials. In addition, it is observed across a number of widely different species, including some invertebrates that do not have a similar blood coagulation system. It is a highly sulfated glycosaminoglycan. It has the highest negativecharge density
In electromagnetism, charge density is the amount of electric charge per unit length, surface area, or volume. Volume charge density (symbolized by the Greek letter ρ) is the quantity of charge per unit volume, measured in the SI system i ...
of any known biological molecule.
Evolutionary conservation
In addition to the bovine and porcine tissue from which pharmaceutical-grade heparin is commonly extracted, it has also been extracted and characterized from: #Turkey
Turkey ( tr, Türkiye ), officially the Republic of Türkiye ( tr, Türkiye Cumhuriyeti, links=no ), is a transcontinental country located mainly on the Anatolian Peninsula in Western Asia, with a small portion on the Balkan Peninsula ...
# Whale
Whales are a widely distributed and diverse group of fully aquatic placental marine mammals. As an informal and colloquial grouping, they correspond to large members of the infraorder Cetacea, i.e. all cetaceans apart from dolphins and ...
# Dromedary camel
# Mouse
A mouse ( : mice) is a small rodent. Characteristically, mice are known to have a pointed snout, small rounded ears, a body-length scaly tail, and a high breeding rate. The best known mouse species is the common house mouse (''Mus musculus' ...
# Humans" \n\n\n\n\nThe robots exclusion standard, also known as the robots exclusion protocol or simply robots.txt, is a standard used by websites to indicate to visiting web crawlers and other web robots which portions of the site they are allowed to visi ...
# Lobster
# Fresh water mussel
# Clam
# Shrimp
Shrimp are crustaceans (a form of shellfish) with elongated bodies and a primarily swimming mode of locomotion – most commonly Caridea and Dendrobranchiata of the decapod order, although some crustaceans outside of this order are ref ...
# Mangrove crab
# Sand dollar
# Atlantic salmon
# Zebra fish
The biological activity of heparin within species 6–11 is unclear and further supports the idea that the main physiological role of heparin is not anticoagulation. These species do not possess any blood coagulation system similar to that present within the species listed 1–5. The above list also demonstrates how heparin has been highly evolutionarily conserved
In evolutionary biology, conserved sequences are identical or similar sequences in nucleic acids ( DNA and RNA) or proteins across species ( orthologous sequences), or within a genome ( paralogous sequences), or between donor and receptor taxa ( ...
, with molecules of a similar structure being produced by a broad range of organisms belonging to many different phyla Phyla, the plural of ''phylum'', may refer to:
* Phylum, a biological taxon between Kingdom and Class
* by analogy, in linguistics, a large division of possibly related languages, or a major language family which is not subordinate to another
Phy ...
.
Pharmacology
Innature
Nature, in the broadest sense, is the physical world or universe. "Nature" can refer to the phenomena of the physical world, and also to life in general. The study of nature is a large, if not the only, part of science. Although humans are ...
, heparin is a polymer
A polymer (; Greek '' poly-'', "many" + '' -mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
of varying chain size. Unfractionated heparin (UFH) as a pharmaceutical is heparin that has not been fractionated
Fractionation is a separation process in which a certain quantity of a mixture (of gases, solids, liquids, enzymes, or isotopes, or a suspension) is divided during a phase transition, into a number of smaller quantities (fractions) in whi ...
to sequester the fraction of molecules with low molecular weight
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bio ...
. In contrast, low-molecular-weight heparin
Low-molecular-weight heparin (LMWH) is a class of anticoagulant medications. They are used in the prevention of blood clots and treatment of venous thromboembolism (deep vein thrombosis and pulmonary embolism) and in the treatment of myocardial in ...
(LMWH) has undergone fractionation for the purpose of making its pharmacodynamics
Pharmacodynamics (PD) is the study of the biochemical and physiologic effects of drugs (especially pharmaceutical drugs). The effects can include those manifested within animals (including humans), microorganisms, or combinations of organisms ...
more predictable. Often either UFH or LMWH can be used; in some situations one or the other is preferable.
Mechanism of action
Heparin binds to the enzyme inhibitor antithrombin III (AT), causing a conformational change that results in its activation through an increase in the flexibility of its reactive site loop. The activated AT then inactivates thrombin,factor Xa
Factor X, also known by the eponym Stuart–Prower factor, is an enzyme () of the coagulation cascade. It is a serine endopeptidase (protease group S1, PA clan). Factor X is synthesized in the liver and requires vitamin K for its synthesis.
...
and other proteases. The rate of inactivation of these proteases by AT can increase by up to 1000-fold due to the binding of heparin. Heparin binds to AT via a specific pentasaccharide sulfation sequence contained within the heparin polymer:
: GlcNAc/NS(6S)-GlcA-GlcNS(3S,6S)-IdoA(2S)-GlcNS(6S)
The conformational change in AT on heparin-binding mediates its inhibition of factor Xa. For thrombin inhibition, however, thrombin must also bind to the heparin polymer at a site proximal to the pentasaccharide. The highly negative charge density of heparin contributes to its very strong electrostatic
Electrostatics is a branch of physics that studies electric charges at rest ( static electricity).
Since classical times, it has been known that some materials, such as amber, attract lightweight particles after rubbing. The Greek word for ...
interaction with thrombin. The formation of a ternary complex
A ternary complex is a protein complex containing three different molecules that are bound together. In structural biology, ''ternary complex'' can also be used to describe a crystal containing a protein with two small molecules bound, for exampl ...
between AT, thrombin, and heparin results in the inactivation of thrombin. For this reason, heparin's activity against thrombin is size-dependent, with the ternary complex requiring at least 18 saccharide units for efficient formation. In contrast, antifactor Xa activity via AT requires only the pentasaccharide-binding site.
This size difference has led to the development of low-molecular-weight heparin
Low-molecular-weight heparin (LMWH) is a class of anticoagulant medications. They are used in the prevention of blood clots and treatment of venous thromboembolism (deep vein thrombosis and pulmonary embolism) and in the treatment of myocardial in ...
s (LMWHs) and fondaparinux
Fondaparinux (trade name Arixtra) is an anticoagulant medication chemically related to low molecular weight heparins. It is marketed by GlaxoSmithKline. A generic version developed by Alchemia is marketed within the US by Dr. Reddy's Laboratories.
...
as anticoagulants. Fondaparinux targets anti-factor Xa activity rather than inhibiting thrombin activity, with the aim of facilitating a more subtle regulation of coagulation and an improved therapeutic index. It is a synthetic pentasaccharide, whose chemical structure is almost identical to the AT binding pentasaccharide sequence that can be found within polymeric heparin and heparan sulfate.
With LMWH and fondaparinux, the risk of osteoporosis and heparin-induced thrombocytopenia
Heparin-induced thrombocytopenia (HIT) is the development of thrombocytopenia (a low platelet count), due to the administration of various forms of heparin, an anticoagulant. HIT predisposes to thrombosis (the abnormal formation of blood clots ...
(HIT) is reduced. Monitoring of the activated partial thromboplastin time
The partial thromboplastin time (PTT), also known as the activated partial thromboplastin time (aPTT or APTT), is a blood test that characterizes coagulation of the blood. A historical name for this measure is the kaolin-cephalin clotting time ( ...
is also not required and does not reflect the anticoagulant effect, as APTT is insensitive to alterations in factor Xa.
Danaparoid, a mixture of heparan sulfate, dermatan sulfate
Dermatan sulfate is a glycosaminoglycan (formerly called a mucopolysaccharide) found mostly in skin, but also in blood vessels, heart valves, tendons, and lungs.
It is also referred to as chondroitin sulfate B, although it is no longer class ...
, and chondroitin sulfate
Chondroitin sulfate is a sulfated glycosaminoglycan (GAG) composed of a chain of alternating sugars ( N-acetylgalactosamine and glucuronic acid). It is usually found attached to proteins as part of a proteoglycan. A chondroitin chain can have ove ...
can be used as an anticoagulant in patients having developed HIT. Because danaparoid does not contain heparin or heparin fragments, cross-reactivity of danaparoid with heparin-induced antibodies is reported as less than 10%.
The effects of heparin are measured in the lab by the partial thromboplastin time (aPTT
The partial thromboplastin time (PTT), also known as the activated partial thromboplastin time (aPTT or APTT), is a blood test that characterizes coagulation of the blood. A historical name for this measure is the kaolin-cephalin clotting time ...
), one of the measures of the time it takes the blood plasma
Blood plasma is a light amber-colored liquid component of blood in which blood cells are absent, but contains proteins and other constituents of whole blood in suspension. It makes up about 55% of the body's total blood volume. It is the int ...
to clot. Partial thromboplastin time should not be confused with prothrombin time, or PT, which measures blood clotting time through a different pathway of the coagulation cascade
Coagulation, also known as clotting, is the process by which blood changes from a liquid to a gel, forming a blood clot. It potentially results in hemostasis, the cessation of blood loss from a damaged vessel, followed by repair. The mechani ...
.
Administration
 Heparin is given
Heparin is given parenteral
A route of administration in pharmacology and toxicology is the way by which a drug, fluid, poison, or other substance is taken into the body.
Routes of administration are generally classified by the location at which the substance is applied. ...
ly because it is not absorbed from the gut, due to its high negative charge and large size. It can be injected intravenously or subcutaneously (under the skin); intramuscular injections (into muscle) are avoided because of the potential for forming hematomas. Because of its short biologic half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ...
of about one hour, heparin must be given frequently or as a continuous infusion. Unfractionated heparin has a half-life of about one to two hours after infusion, whereas LMWH has a half-life of four to five hours. The use of LMWH has allowed once-daily dosing, thus not requiring a continuous infusion of the drug. If long-term anticoagulation is required, heparin is often used only to commence anticoagulation therapy until an oral anticoagulant e.g. warfarin takes effect.
The American College of Chest Physicians
The American College of Chest Physicians (CHEST) is a medical association in the United States consisting of physician
A physician (American English), medical practitioner (Commonwealth English), medical doctor, or simply doctor, is a ...
publishes clinical guidelines on heparin dosing.
Natural degradation or clearance
Unfractionated heparin has a half-life of about one to two hours after infusion, whereaslow-molecular-weight heparin
Low-molecular-weight heparin (LMWH) is a class of anticoagulant medications. They are used in the prevention of blood clots and treatment of venous thromboembolism (deep vein thrombosis and pulmonary embolism) and in the treatment of myocardial in ...
's half-life is about four times longer. Lower doses of heparin have a much shorter half-life than larger ones. Heparin binding to macrophage
Macrophages (abbreviated as M φ, MΦ or MP) ( el, large eaters, from Greek ''μακρός'' (') = large, ''φαγεῖν'' (') = to eat) are a type of white blood cell of the immune system that engulfs and digests pathogens, such as cancer ce ...
cells is internalized and depolymerized by the macrophages. It also rapidly binds to endothelial cells, which precludes the binding to antithrombin that results in anticoagulant action. For higher doses of heparin, endothelial cell binding will be saturated, such that clearance of heparin from the bloodstream by the kidneys will be a slower process.
Chemistry
Heparin structure
 Native heparin is a polymer with a
Native heparin is a polymer with a molecular weight
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bio ...
ranging from 3 to 30 kDa, although the average molecular weight of most commercial heparin preparations is in the range of 12 to 15 kDa. Heparin is a member of the glycosaminoglycan
Glycosaminoglycans (GAGs) or mucopolysaccharides are long, linear polysaccharides consisting of repeating disaccharide units (i.e. two-sugar units). The repeating two-sugar unit consists of a uronic sugar and an amino sugar, except in the case ...
family of carbohydrates
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may or m ...
(which includes the closely related molecule heparan sulfate) and consists of a variably sulfated repeating disaccharide unit.
The main disaccharide units that occur in heparin are shown below. The most common disaccharide unit* (see below) is composed of a 2-O-sulfated iduronic acid
-Iduronic acid (IUPAC abbr.: IdoA) is the major uronic acid component of the glycosaminoglycans (GAGs) dermatan sulfate, and heparin. It is also present in heparan sulfate
Heparan sulfate (HS) is a linear polysaccharide found in all animal tiss ...
and 6-O-sulfated, N-sulfated glucosamine, IdoA(2S)-GlcNS(6S). For example, this makes up 85% of heparins from beef lung and about 75% of those from porcine intestinal mucosa.
Not shown below are the rare disaccharides containing a 3-O-sulfated glucosamine (GlcNS(3S,6S)) or a free amine group (GlcNH3+). Under physiological conditions, the ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides ...
and amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it i ...
sulfate groups are deprotonated and attract positively charged counterions to form a heparin salt. Heparin is usually administered in this form as an anticoagulant.
glucuronic acid
Glucuronic acid (from Greek γλεῦκος "''wine, must''" and οὖρον "''urine''") is a uronic acid that was first isolated from urine (hence the name). It is found in many gums such as gum arabic (c. 18%), xanthan, and kombucha tea ...
, IdoA = α--iduronic acid
-Iduronic acid (IUPAC abbr.: IdoA) is the major uronic acid component of the glycosaminoglycans (GAGs) dermatan sulfate, and heparin. It is also present in heparan sulfate
Heparan sulfate (HS) is a linear polysaccharide found in all animal tiss ...
, IdoA(2S) = 2-''O''-sulfo-α--iduronic acid, GlcNAc = 2-deoxy-2-acetamido-α--glucopyranosyl, GlcNS = 2-deoxy-2-sulfamido-α--glucopyranosyl, GlcNS(6S) = 2-deoxy-2-sulfamido-α--glucopyranosyl-6-''O''-sulfate
One unit of heparin (the " Howell unit") is an amount approximately equivalent to 0.002 mg of pure heparin, which is the quantity required to keep 1 ml of cat's blood fluid for 24 hours at 0 °C.
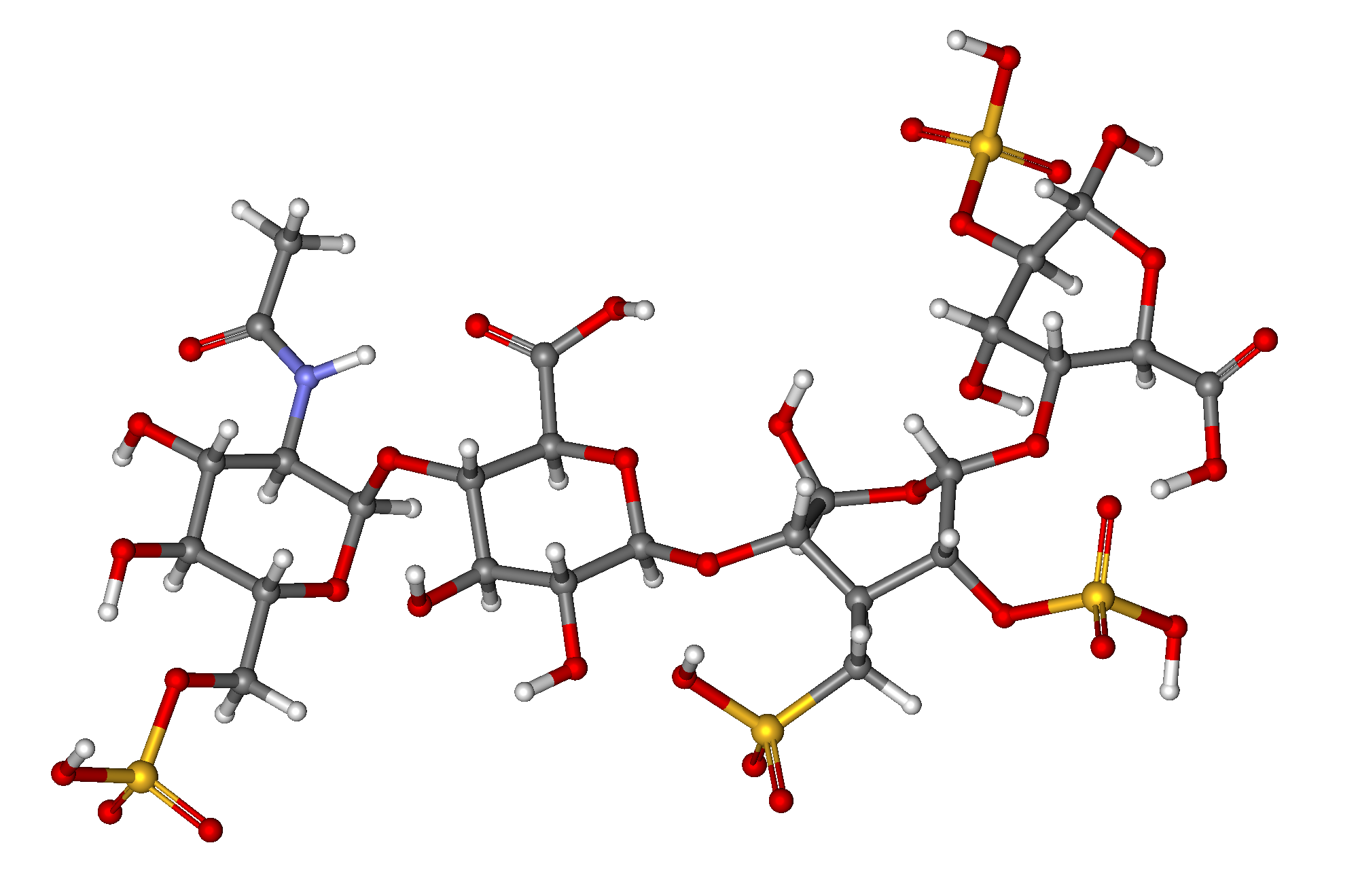
Three-dimensional structure
The three-dimensional structure of heparin is complicated becauseiduronic acid
-Iduronic acid (IUPAC abbr.: IdoA) is the major uronic acid component of the glycosaminoglycans (GAGs) dermatan sulfate, and heparin. It is also present in heparan sulfate
Heparan sulfate (HS) is a linear polysaccharide found in all animal tiss ...
may be present in either of two low-energy conformations when internally positioned within an oligosaccharide. The conformational equilibrium is influenced by sulfation state of adjacent glucosamine sugars. Nevertheless, the solution structure of a heparin dodecasaccharide composed solely of six GlcNS(6S)-IdoA(2S) repeat units has been determined using a combination of NMR spectroscopy and molecular modeling techniques. Two models were constructed, one in which all IdoA(2S) were in the 2S0 conformation (A and B below), and one in which they are in the 1C4 conformation (C and D below). However, no evidence suggests that changes between these conformations occur in a concerted fashion. These models correspond to the protein data bank code 1HPN.
In the image above:
* A = 1HPN (all IdoA(2S) residues in 2S0 conformationJmol viewer
* B = van der Waals radius space filling model of ''A'' * C = 1HPN (all IdoA(2S) residues in 1C4 conformation
Jmol viewer
* D = van der Waals radius space filling model of ''C'' In these models, heparin adopts a helical conformation, the rotation of which places clusters of sulfate groups at regular intervals of about 17
angstrom
The angstromEntry "angstrom" in the Oxford online dictionary. Retrieved on 2019-03-02 from https://en.oxforddictionaries.com/definition/angstrom.Entry "angstrom" in the Merriam-Webster online dictionary. Retrieved on 2019-03-02 from https://www.m ...
s (1.7 nm) on either side of the helical axis.
Depolymerization techniques
Either chemical or enzymatic depolymerization techniques or a combination of the two underlie the vast majority of analyses carried out on the structure and function of heparin and heparan sulfate (HS).Enzymatic
The enzymes traditionally used to digest heparin or HS are naturally produced by the soil bacterium ''Pedobacter heparinus'' (formerly named ''Flavobacterium heparinum''). This bacterium is capable of using either heparin or HS as its sole carbon and nitrogen source. To do so, it produces a range of enzymes such as lyases, glucuronidases, sulfoesterases, and sulfamidases. The lyases have mainly been used in heparin/HS studies. The bacterium produces three lyases, heparinases I (), II (no EC number assigned) and III () and each has distinct substrate specificities as detailed below.Chemical
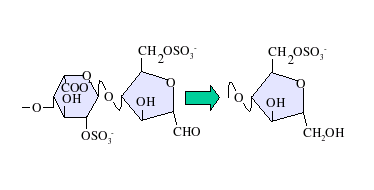
Nitrous acid can be used to chemically depolymerize heparin/HS. Nitrous acid can be used at pH 1.5 or at a higher pH of 4. Under both conditions, nitrous acid effects deaminative cleavage of the chain. At both 'high' (4) and 'low' (1.5) pH, deaminative cleavage occurs between GlcNS-GlcA and GlcNS-IdoA, albeit at a slower rate at the higher pH. The deamination reaction, and therefore chain cleavage, is regardless of O-sulfation carried by either monosaccharide unit.
At low pH, deaminative cleavage results in the release of inorganic SO4, and the conversion of GlcNS into anhydromannose (aMan). Low-pH nitrous acid treatment is an excellent method to distinguish N-sulfated polysaccharides such as heparin and HS from non N-sulfated polysaccharides such as
At both 'high' (4) and 'low' (1.5) pH, deaminative cleavage occurs between GlcNS-GlcA and GlcNS-IdoA, albeit at a slower rate at the higher pH. The deamination reaction, and therefore chain cleavage, is regardless of O-sulfation carried by either monosaccharide unit.
At low pH, deaminative cleavage results in the release of inorganic SO4, and the conversion of GlcNS into anhydromannose (aMan). Low-pH nitrous acid treatment is an excellent method to distinguish N-sulfated polysaccharides such as heparin and HS from non N-sulfated polysaccharides such as chondroitin sulfate
Chondroitin sulfate is a sulfated glycosaminoglycan (GAG) composed of a chain of alternating sugars ( N-acetylgalactosamine and glucuronic acid). It is usually found attached to proteins as part of a proteoglycan. A chondroitin chain can have ove ...
and dermatan sulfate
Dermatan sulfate is a glycosaminoglycan (formerly called a mucopolysaccharide) found mostly in skin, but also in blood vessels, heart valves, tendons, and lungs.
It is also referred to as chondroitin sulfate B, although it is no longer class ...
, chondroitin sulfate and dermatan sulfate not being susceptible to nitrous acid cleavage.
Detection in body fluids
Current clinical laboratory assays for heparin rely on an indirect measurement of the effect of the drug, rather than on a direct measure of its chemical presence. These includeactivated partial thromboplastin time
The partial thromboplastin time (PTT), also known as the activated partial thromboplastin time (aPTT or APTT), is a blood test that characterizes coagulation of the blood. A historical name for this measure is the kaolin-cephalin clotting time ( ...
(APTT) and antifactor Xa activity. The specimen of choice is usually fresh, nonhemolyzed plasma from blood that has been anticoagulated with citrate, fluoride, or oxalate.
Other functions
* Blood specimen test tubes, vacutainers, andcapillary
A capillary is a small blood vessel from 5 to 10 micrometres (μm) in diameter. Capillaries are composed of only the tunica intima, consisting of a thin wall of simple squamous endothelial cells. They are the smallest blood vessels in the bod ...
tubes that use the lithium
Lithium (from el, λίθος, lithos, lit=stone) is a chemical element with the symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard conditions, it is the least dense metal and the least dense soli ...
salt of heparin (lithium heparin) as an anticoagulant are usually marked with green stickers and green tops. Heparin has the advantage over EDTA of not affecting levels of most ions. However, the concentration of ionized calcium may be decreased if the concentration of heparin in the blood specimen is too high. Heparin can interfere with some immunoassay
An immunoassay (IA) is a biochemical test that measures the presence or concentration of a macromolecule or a small molecule in a solution through the use of an antibody (usually) or an antigen (sometimes). The molecule detected by the immunoa ...
s, however. As lithium heparin is usually used, a person's lithium levels cannot be obtained from these tubes; for this purpose, royal-blue-topped (and dark green-topped) vacutainers containing sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable ...
heparin are used.
* Heparin-coated blood oxygenator
An oxygenator is a medical device that is capable of exchanging oxygen and carbon dioxide in the blood of human patient during surgical procedures that may necessitate the interruption or cessation of blood flow in the body, a critical organ or ...
s are available for use in heart-lung machines. Among other things, these specialized oxygenators are thought to improve overall biocompatibility and host homeostasis by providing characteristics similar to those of native endothelium.
* The DNA binding sites on RNA polymerase can be occupied by heparin, preventing the polymerase from binding to promoter DNA. This property is exploited in a range of molecular biological assays.
* Common diagnostic procedures require PCR amplification of a patient's DNA, which is easily extracted from white blood cells treated with heparin. This poses a potential problem, since heparin may be extracted along with the DNA, and it has been found to interfere with the PCR reaction at levels as low as 0.002 U in a 50 μL reaction mixture.
* Heparin has been used as a chromatography resin, acting as both an affinity ligand and an ion exchanger. Its polyanionic structure can mimic nucleic acids like DNA and RNA, making it useful for purification of nucleic acid-binding proteins including DNA and RNA polymerases and transcription factor
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription of genetic information from DNA to messenger RNA, by binding to a specific DNA sequence. The f ...
s. Heparin's specific affinity for VSV-G, a viral envelope glycoprotein
Glycoproteins are proteins which contain oligosaccharide chains covalently attached to amino acid side-chains. The carbohydrate is attached to the protein in a cotranslational or posttranslational modification. This process is known as glyco ...
often used to pseudotype Pseudotyping is the process of producing viruses or viral vectors in combination with foreign viral envelope proteins. The result is a pseudotyped virus particle, also called a pseudovirus. With this method, the foreign viral envelope proteins can ...
retroviral
A retrovirus is a type of virus that inserts a DNA copy of its RNA genome into the DNA of a host cell that it invades, thus changing the genome of that cell. Once inside the host cell's cytoplasm, the virus uses its own reverse transcriptase ...
and lentiviral vectors for gene therapy
Gene therapy is a Medicine, medical field which focuses on the genetic modification of cells to produce a therapeutic effect or the treatment of disease by repairing or reconstructing defective genetic material. The first attempt at modifying ...
, allows it to be used for downstream purification of viral vectors.
* Heparin is being trialed in a nasal spray form as prophylaxis against COVID-19 infection. Furthermore, its reported from trials that due to anti-viral, anti-inflammatory and its anti-clotting effects its inhalation could improve at a 70% rate on patients that were actively struck by a COVID-19 infection.
Society and culture
Contamination recalls
Considering the animal source of pharmaceutical heparin, the numbers of potential impurities are relatively large compared with a wholly synthetic therapeutic agent. The range of possible biological contaminants includes viruses, bacterial endotoxins, transmissible spongiform encephalopathy (TSE) agents, lipids, proteins, and DNA. During the preparation of pharmaceutical-grade heparin from animal tissues, impurities such as solvents, heavy metals, and extraneous cations can be introduced. However, the methods employed to minimize the occurrence and to identify and/or eliminate these contaminants are well established and listed in guidelines and pharmacopoeias. The major challenge in the analysis of heparin impurities is the detection and identification of structurally related impurities. The most prevalent impurity in heparin is dermatan sulfate (DS), also known as chondroitin sulfate B. The building-block of DS is a disaccharide composed of 1,3-linked N-acetyl galactosamine (GalN) and a uronic acid residue, connected via 1,4 linkages to form the polymer. DS is composed of three possible uronic acid (GlcA, IdoA or IdoA2S) and four possible hexosamine (GalNAc, Gal- NAc4S, GalNAc6S, or GalNAc4S6S) building-blocks. The presence of iduronic acid in DS distinguishes it from chrondroitin sulfate A and C and likens it to heparin and HS. DS has a lower negative charge density overall compared to heparin. A common natural contaminant, DS is present at levels of 1–7% in heparin API, but has no proven biological activity that influences the anticoagulation effect of heparin. In December 2007, the Food and Drug Administration (United States), US Food and Drug Administration (FDA) recalled a shipment of heparin because of bacterial growth (''Serratia marcescens'') in several unopened syringes of this product. ''S. marcescens'' can lead to life-threatening injuries and/or death.2008 recall due to adulteration in drug from China
In March 2008, major Product recall, recalls of heparin were announced by the FDA due to contamination of the raw heparin stock imported from China. According to the FDA, the adulterated heparin killed nearly 80 people in the United States. The adulterant was identified as an "over-sulphated" derivative ofchondroitin sulfate
Chondroitin sulfate is a sulfated glycosaminoglycan (GAG) composed of a chain of alternating sugars ( N-acetylgalactosamine and glucuronic acid). It is usually found attached to proteins as part of a proteoglycan. A chondroitin chain can have ove ...
, a popular shellfish-derived supplement often used for arthritis, which was intended to substitute for actual heparin in potency tests.
According to the ''New York Times'': "Problems with heparin reported to the agency include difficulty breathing, nausea, vomiting, excessive sweating and rapidly falling blood pressure that in some cases led to life-threatening shock".
Use in homicide
In 2006, Petr Zelenka (serial killer), Petr Zelenka, a nurse in the Czech Republic, deliberately administered large doses to patients, killing seven, and attempting to kill ten others.Overdose issues
In 2007, a nurse at Cedars-Sinai Medical Center mistakenly gave the 12-day-old twins of actor Dennis Quaid a dose of heparin that was 1,000 times the recommended dose for infants. The overdose allegedly arose because the labeling and design of the adult and infant versions of the product were similar. The Quaid family subsequently sued the manufacturer, Baxter Healthcare Corp., and settled with the hospital for $750,000. Prior to the Quaid accident, six newborn babies at Methodist Hospital in Indianapolis, Indiana, were given an overdose. Three of the babies died after the mistake. In July 2008, another set of twins born at Christus Spohn Hospital South, in Corpus Christi, Texas, died after an accidentally administered overdose of the drug. The overdose was due to a mixing error at the hospital pharmacy and was unrelated to the product's packaging or labeling. , the exact cause of the twins' death was under investigation. In March 2010, a two-year-old transplant patient from Texas was given a lethal dose of heparin at the University of Nebraska Medical Center. The exact circumstances surrounding her death are still under investigation.Production
Pharmaceutical-grade heparin is derived from mucosal tissues of slaughterhouse, slaughtered meat animals such as porcine (pig) intestines or bovine (cattle) lungs. Advances to produce heparin synthetically have been made in 2003 and 2008. In 2011, a chemoenzymatic process of synthesizing low molecular weight heparins from simple disaccharides was reported.Research
As detailed in the table below, the potential is great for the development of heparin-like structures as drugs to treat a wide range of diseases, in addition to their current use as anticoagulants. : – indicates that no information is available As a result of heparin's effect on such a wide variety of disease states, a number of drugs are indeed in development whose molecular structures are identical or similar to those found within parts of the polymeric heparin chain.References
Further reading
* *External links
*History of heparin
{{Authority control Chemical substances for emergency medicine Glycosaminoglycans Heparins World Health Organization essential medicines Wikipedia medicine articles ready to translate Aldols Ophthalmology drugs