Heat flow on:
[Wikipedia]
[Google]
[Amazon]
 Heat transfer is a discipline of
Heat transfer is a discipline of
 Heat transfer is the energy exchanged between materials (solid/liquid/gas) as a result of a temperature difference. The
Heat transfer is the energy exchanged between materials (solid/liquid/gas) as a result of a temperature difference. The
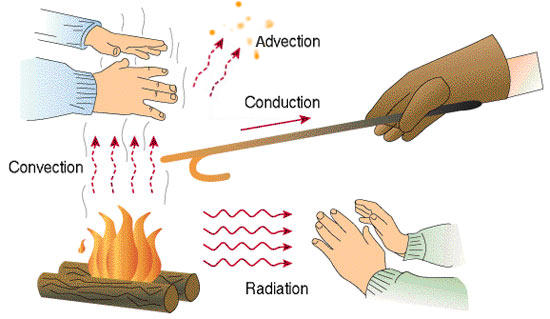 The fundamental modes of heat transfer are:
;
The fundamental modes of heat transfer are:
;
 Radiative heat transfer is the transfer of energy via
Radiative heat transfer is the transfer of energy via

 Heat transfer has broad application to the functioning of numerous devices and systems. Heat-transfer principles may be used to preserve, increase, or decrease temperature in a wide variety of circumstances. Heat transfer methods are used in numerous disciplines, such as
Heat transfer has broad application to the functioning of numerous devices and systems. Heat-transfer principles may be used to preserve, increase, or decrease temperature in a wide variety of circumstances. Heat transfer methods are used in numerous disciplines, such as

 The
The

A Heat Transfer Textbook
- (free download).
Thermal-FluidsPedia
- An online thermal fluids encyclopedia.
- Overview
- a practical example of how heat transfer is used to heat buildings without burning fossil fuels.
Thermal-Fluids CentralEnergy2D: Interactive Heat Transfer Simulations for Everyone
{{DEFAULTSORT:Heat Transfer Chemical engineering Mechanical engineering Unit operations Transport phenomena
 Heat transfer is a discipline of
Heat transfer is a discipline of thermal engineering
Thermal engineering is a specialized sub-discipline of mechanical engineering that deals with the movement of heat energy and transfer. The energy can be transferred between two mediums or transformed into other forms of energy. A thermal engineer ...
that concerns the generation, use, conversion, and exchange of thermal energy
The term "thermal energy" is used loosely in various contexts in physics and engineering. It can refer to several different well-defined physical concepts. These include the internal energy or enthalpy of a body of matter and radiation; heat, de ...
(heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is ...
) between physical systems. Heat transfer is classified into various mechanisms, such as thermal conduction
Conduction is the process by which heat is transferred from the hotter end to the colder end of an object. The ability of the object to conduct heat is known as its ''thermal conductivity'', and is denoted .
Heat spontaneously flows along a te ...
, thermal convection
Convection (or convective heat transfer) is the transfer of heat from one place to another due to the movement of fluid. Although often discussed as a distinct method of heat transfer, convective heat transfer involves the combined processes o ...
, thermal radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of particles in matter. Thermal radiation is generated when heat from the movement of charges in the material (electrons and protons in common forms of matter) i ...
, and transfer of energy by phase changes. Engineers also consider the transfer of mass of differing chemical species (mass transfer in the form of advection), either cold or hot, to achieve heat transfer. While these mechanisms have distinct characteristics, they often occur simultaneously in the same system.
Heat conduction, also called diffusion, is the direct microscopic exchanges of kinetic energy of particles (such as molecules) or quasiparticles (such as lattice waves) through the boundary between two systems. When an object is at a different temperature
Temperature is a physical quantity that expresses quantitatively the perceptions of hotness and coldness. Temperature is measured with a thermometer.
Thermometers are calibrated in various temperature scales that historically have relied o ...
from another body or its surroundings, heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is ...
flows so that the body and the surroundings reach the same temperature, at which point they are in thermal equilibrium
Two physical systems are in thermal equilibrium if there is no net flow of thermal energy between them when they are connected by a path permeable to heat. Thermal equilibrium obeys the zeroth law of thermodynamics. A system is said to be in ...
. Such spontaneous heat transfer always occurs from a region of high temperature to another region of lower temperature, as described in the second law of thermodynamics
The second law of thermodynamics is a physical law based on universal experience concerning heat and energy interconversions. One simple statement of the law is that heat always moves from hotter objects to colder objects (or "downhill"), unle ...
.
Heat convection occurs when the bulk flow of a fluid (gas or liquid) carries its heat through the fluid. All convective processes also move heat partly by diffusion, as well. The flow of fluid may be forced by external processes, or sometimes (in gravitational fields) by buoyancy forces caused when thermal energy expands the fluid (for example in a fire plume), thus influencing its own transfer. The latter process is often called "natural convection". The former process is often called "forced convection." In this case, the fluid is forced to flow by use of a pump, fan, or other mechanical means.
Thermal radiation occurs through a vacuum
A vacuum is a space devoid of matter. The word is derived from the Latin adjective ''vacuus'' for "vacant" or " void". An approximation to such vacuum is a region with a gaseous pressure much less than atmospheric pressure. Physicists often ...
or any transparent medium
Medium may refer to:
Science and technology
Aviation
* Medium bomber, a class of war plane
* Tecma Medium, a French hang glider design
Communication
* Media (communication), tools used to store and deliver information or data
* Medium ...
(solid
Solid is one of the four fundamental states of matter (the others being liquid, gas, and plasma). The molecules in a solid are closely packed together and contain the least amount of kinetic energy. A solid is characterized by structur ...
or fluid
In physics, a fluid is a liquid, gas, or other material that continuously deforms (''flows'') under an applied shear stress, or external force. They have zero shear modulus, or, in simpler terms, are substances which cannot resist any shear ...
or gas). It is the transfer of energy by means of photons
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are Massless particle, massless ...
or electromagnetic waves
In physics, electromagnetic radiation (EMR) consists of waves of the electromagnetic (EM) field, which propagate through space and carry momentum and electromagnetic radiant energy. It includes radio waves, microwaves, infrared, (visible) ...
governed by the same laws.
Overview
 Heat transfer is the energy exchanged between materials (solid/liquid/gas) as a result of a temperature difference. The
Heat transfer is the energy exchanged between materials (solid/liquid/gas) as a result of a temperature difference. The thermodynamic free energy
The thermodynamic free energy is a concept useful in the thermodynamics of chemical or thermal processes in engineering and science. The change in the free energy is the maximum amount of work that a thermodynamic system can perform in a process ...
is the amount of work that a thermodynamic system can perform. Enthalpy
Enthalpy , a property of a thermodynamic system, is the sum of the system's internal energy and the product of its pressure and volume. It is a state function used in many measurements in chemical, biological, and physical systems at a constant ...
is a thermodynamic potential
A thermodynamic potential (or more accurately, a thermodynamic potential energy)ISO/IEC 80000-5, Quantities an units, Part 5 - Thermodynamics, item 5-20.4 Helmholtz energy, Helmholtz functionISO/IEC 80000-5, Quantities an units, Part 5 - Thermod ...
, designated by the letter "H", that is the sum of the internal energy
The internal energy of a thermodynamic system is the total energy contained within it. It is the energy necessary to create or prepare the system in its given internal state, and includes the contributions of potential energy and internal kinet ...
of the system (U) plus the product of pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country a ...
(P) and volume
Volume is a measure of occupied three-dimensional space. It is often quantified numerically using SI derived units (such as the cubic metre and litre) or by various imperial or US customary units (such as the gallon, quart, cubic inch). ...
(V). Joule
The joule ( , ; symbol: J) is the unit of energy in the International System of Units (SI). It is equal to the amount of work done when a force of 1 newton displaces a mass through a distance of 1 metre in the direction of the force appli ...
is a unit to quantify energy
In physics, energy (from Ancient Greek: ἐνέργεια, ''enérgeia'', “activity”) is the quantitative property that is transferred to a body or to a physical system, recognizable in the performance of work and in the form of ...
, work, or the amount of heat.
Heat transfer is a process function
In thermodynamics, a quantity that is well defined so as to describe the path of a process through the equilibrium state space of a thermodynamic system is termed a process function, or, alternatively, a process quantity, or a path function. As ...
(or path function), as opposed to functions of state; therefore, the amount of heat transferred in a thermodynamic process
Classical thermodynamics considers three main kinds of thermodynamic process: (1) changes in a system, (2) cycles in a system, and (3) flow processes.
(1)A Thermodynamic process is a process in which the thermodynamic state of a system is change ...
that changes the state
State may refer to:
Arts, entertainment, and media Literature
* ''State Magazine'', a monthly magazine published by the U.S. Department of State
* ''The State'' (newspaper), a daily newspaper in Columbia, South Carolina, United States
* ''Our S ...
of a system
A system is a group of interacting or interrelated elements that act according to a set of rules to form a unified whole. A system, surrounded and influenced by its environment, is described by its boundaries, structure and purpose and express ...
depends on how that process occurs, not only the net difference between the initial and final states of the process.
Thermodynamic and mechanical
Mechanical may refer to:
Machine
* Machine (mechanical), a system of mechanisms that shape the actuator input to achieve a specific application of output forces and movement
* Mechanical calculator, a device used to perform the basic operations ...
heat transfer is calculated with the heat transfer coefficient
In thermodynamics, the heat transfer coefficient or film coefficient, or film effectiveness, is the proportionality constant between the heat flux and the thermodynamic driving force for the flow of heat (i.e., the temperature difference, ). ...
, the proportionality between the heat flux
Heat flux or thermal flux, sometimes also referred to as ''heat flux density'', heat-flow density or ''heat flow rate intensity'' is a flow of energy per unit area per unit time. In SI its units are watts per square metre (W/m2). It has both a ...
and the thermodynamic driving force for the flow of heat. Heat flux is a quantitative, vectorial representation of heat-flow through a surface.
In engineering contexts, the term ''heat'' is taken as synonymous to thermal energy. This usage has its origin in the historical interpretation of heat as a fluid (''caloric'') that can be transferred by various causes,
and that is also common in the language of laymen and everyday life.
The transport
Transport (in British English), or transportation (in American English), is the intentional movement of humans, animals, and goods from one location to another. Modes of transport include air, land ( rail and road), water, cable, pipelin ...
equations for thermal energy (Fourier's law
Conduction is the process by which heat is transferred from the hotter end to the colder end of an object. The ability of the object to conduct heat is known as its '' thermal conductivity'', and is denoted .
Heat spontaneously flows along a t ...
), mechanical momentum ( Newton's law for fluids), and mass transfer (Fick's laws of diffusion
Fick's laws of diffusion describe diffusion and were derived by Adolf Fick in 1855. They can be used to solve for the diffusion coefficient, . Fick's first law can be used to derive his second law which in turn is identical to the diffusion equ ...
) are similar,
and analogies among these three transport processes have been developed to facilitate prediction of conversion from any one to the others.
Thermal engineering
Thermal engineering is a specialized sub-discipline of mechanical engineering that deals with the movement of heat energy and transfer. The energy can be transferred between two mediums or transformed into other forms of energy. A thermal engineer ...
concerns the generation, use, conversion, storage, and exchange of heat transfer. As such, heat transfer is involved in almost every sector of the economy. Heat transfer is classified into various mechanisms, such as thermal conduction
Conduction is the process by which heat is transferred from the hotter end to the colder end of an object. The ability of the object to conduct heat is known as its ''thermal conductivity'', and is denoted .
Heat spontaneously flows along a te ...
, thermal convection
Convection (or convective heat transfer) is the transfer of heat from one place to another due to the movement of fluid. Although often discussed as a distinct method of heat transfer, convective heat transfer involves the combined processes o ...
, thermal radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of particles in matter. Thermal radiation is generated when heat from the movement of charges in the material (electrons and protons in common forms of matter) i ...
, and transfer of energy by phase changes.
Mechanisms
 The fundamental modes of heat transfer are:
;
The fundamental modes of heat transfer are:
;Advection
In the field of physics, engineering, and earth sciences, advection is the transport of a substance or quantity by bulk motion of a fluid. The properties of that substance are carried with it. Generally the majority of the advected substance is al ...
: Advection is the transport mechanism of a fluid
In physics, a fluid is a liquid, gas, or other material that continuously deforms (''flows'') under an applied shear stress, or external force. They have zero shear modulus, or, in simpler terms, are substances which cannot resist any shear ...
from one location to another, and is dependent on motion
In physics, motion is the phenomenon in which an object changes its position with respect to time. Motion is mathematically described in terms of displacement, distance, velocity, acceleration, speed and frame of reference to an observer and m ...
and momentum
In Newtonian mechanics, momentum (more specifically linear momentum or translational momentum) is the product of the mass and velocity of an object. It is a vector quantity, possessing a magnitude and a direction. If is an object's mass ...
of that fluid.
;Conduction
Conductor or conduction may refer to:
Music
* Conductor (music), a person who leads a musical ensemble, such as an orchestra.
* ''Conductor'' (album), an album by indie rock band The Comas
* Conduction, a type of structured free improvisation ...
or diffusion
Diffusion is the net movement of anything (for example, atoms, ions, molecules, energy) generally from a region of higher concentration to a region of lower concentration. Diffusion is driven by a gradient in Gibbs free energy or chemical ...
: The transfer of energy between objects that are in physical contact. Thermal conductivity
The thermal conductivity of a material is a measure of its ability to conduct heat. It is commonly denoted by k, \lambda, or \kappa.
Heat transfer occurs at a lower rate in materials of low thermal conductivity than in materials of high thermal ...
is the property of a material to conduct heat and evaluated primarily in terms of Fourier's Law
Conduction is the process by which heat is transferred from the hotter end to the colder end of an object. The ability of the object to conduct heat is known as its '' thermal conductivity'', and is denoted .
Heat spontaneously flows along a t ...
for heat conduction.
;Convection
Convection is single or multiphase fluid flow that occurs spontaneously due to the combined effects of material property heterogeneity and body forces on a fluid, most commonly density and gravity (see buoyancy). When the cause of the c ...
: The transfer of energy between an object and its environment, due to fluid motion. The average temperature is a reference for evaluating properties related to convective heat transfer.
;Radiation
In physics, radiation is the emission or transmission of energy in the form of waves or particles through space or through a material medium. This includes:
* ''electromagnetic radiation'', such as radio waves, microwaves, infrared, visi ...
: The transfer of energy by the emission of electromagnetic radiation
In physics, electromagnetic radiation (EMR) consists of waves of the electromagnetic (EM) field, which propagate through space and carry momentum and electromagnetic radiant energy. It includes radio waves, microwaves, infrared, (visib ...
.
Advection
By transferring matter, energy—including thermal energy—is moved by the physical transfer of a hot or cold object from one place to another. This can be as simple as placing hot water in a bottle and heating a bed, or the movement of an iceberg in changing ocean currents. A practical example isthermal hydraulics
Thermal hydraulics (also called thermohydraulics) is the study of hydraulic flow in thermal fluids. The area can be mainly divided into three parts: thermodynamics, fluid mechanics, and heat transfer, but they are often closely linked to each oth ...
. This can be described by the formula:
where
* is heat flux
Heat flux or thermal flux, sometimes also referred to as ''heat flux density'', heat-flow density or ''heat flow rate intensity'' is a flow of energy per unit area per unit time. In SI its units are watts per square metre (W/m2). It has both a ...
(W/m2),
* is density (kg/m3),
* is heat capacity at constant pressure (J/kg·K),
* is the difference in temperature (K),
* is velocity (m/s).
Conduction
On a microscopic scale, heat conduction occurs as hot, rapidly moving or vibrating atoms and molecules interact with neighboring atoms and molecules, transferring some of their energy (heat) to these neighboring particles. In other words, heat is transferred by conduction when adjacent atoms vibrate against one another, or as electrons move from one atom to another. Conduction is the most significant means of heat transfer within a solid or between solid objects inthermal contact
In heat transfer and thermodynamics, a thermodynamic system is said to be in thermal contact with another system if it can exchange energy through the process of heat. Perfect thermal isolation is an idealization as real systems are always in therm ...
. Fluids—especially gases—are less conductive. Thermal contact conductance is the study of heat conduction between solid bodies in contact. The process of heat transfer from one place to another place without the movement of particles is called conduction, such as when placing a hand on a cold glass of water—heat is conducted from the warm skin to the cold glass, but if the hand is held a few inches from the glass, little conduction would occur since air is a poor conductor of heat. Steady state conduction is an idealized model of conduction that happens when the temperature difference driving the conduction is constant, so that after a time, the spatial distribution of temperatures in the conducting object does not change any further (see Fourier's law
Conduction is the process by which heat is transferred from the hotter end to the colder end of an object. The ability of the object to conduct heat is known as its '' thermal conductivity'', and is denoted .
Heat spontaneously flows along a t ...
). In steady state conduction, the amount of heat entering a section is equal to amount of heat coming out, since the change in temperature (a measure of heat energy) is zero. An example of steady state conduction is the heat flow through walls of a warm house on a cold day—inside the house is maintained at a high temperature and, outside, the temperature stays low, so the transfer of heat per unit time stays near a constant rate determined by the insulation in the wall and the spatial distribution of temperature in the walls will be approximately constant over time.
''Transient conduction'' (see Heat equation
In mathematics and physics, the heat equation is a certain partial differential equation. Solutions of the heat equation are sometimes known as caloric functions. The theory of the heat equation was first developed by Joseph Fourier in 1822 for ...
) occurs when the temperature within an object changes as a function of time. Analysis of transient systems is more complex, and analytic solutions of the heat equation are only valid for idealized model systems. Practical applications are generally investigated using numerical methods, approximation techniques, or empirical study.
Convection
The flow of fluid may be forced by external processes, or sometimes (in gravitational fields) by buoyancy forces caused when thermal energy expands the fluid (for example in a fire plume), thus influencing its own transfer. The latter process is often called "natural convection". All convective processes also move heat partly by diffusion, as well. Another form of convection is forced convection. In this case the fluid is forced to flow by using a pump, fan or other mechanical means.Convective heat transfer
Convection (or convective heat transfer) is the transfer of heat from one place to another due to the movement of fluid. Although often discussed as a distinct method of heat transfer, convective heat transfer involves the combined processes o ...
, or simply, convection, is the transfer of heat from one place to another by the movement of fluids
In physics, a fluid is a liquid, gas, or other material that continuously deforms (''flows'') under an applied shear stress, or external force. They have zero shear modulus, or, in simpler terms, are substances which cannot resist any she ...
, a process that is essentially the transfer of heat via mass transfer
Mass transfer is the net movement of mass from one location (usually meaning stream, phase, fraction or component) to another. Mass transfer occurs in many processes, such as absorption, evaporation, drying, precipitation, membrane filtration ...
. Bulk motion of fluid enhances heat transfer in many physical situations, such as (for example) between a solid surface and the fluid. Convection is usually the dominant form of heat transfer in liquids and gases. Although sometimes discussed as a third method of heat transfer, convection is usually used to describe the combined effects of heat conduction within the fluid (diffusion) and heat transference by bulk fluid flow streaming. The process of transport by fluid streaming is known as advection, but pure advection is a term that is generally associated only with mass transport in fluids, such as advection of pebbles in a river. In the case of heat transfer in fluids, where transport by advection in a fluid is always also accompanied by transport via heat diffusion (also known as heat conduction) the process of heat convection is understood to refer to the sum of heat transport by advection and diffusion/conduction.
Free, or natural, convection occurs when bulk fluid motions (streams and currents) are caused by buoyancy forces that result from density variations due to variations of temperature in the fluid. ''Forced'' convection is a term used when the streams and currents in the fluid are induced by external means—such as fans, stirrers, and pumps—creating an artificially induced convection current.
Convection-cooling
Convective cooling is sometimes described asNewton's law of cooling
In the study of heat transfer, Newton's law of cooling is a physical law which states that
The rate of heat loss of a body is directly proportional to the difference in the temperatures between the body and its environment.
The law is frequently q ...
:
However, by definition, the validity of Newton's law of Cooling requires that the rate of heat loss from convection be a linear function of ("proportional to") the temperature difference that drives heat transfer, and in convective cooling this is sometimes not the case. In general, convection is not linearly dependent on temperature gradient
A temperature gradient is a physical quantity that describes in which direction and at what rate the temperature changes the most rapidly around a particular location. The temperature gradient is a dimensional quantity expressed in units of degre ...
s, and in some cases is strongly nonlinear. In these cases, Newton's law does not apply.
Convection vs. conduction
In a body of fluid that is heated from underneath its container, conduction and convection can be considered to compete for dominance. If heat conduction is too great, fluid moving down by convection is heated by conduction so fast that its downward movement will be stopped due to itsbuoyancy
Buoyancy (), or upthrust, is an upward force exerted by a fluid that opposes the weight of a partially or fully immersed object. In a column of fluid, pressure increases with depth as a result of the weight of the overlying fluid. Thus the ...
, while fluid moving up by convection is cooled by conduction so fast that its driving buoyancy will diminish. On the other hand, if heat conduction is very low, a large temperature gradient may be formed and convection might be very strong.
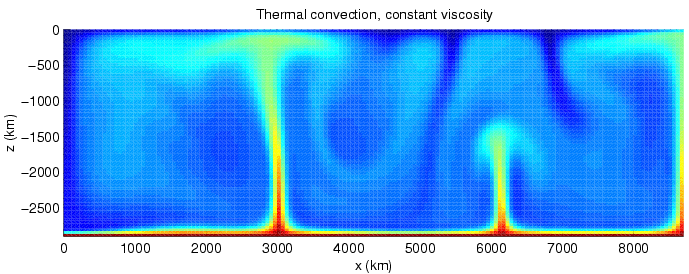
The Rayleigh number
In fluid mechanics, the Rayleigh number (, after Lord Rayleigh) for a fluid is a dimensionless number associated with buoyancy-driven flow, also known as free (or natural) convection. It characterises the fluid's flow regime: a value in a certai ...
() is the product of the Grashof () and Prandtl () numbers. It is a measure which determines the relative strength of conduction and convection.
where
*''g'' is acceleration due to gravity,
*''ρ'' is the density with being the density difference between the lower and upper ends,
*''μ'' is the dynamic viscosity
The viscosity of a fluid is a measure of its resistance to deformation at a given rate. For liquids, it corresponds to the informal concept of "thickness": for example, syrup has a higher viscosity than water.
Viscosity quantifies the int ...
,
*''α'' is the Thermal diffusivity
In heat transfer analysis, thermal diffusivity is the thermal conductivity divided by density and specific heat capacity at constant pressure. It measures the rate of transfer of heat of a material from the hot end to the cold end. It has the SI ...
,
*''β'' is the volume thermal expansivity (sometimes denoted ''α'' elsewhere),
*''T'' is the temperature,
*''ν'' is the kinematic viscosity
The viscosity of a fluid is a measure of its resistance to deformation at a given rate. For liquids, it corresponds to the informal concept of "thickness": for example, syrup has a higher viscosity than water.
Viscosity quantifies the int ...
, and
*''L'' is characteristic length.
The Rayleigh number can be understood as the ratio between the rate of heat transfer by convection to the rate of heat transfer by conduction; or, equivalently, the ratio between the corresponding timescales (i.e. conduction timescale divided by convection timescale), up to a numerical factor. This can be seen as follows, where all calculations are up to numerical factors depending on the geometry of the system.
The buoyancy force driving the convection is roughly , so the corresponding pressure is roughly . In steady state
In systems theory, a system or a process is in a steady state if the variables (called state variables) which define the behavior of the system or the process are unchanging in time. In continuous time, this means that for those properties ''p' ...
, this is canceled by the shear stress due to viscosity, and therefore roughly equals , where ''V'' is the typical fluid velocity due to convection and the order of its timescale. The conduction timescale, on the other hand, is of the order of .
Convection occurs when the Rayleigh number is above 1,000–2,000.
Radiation
 Radiative heat transfer is the transfer of energy via
Radiative heat transfer is the transfer of energy via thermal radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of particles in matter. Thermal radiation is generated when heat from the movement of charges in the material (electrons and protons in common forms of matter) i ...
, i.e., electromagnetic waves
In physics, electromagnetic radiation (EMR) consists of waves of the electromagnetic (EM) field, which propagate through space and carry momentum and electromagnetic radiant energy. It includes radio waves, microwaves, infrared, (visible) ...
. It occurs across vacuum
A vacuum is a space devoid of matter. The word is derived from the Latin adjective ''vacuus'' for "vacant" or " void". An approximation to such vacuum is a region with a gaseous pressure much less than atmospheric pressure. Physicists often ...
or any transparent medium
Medium may refer to:
Science and technology
Aviation
* Medium bomber, a class of war plane
* Tecma Medium, a French hang glider design
Communication
* Media (communication), tools used to store and deliver information or data
* Medium ...
(solid
Solid is one of the four fundamental states of matter (the others being liquid, gas, and plasma). The molecules in a solid are closely packed together and contain the least amount of kinetic energy. A solid is characterized by structur ...
or fluid
In physics, a fluid is a liquid, gas, or other material that continuously deforms (''flows'') under an applied shear stress, or external force. They have zero shear modulus, or, in simpler terms, are substances which cannot resist any shear ...
or gas). Thermal radiation is emitted by all objects at temperatures above absolute zero
Absolute zero is the lowest limit of the thermodynamic temperature scale, a state at which the enthalpy and entropy of a cooled ideal gas reach their minimum value, taken as zero kelvin. The fundamental particles of nature have minimum vibra ...
, due to random movements of atoms and molecules in matter. Since these atoms and molecules are composed of charged particles (proton
A proton is a stable subatomic particle, symbol , H+, or 1H+ with a positive electric charge of +1 ''e'' elementary charge. Its mass is slightly less than that of a neutron and 1,836 times the mass of an electron (the proton–electron mass ...
s and electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have n ...
s), their movement results in the emission of electromagnetic radiation
In physics, electromagnetic radiation (EMR) consists of waves of the electromagnetic (EM) field, which propagate through space and carry momentum and electromagnetic radiant energy. It includes radio waves, microwaves, infrared, (visib ...
which carries away energy. Radiation is typically only important in engineering applications for very hot objects, or for objects with a large temperature difference.
When the objects and distances separating them are large in size and compared to the wavelength of thermal radiation, the rate of transfer of radiant energy
Radiant may refer to:
Computers, software, and video games
* Radiant (software), a content management system
* GtkRadiant, a level editor created by id Software for their games
* Radiant AI, a technology developed by Bethesda Softworks for ''Th ...
is best described by the Stefan-Boltzmann equation. For an object in vacuum, the equation is:
For radiative transfer
Radiative transfer is the physical phenomenon of energy transfer in the form of electromagnetic radiation. The propagation of radiation through a medium is affected by absorption, emission, and scattering processes. The equation of radiative trans ...
between two objects, the equation is as follows:
where
* is the heat flux
Heat flux or thermal flux, sometimes also referred to as ''heat flux density'', heat-flow density or ''heat flow rate intensity'' is a flow of energy per unit area per unit time. In SI its units are watts per square metre (W/m2). It has both a ...
,
* is the emissivity
The emissivity of the surface of a material is its effectiveness in emitting energy as thermal radiation. Thermal radiation is electromagnetic radiation that most commonly includes both visible radiation (light) and infrared radiation, which is n ...
(unity for a black body
A black body or blackbody is an idealized physical body that absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence. The name "black body" is given because it absorbs all colors of light. A black body ...
),
* is the Stefan–Boltzmann constant
The Stefan–Boltzmann constant (also Stefan's constant), a physical constant denoted by the Greek letter ''σ'' (sigma), is the constant of proportionality in the Stefan–Boltzmann law: "the total intensity radiated over all wavelengths inc ...
,
* is the view factor between two surfaces a and b, and
* and are the absolute temperatures (in kelvin
The kelvin, symbol K, is the primary unit of temperature in the International System of Units (SI), used alongside its prefixed forms and the degree Celsius. It is named after the Belfast-born and University of Glasgow-based engineer and ...
s or degrees Rankine
The Rankine scale () is an absolute scale of thermodynamic temperature named after the University of Glasgow engineer and physicist Macquorn Rankine, who proposed it in 1859.
History
Similar to the Kelvin scale, which was first proposed in 18 ...
) for the two objects.
The blackbody limit established by the Stefan-Boltzmann equation can be exceeded when the objects exchanging thermal radiation or the distances separating them are comparable in scale or smaller than the dominant thermal wavelength. The study of these cases is called near-field radiative heat transfer
Near-field radiative heat transfer (NFRHT) is a branch of radiative heat transfer which deals with situations for which the objects and/or distances separating objects are comparable or smaller in scale or to the dominant wavelength of thermal ra ...
.
Radiation from the sun, or solar radiation, can be harvested for heat and power. Unlike conductive and convective forms of heat transfer, thermal radiation – arriving within a narrow angle i.e. coming from a source much smaller than its distance – can be concentrated in a small spot by using reflecting mirrors, which is exploited in concentrating solar power
Concentrated solar power (CSP, also known as concentrating solar power, concentrated solar thermal) systems generate solar power by using mirrors or lenses to concentrate a large area of sunlight into a receiver. Electricity is generated when ...
generation or a burning glass
A burning glass or burning lens is a large convex lens that can concentrate the sun's rays onto a small area, heating up the area and thus resulting in ignition of the exposed surface. Burning mirrors achieve a similar effect by using reflecting ...
. For example, the sunlight reflected from mirrors heats the PS10 solar power tower
The PS10 Solar Power Plant ( es, Planta Solar 10), is the world's first commercial concentrating solar power tower operating near Seville, in Andalusia, Spain. The 11 megawatt (MW) solar power tower produces electricity with 624 large movab ...
and during the day it can heat water to .
The reachable temperature at the target is limited by the temperature of the hot source of radiation. (T4-law lets the reverse-flow of radiation back to the source rise.) The (on its surface) somewhat 4000 K hot sun
The Sun is the star at the center of the Solar System. It is a nearly perfect ball of hot plasma, heated to incandescence by nuclear fusion reactions in its core. The Sun radiates this energy mainly as light, ultraviolet, and infrared radi ...
allows to reach coarsely 3000 K (or 3000 °C, which is about 3273 K) at a small probe in the focus spot of a big concave, concentrating mirror of the Mont-Louis Solar Furnace
The Mont-Louis Solar Furnace is an experimental solar furnace - a solar thermal energy facility that was built in 1949. It was the first facility of its kind in the world, and was a precursor of the Odeillo Solar Furnace. It provides a thermal ...
in France.
Phase transition
Phase transition
In chemistry, thermodynamics, and other related fields, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. Commonly the term is used to refer to changes among the basic states ...
or phase change, takes place in a thermodynamic system
A thermodynamic system is a body of matter and/or radiation, confined in space by walls, with defined permeabilities, which separate it from its surroundings. The surroundings may include other thermodynamic systems, or physical systems that are ...
from one phase or state of matter
In physics, a state of matter is one of the distinct forms in which matter can exist. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma. Many intermediate states are known to exist, such as liquid crystal, ...
to another one by heat transfer. Phase change examples are the melting of ice or the boiling of water.
The Mason equation explains the growth of a water droplet based on the effects of heat transport on evaporation
Evaporation is a type of vaporization that occurs on the surface of a liquid as it changes into the gas phase. High concentration of the evaporating substance in the surrounding gas significantly slows down evaporation, such as when h ...
and condensation.
Phase transitions involve the four fundamental states of matter
In physics, a state of matter is one of the distinct forms in which matter can exist. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma. Many intermediate states are known to exist, such as liquid crystal, ...
:
*Solid
Solid is one of the four fundamental states of matter (the others being liquid, gas, and plasma). The molecules in a solid are closely packed together and contain the least amount of kinetic energy. A solid is characterized by structur ...
– Deposition, freezing and solid to solid transformation.
* Gas – Boiling / evaporation, recombination / deionization
Purified water is water that has been mechanically filtered or processed to remove impurities and make it suitable for use. Distilled water was, formerly, the most common form of purified water, but, in recent years, water is more frequently puri ...
, and sublimation.
*Liquid
A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. As such, it is one of the four fundamental states of matter (the others being solid, gas, ...
– Condensation and melting / fusion.
* Plasma – Ionization
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecul ...
.
Boiling
Theboiling point
The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid and the liquid changes into a vapor.
The boiling point of a liquid varies depending upon the surrounding env ...
of a substance is the temperature at which the vapor pressure
Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed pha ...
of the liquid equals the pressure surrounding the liquid and the liquid evaporates
Evaporation is a type of vaporization that occurs on the surface of a liquid as it changes into the gas phase. High concentration of the evaporating substance in the surrounding gas significantly slows down evaporation, such as when humidi ...
resulting in an abrupt change in vapor volume.
In a closed system
A closed system is a natural physical system that does not allow transfer of matter in or out of the system, although — in contexts such as physics, chemistry or engineering — the transfer of energy (''e.g.'' as work or heat) is allowed.
In ...
, ''saturation temperature'' and ''boiling point'' mean the same thing. The saturation temperature is the temperature for a corresponding saturation pressure at which a liquid boils into its vapor phase. The liquid can be said to be saturated with thermal energy. Any addition of thermal energy results in a phase transition.
At standard atmospheric pressure and low temperatures, no boiling occurs and the heat transfer rate is controlled by the usual single-phase mechanisms. As the surface temperature is increased, local boiling occurs and vapor bubbles nucleate, grow into the surrounding cooler fluid, and collapse. This is ''sub-cooled nucleate boiling'', and is a very efficient heat transfer mechanism. At high bubble generation rates, the bubbles begin to interfere and the heat flux no longer increases rapidly with surface temperature (this is the departure from nucleate boiling Nucleate boiling is a type of boiling that takes place when the surface temperature is hotter than the saturated fluid temperature by a certain amount but where the heat flux is below the critical heat flux. For water, as shown in the graph below, ...
, or DNB).
At similar standard atmospheric pressure and high temperatures, the hydrodynamically-quieter regime of film boiling
The Leidenfrost effect is a physical phenomenon in which a liquid, close to a surface that is significantly hotter than the liquid's boiling point, produces an insulating vapor layer that keeps the liquid from boiling rapidly. Because of this re ...
is reached. Heat fluxes across the stable vapor layers are low, but rise slowly with temperature. Any contact between fluid and the surface that may be seen probably leads to the extremely rapid nucleation of a fresh vapor layer ("spontaneous nucleation
In thermodynamics, nucleation is the first step in the formation of either a new thermodynamic phase or structure via self-assembly or self-organization within a substance or mixture. Nucleation is typically defined to be the process that deter ...
"). At higher temperatures still, a maximum in the heat flux is reached (the critical heat flux
Critical heat flux (CHF) describes the thermal limit of a phenomenon where a phase change occurs during heating (such as bubbles forming on a metal surface used to heat water), which suddenly decreases the efficiency of heat transfer, thus causin ...
, or CHF).
The Leidenfrost Effect demonstrates how nucleate boiling slows heat transfer due to gas bubbles on the heater's surface. As mentioned, gas-phase thermal conductivity is much lower than liquid-phase thermal conductivity, so the outcome is a kind of "gas thermal barrier".
Condensation
Condensation
Condensation is the change of the state of matter from the gas phase into the liquid phase, and is the reverse of vaporization. The word most often refers to the water cycle. It can also be defined as the change in the state of water vapo ...
occurs when a vapor is cooled and changes its phase to a liquid. During condensation, the latent heat of vaporization must be released. The amount of the heat is the same as that absorbed during vaporization at the same fluid pressure.
There are several types of condensation:
* Homogeneous condensation, as during a formation of fog.
* Condensation in direct contact with subcooled liquid.
* Condensation on direct contact with a cooling wall of a heat exchanger: This is the most common mode used in industry: Dropwise condensation is difficult to sustain reliably; therefore, industrial equipment is normally designed to operate in filmwise condensation mode.
Melting

Melting
Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. This occurs when the internal energy of the solid increases, typically by the application of heat or pressure, which in ...
is a thermal process that results in the phase transition of a substance from a solid
Solid is one of the four fundamental states of matter (the others being liquid, gas, and plasma). The molecules in a solid are closely packed together and contain the least amount of kinetic energy. A solid is characterized by structur ...
to a liquid
A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. As such, it is one of the four fundamental states of matter (the others being solid, gas, ...
. The internal energy
The internal energy of a thermodynamic system is the total energy contained within it. It is the energy necessary to create or prepare the system in its given internal state, and includes the contributions of potential energy and internal kinet ...
of a substance is increased, typically with in heat or pressure, resulting in a rise of its temperature to the melting point
The melting point (or, rarely, liquefaction point) of a substance is the temperature at which it changes state from solid to liquid. At the melting point the solid and liquid phase exist in equilibrium. The melting point of a substance depen ...
, at which the ordering of ionic or molecular entities in the solid breaks down to a less ordered state and the solid liquefies. Molten substances generally have reduced viscosity with elevated temperature; an exception to this maxim is the element sulfur
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formul ...
, whose viscosity increases to a point due to polymerization
In polymer chemistry, polymerization (American English), or polymerisation (British English), is a process of reacting monomer molecules together in a chemical reaction to form polymer chains or three-dimensional networks. There are many f ...
and then decreases with higher temperatures in its molten state.
Modeling approaches
Heat transfer can be modeled in various ways.Heat equation
Theheat equation
In mathematics and physics, the heat equation is a certain partial differential equation. Solutions of the heat equation are sometimes known as caloric functions. The theory of the heat equation was first developed by Joseph Fourier in 1822 for ...
is an important partial differential equation
In mathematics, a partial differential equation (PDE) is an equation which imposes relations between the various partial derivatives of a multivariable function.
The function is often thought of as an "unknown" to be solved for, similarly to h ...
that describes the distribution of heat (or variation in temperature) in a given region over time. In some cases, exact solutions of the equation are available; in other cases the equation must be solved numerically using computational methods such as DEM-based models for thermal/reacting particulate systems (as critically reviewed by Peng et al.).
Lumped system analysis
Lumped system analysis often reduces the complexity of the equations to one first-order linear differential equation, in which case heating and cooling are described by a simple exponential solution, often referred to asNewton's law of cooling
In the study of heat transfer, Newton's law of cooling is a physical law which states that
The rate of heat loss of a body is directly proportional to the difference in the temperatures between the body and its environment.
The law is frequently q ...
.
System analysis by the lumped capacitance model
The lumped-element model (also called lumped-parameter model, or lumped-component model) simplifies the description of the behaviour of spatially distributed physical systems, such as electrical circuits, into a topology consisting of discrete e ...
is a common approximation in transient conduction that may be used whenever heat conduction within an object is much faster than heat conduction across the boundary of the object. This is a method of approximation that reduces one aspect of the transient conduction system—that within the object—to an equivalent steady state system. That is, the method assumes that the temperature within the object is completely uniform, although its value may be changing in time.
In this method, the ratio of the conductive heat resistance within the object to the convective heat transfer resistance across the object's boundary, known as the ''Biot number
The Biot number (Bi) is a dimensionless quantity used in heat transfer calculations. It is named after the eighteenth century French physicist Jean-Baptiste Biot (1774–1862), and gives a simple index of the ratio of the thermal resistances ''ins ...
'', is calculated. For small Biot numbers, the approximation of ''spatially uniform temperature within the object'' can be used: it can be presumed that heat transferred into the object has time to uniformly distribute itself, due to the lower resistance to doing so, as compared with the resistance to heat entering the object.
Climate models
Climate models study the radiant heat transfer by using quantitative methods to simulate the interactions of the atmosphere, oceans, land surface, and ice.Engineering
 Heat transfer has broad application to the functioning of numerous devices and systems. Heat-transfer principles may be used to preserve, increase, or decrease temperature in a wide variety of circumstances. Heat transfer methods are used in numerous disciplines, such as
Heat transfer has broad application to the functioning of numerous devices and systems. Heat-transfer principles may be used to preserve, increase, or decrease temperature in a wide variety of circumstances. Heat transfer methods are used in numerous disciplines, such as automotive engineering
Automotive engineering, along with aerospace engineering and naval architecture, is a branch of vehicle engineering, incorporating elements of mechanical, electrical, electronic, software, and safety engineering as applied to the design, manufac ...
, thermal management of electronic devices and systems
All electronic devices and circuitry generate excess heat and thus require thermal management to improve reliability and prevent premature failure. The amount of heat output is equal to the power input, if there are no other energy int ...
, climate control
Heating, ventilation, and air conditioning (HVAC) is the use of various technologies to control the temperature, humidity, and purity of the air in an enclosed space. Its goal is to provide thermal comfort and acceptable indoor air quality. HV ...
, insulation, materials processing
In engineering, a process is a series of interrelated tasks that, together, transform inputs into a given output. These tasks may be carried out by people, nature or machines using various resources; an engineering process must be considered in t ...
, chemical engineering
Chemical engineering is an engineering field which deals with the study of operation and design of chemical plants as well as methods of improving production. Chemical engineers develop economical commercial processes to convert raw materials in ...
and power station
A power station, also referred to as a power plant and sometimes generating station or generating plant, is an industrial facility for the generation of electric power. Power stations are generally connected to an electrical grid.
Many ...
engineering.
Insulation, radiance and resistance
Thermal insulators are materials specifically designed to reduce the flow of heat by limiting conduction, convection, or both.Thermal resistance
Thermal resistance is a heat property and a measurement of a temperature difference by which an object or material resists a heat flow. Thermal resistance is the reciprocal of thermal conductance.
* (Absolute) thermal resistance ''R'' in kelvi ...
is a heat property and the measurement by which an object or material resists to heat flow (heat per time unit or thermal resistance) to temperature difference.
Radiance
In radiometry, radiance is the radiant flux emitted, reflected, transmitted or received by a given surface, per unit solid angle per unit projected area. Radiance is used to characterize diffuse emission and reflection of electromagnetic radiati ...
or spectral radiance are measures of the quantity of radiation that passes through or is emitted. Radiant barrier
A radiant barrier is a type of building material that reflects thermal radiation and reduces heat transfer. Because thermal energy is also transferred by conduction and convection, in addition radiation, radiant barriers are often supplemented wit ...
s are materials that reflect radiation, and therefore reduce the flow of heat from radiation sources. Good insulators are not necessarily good radiant barriers, and vice versa. Metal, for instance, is an excellent reflector and a poor insulator.
The effectiveness of a radiant barrier is indicated by its reflectivity, which is the fraction of radiation reflected. A material with a high reflectivity (at a given wavelength) has a low emissivity (at that same wavelength), and vice versa. At any specific wavelength, reflectivity=1 - emissivity. An ideal radiant barrier would have a reflectivity of 1, and would therefore reflect 100 percent of incoming radiation. Vacuum flasks, or Dewars, are silvered
Silvering is the chemical process of coating a non-conductive substrate such as glass with a reflective substance, to produce a mirror. While the metal is often silver, the term is used for the application of any reflective metal.
Process
Mos ...
to approach this ideal. In the vacuum of space, satellites use multi-layer insulation, which consists of many layers of aluminized (shiny) Mylar
BoPET (biaxially-oriented polyethylene terephthalate) is a polyester film made from stretched polyethylene terephthalate (PET) and is used for its high tensile strength, chemical and dimensional stability, transparency, reflectivity, gas and a ...
to greatly reduce radiation heat transfer and control satellite temperature.
Devices
Aheat engine
In thermodynamics and engineering, a heat engine is a system that converts heat to mechanical energy, which can then be used to do mechanical work. It does this by bringing a working substance from a higher state temperature to a lower stat ...
is a system that performs the conversion of a flow of thermal energy
The term "thermal energy" is used loosely in various contexts in physics and engineering. It can refer to several different well-defined physical concepts. These include the internal energy or enthalpy of a body of matter and radiation; heat, de ...
(heat) to mechanical energy
In physical sciences, mechanical energy is the sum of potential energy and kinetic energy. The principle of conservation of mechanical energy states that if an isolated system is subject only to conservative forces, then the mechanical energy is ...
to perform mechanical work
In physics, work is the energy transferred to or from an object via the application of force along a displacement. In its simplest form, for a constant force aligned with the direction of motion, the work equals the product of the force stre ...
.
A thermocouple
A thermocouple, also known as a "thermoelectrical thermometer", is an electrical device consisting of two dissimilar electrical conductors forming an electrical junction. A thermocouple produces a temperature-dependent voltage as a result of th ...
is a temperature-measuring device and widely used type of temperature sensor for measurement and control, and can also be used to convert heat into electric power.
A thermoelectric cooler
Thermoelectric cooling uses the Peltier effect to create a heat flux at the junction of two different types of materials. A Peltier cooler, heater, or thermoelectric heat pump is a solid-state active heat pump which transfers heat from one side o ...
is a solid state electronic device that pumps (transfers) heat from one side of the device to the other when electric current is passed through it. It is based on the Peltier effect
The thermoelectric effect is the direct conversion of temperature differences to electric voltage and vice versa via a thermocouple. A thermoelectric device creates a voltage when there is a different temperature on each side. Conversely, when ...
.
A thermal diode or thermal rectifier is a device that causes heat to flow preferentially in one direction.
Heat exchangers
Aheat exchanger
A heat exchanger is a system used to transfer heat between a source and a working fluid. Heat exchangers are used in both cooling and heating processes. The fluids may be separated by a solid wall to prevent mixing or they may be in direct conta ...
is used for more efficient heat transfer or to dissipate heat. Heat exchangers are widely used in refrigeration
The term refrigeration refers to the process of removing heat from an enclosed space or substance for the purpose of lowering the temperature.International Dictionary of Refrigeration, http://dictionary.iifiir.org/search.phpASHRAE Terminology, ht ...
, air conditioning
Air conditioning, often abbreviated as A/C or AC, is the process of removing heat from an enclosed space to achieve a more comfortable interior environment (sometimes referred to as 'comfort cooling') and in some cases also strictly controlling ...
, space heating, power generation
Electricity generation is the process of generating electric power from sources of primary energy. For utilities in the electric power industry, it is the stage prior to its delivery ( transmission, distribution, etc.) to end users or its stor ...
, and chemical processing. One common example of a heat exchanger is a car's radiator, in which the hot coolant fluid is cooled by the flow of air over the radiator's surface.
Common types of heat exchanger flows include parallel flow, counter flow, and cross flow. In parallel flow, both fluids move in the same direction while transferring heat; in counter flow, the fluids move in opposite directions; and in cross flow, the fluids move at right angle
In geometry and trigonometry, a right angle is an angle of exactly 90 degrees or radians corresponding to a quarter turn. If a ray is placed so that its endpoint is on a line and the adjacent angles are equal, then they are right angles. Th ...
s to each other. Common types of heat exchangers include shell and tube, double pipe, extruded finned pipe, spiral fin pipe, u-tube, and stacked plate. Each type has certain advantages and disadvantages over other types.
A heat sink
A heat sink (also commonly spelled heatsink) is a passive heat exchanger that transfers the heat generated by an electronic or a mechanical device to a fluid medium, often air or a liquid coolant, where it is dissipated away from the device, ...
is a component that transfers heat generated within a solid material to a fluid medium, such as air or a liquid. Examples of heat sinks are the heat exchangers used in refrigeration and air conditioning systems or the radiator in a car. A heat pipe
A heat pipe is a heat-transfer device that employs phase transition to transfer heat between two solid interfaces.
At the hot interface of a heat pipe, a volatile liquid in contact with a thermally conductive solid surface turns into a vapor b ...
is another heat-transfer device that combines thermal conductivity and phase transition to efficiently transfer heat between two solid interfaces.
Applications
Architecture
Efficient energy use
Efficient energy use, sometimes simply called energy efficiency, is the process of reducing the amount of energy required to provide products and services. For example, insulating a building allows it to use less heating and cooling energy to ...
is the goal to reduce the amount of energy required in heating or cooling. In architecture, condensation and air current
In meteorology, air currents are concentrated areas of winds. They are mainly due to differences in atmospheric pressure or temperature. They are divided into horizontal and vertical currents; both are present at mesoscale while horizontal ones ...
s can cause cosmetic or structural damage. An energy audit
An energy audit is an inspection survey and an analysis of energy flows for energy conservation in a building. It may include a process or system to reduce the amount of energy input into the system without negatively affecting the output. In co ...
can help to assess the implementation of recommended corrective procedures. For instance, insulation improvements, air sealing of structural leaks or the addition of energy-efficient windows and doors.
* Smart meter
A smart meter is an electronic device that records information such as consumption of electric energy, voltage levels, current, and power factor. Smart meters communicate the information to the consumer for greater clarity of consumption be ...
is a device that records electric energy consumption in intervals.
* Thermal transmittance
Thermal transmittance is the rate of transfer of heat through matter. The thermal transmittance of a material (such as insulation or concrete) or an assembly (such as a wall or window) is expressed as a U-value. The thermal insulance of a struct ...
is the rate of transfer of heat through a structure divided by the difference in temperature across the structure. It is expressed in watts per square meter per kelvin, or W/(m2K). Well-insulated parts of a building have a low thermal transmittance, whereas poorly-insulated parts of a building have a high thermal transmittance.
* Thermostat
A thermostat is a regulating device component which senses the temperature of a physical system and performs actions so that the system's temperature is maintained near a desired setpoint (control system), setpoint.
Thermostats are used i ...
is a device to monitor and control temperature.
Climate engineering

Climate engineering
Climate engineering (also called geoengineering) is a term used for both carbon dioxide removal (CDR) and solar radiation management (SRM), also called solar geoengineering, when applied at a planetary scale.IPCC (2022Chapter 1: Introduction and ...
consists of carbon dioxide removal
Carbon dioxide removal (CDR), also known as negative emissions, is a process in which carbon dioxide gas () is removed from the atmosphere and sequestered for long periods of time. Similarly, greenhouse gas removal (GGR) or negative greenh ...
and solar radiation management
Solar geoengineering, or solar radiation modification (SRM), is a type of climate engineering in which sunlight (solar radiation) would be reflected back to outer space to limit or reverse human-caused climate change. It is not a substitute for ...
. Since the amount of carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is t ...
determines the radiative balance
Earth's energy budget accounts for the balance between the energy that Earth receives from the Sun and the energy the Earth loses back into outer space. Smaller energy sources, such as Earth's internal heat, are taken into consideration, but ma ...
of Earth atmosphere, carbon dioxide removal techniques can be applied to reduce the radiative forcing
Radiative forcing (or climate forcing) is the change in energy flux in the atmosphere caused by natural or anthropogenic factors of climate change as measured by watts / metre2. It is a scientific concept used to quantify and compare the extern ...
. Solar radiation management is the attempt to absorb less solar radiation to offset the effects of greenhouse gases
A greenhouse gas (GHG or GhG) is a gas that absorbs and emits radiant energy within the thermal infrared range, causing the greenhouse effect. The primary greenhouse gases in Earth's atmosphere are water vapor (), carbon dioxide (), methane ...
.
An alternative method is passive daytime radiative cooling
Passive daytime radiative cooling (PDRC) is a renewable cooling method proposed as a solution to global warming of enhancing terrestrial heat flow to outer space through the installation of thermally-emissive surfaces on Earth that require zer ...
, which enhances terrestrial heat flow to outer space through the infrared window
The infrared atmospheric window refers to a region of the Infrared spectrum where there is relatively little absorption of terrestrial thermal radiation by atmospheric gases. The window plays an important role in the atmospheric greenhouse effe ...
(8–13 µm). Rather than merely blocking solar radiation, this method increases outgoing longwave infrared (LWIR) thermal radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of particles in matter. Thermal radiation is generated when heat from the movement of charges in the material (electrons and protons in common forms of matter) i ...
heat transfer with the extremely cold temperature of outer space (~2.7 K) to lower ambient temperatures while requiring zero energy input.
Greenhouse effect
greenhouse effect
The greenhouse effect is a process that occurs when energy from a planet's host star goes through the planet's atmosphere and heats the planet's surface, but greenhouse gases in the atmosphere prevent some of the heat from returning directly ...
is a process by which thermal radiation from a planetary surface is absorbed by atmospheric greenhouse gases, and is re-radiated in all directions. Since part of this re-radiation is back towards the surface and the lower atmosphere, it results in an elevation of the average surface temperature above what it would be in the absence of the gases.
Heat transfer in the human body
The principles of heat transfer in engineering systems can be applied to the human body in order to determine how the body transfers heat. Heat is produced in the body by the continuous metabolism of nutrients which provides energy for the systems of the body. The human body must maintain a consistent internal temperature in order to maintain healthy bodily functions. Therefore, excess heat must be dissipated from the body to keep it from overheating. When a person engages in elevated levels of physical activity, the body requires additional fuel which increases the metabolic rate and the rate of heat production. The body must then use additional methods to remove the additional heat produced in order to keep the internal temperature at a healthy level. Heat transfer by convection is driven by the movement of fluids over the surface of the body. This convective fluid can be either a liquid or a gas. For heat transfer from the outer surface of the body, the convection mechanism is dependent on the surface area of the body, the velocity of the air, and the temperature gradient between the surface of the skin and the ambient air.Cengel, Yunus A. and Ghajar, Afshin J. "Heat and Mass Transfer: Fundamentals and Applications", McGraw-Hill, 4th Edition, 2010. The normal temperature of the body is approximately 37 °C. Heat transfer occurs more readily when the temperature of the surroundings is significantly less than the normal body temperature. This concept explains why a person feels cold when not enough covering is worn when exposed to a cold environment. Clothing can be considered an insulator which provides thermal resistance to heat flow over the covered portion of the body. This thermal resistance causes the temperature on the surface of the clothing to be less than the temperature on the surface of the skin. This smaller temperature gradient between the surface temperature and the ambient temperature will cause a lower rate of heat transfer than if the skin were not covered. In order to ensure that one portion of the body is not significantly hotter than another portion, heat must be distributed evenly through the bodily tissues. Blood flowing through blood vessels acts as a convective fluid and helps to prevent any buildup of excess heat inside the tissues of the body. This flow of blood through the vessels can be modeled as pipe flow in an engineering system. The heat carried by the blood is determined by the temperature of the surrounding tissue, the diameter of the blood vessel, the thickness of the fluid, velocity of the flow, and the heat transfer coefficient of the blood. The velocity, blood vessel diameter, and the fluid thickness can all be related with theReynolds Number
In fluid mechanics, the Reynolds number () is a dimensionless quantity that helps predict fluid flow patterns in different situations by measuring the ratio between inertial and viscous forces. At low Reynolds numbers, flows tend to be dom ...
, a dimensionless number used in fluid mechanics to characterize the flow of fluids.
Latent heat
Latent heat (also known as latent energy or heat of transformation) is energy released or absorbed, by a body or a thermodynamic system, during a constant-temperature process — usually a first-order phase transition.
Latent heat can be underst ...
loss, also known as evaporative heat loss, accounts for a large fraction of heat loss from the body. When the core temperature of the body increases, the body triggers sweat glands in the skin to bring additional moisture to the surface of the skin. The liquid is then transformed into vapor which removes heat from the surface of the body. The rate of evaporation heat loss is directly related to the vapor pressure
Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed pha ...
at the skin surface and the amount of moisture present on the skin. Therefore, the maximum of heat transfer will occur when the skin is completely wet. The body continuously loses water by evaporation but the most significant amount of heat loss occurs during periods of increased physical activity.
Cooling techniques
Evaporative cooling

Evaporative cooling
An evaporative cooler (also known as evaporative air conditioner, swamp cooler, swamp box, desert cooler and wet air cooler) is a device that cools air through the evaporation of water. Evaporative cooling differs from other air conditioning ...
happens when water vapor is added to the surrounding air. The energy needed to evaporate the water is taken from the air in the form of sensible heat and converted into latent heat, while the air remains at a constant enthalpy
Enthalpy , a property of a thermodynamic system, is the sum of the system's internal energy and the product of its pressure and volume. It is a state function used in many measurements in chemical, biological, and physical systems at a constant ...
. Latent heat describes the amount of heat that is needed to evaporate the liquid; this heat comes from the liquid itself and the surrounding gas and surfaces. The greater the difference between the two temperatures, the greater the evaporative cooling effect. When the temperatures are the same, no net evaporation of water in air occurs; thus, there is no cooling effect.
Laser cooling
Inquantum physics
Quantum mechanics is a fundamental theory in physics that provides a description of the physical properties of nature at the scale of atoms and subatomic particles. It is the foundation of all quantum physics including quantum chemistry, qua ...
, laser cooling
Laser cooling includes a number of techniques in which atoms, molecules, and small mechanical systems are cooled, often approaching temperatures near absolute zero. Laser cooling techniques rely on the fact that when an object (usually an atom) ...
is used to achieve temperatures of near absolute zero
Absolute zero is the lowest limit of the thermodynamic temperature scale, a state at which the enthalpy and entropy of a cooled ideal gas reach their minimum value, taken as zero kelvin. The fundamental particles of nature have minimum vibra ...
(−273.15 °C, −459.67 °F) of atomic and molecular samples to observe unique quantum effects that can only occur at this heat level.
* Doppler cooling is the most common method of laser cooling.
* Sympathetic cooling is a process in which particles of one type cool particles of another type. Typically, atomic ions that can be directly laser-cooled are used to cool nearby ions or atoms. This technique allows cooling of ions and atoms that cannot be laser cooled directly.
Magnetic cooling
Magnetic evaporative cooling
Evaporative cooling is an atomic physics technique to achieve high phase space densities which optical cooling techniques alone typically can not reach.
Atoms trapped in optical or magnetic traps can be evaporatively cooled via two primary mechan ...
is a process for lowering the temperature of a group of atoms, after pre-cooled by methods such as laser cooling. Magnetic refrigeration cools below 0.3K, by making use of the magnetocaloric effect
Magnetic refrigeration is a cooling technology based on the magnetocaloric effect. This technique can be used to attain extremely low temperatures, as well as the ranges used in common refrigerators.
A magnetocaloric material warms up when a m ...
.
Radiative cooling
Radiative cooling
In the study of heat transfer, radiative cooling is the process by which a body loses heat by thermal radiation. As Planck's law describes, every physical body spontaneously and continuously emits electromagnetic radiation.
Radiative coolin ...
is the process by which a body loses heat by radiation. Outgoing energy is an important effect in the Earth's energy budget
Earth's energy budget accounts for the balance between the energy that Earth receives from the Sun and the energy the Earth loses back into outer space. Smaller energy sources, such as Earth's internal heat, are taken into consideration, but m ...
. In the case of the Earth-atmosphere system, it refers to the process by which long-wave (infrared) radiation is emitted to balance the absorption of short-wave (visible) energy from the Sun. The thermosphere (top of atmosphere) cools to space primarily by infrared energy radiated by carbon dioxide () at 15 μm and by nitric oxide (NO) at 5.3 μm.The global infrared energy budget of the thermosphere from 1947 to 2016 and implications for solar variability
Martin G. Mlynczak Linda A. Hunt James M. Russell III B. Thomas Marshall Christopher J. Mertens R. Earl Thompson https://agupubs.onlinelibrary.wiley.com/doi/full/10.1002/2016GL070965 Convective transport of heat and evaporative transport of latent heat both remove heat from the surface and redistribute it in the atmosphere.
Thermal energy storage
Thermal energy storage
Thermal energy storage (TES) is achieved with widely different technologies. Depending on the specific technology, it allows excess thermal energy to be stored and used hours, days, months later, at scales ranging from the individual process, ...
includes technologies for collecting and storing energy for later use. It may be employed to balance energy demand between day and nighttime. The thermal reservoir may be maintained at a temperature above or below that of the ambient environment. Applications include space heating, domestic or process hot water systems, or generating electricity.
See also
*Combined forced and natural convection
In fluid thermodynamics, combined forced convection and natural convection, or mixed convection, occurs when natural convection and forced convection mechanisms act together to transfer heat. This is also defined as situations where both pres ...
*Heat capacity
Heat capacity or thermal capacity is a physical property of matter, defined as the amount of heat to be supplied to an object to produce a unit change in its temperature. The SI unit of heat capacity is joule per kelvin (J/K).
Heat cap ...
* Heat transfer physics
*Stefan–Boltzmann law
The Stefan–Boltzmann law describes the power radiated from a black body in terms of its temperature. Specifically, the Stefan–Boltzmann law states that the total energy radiated per unit surface area of a black body across all wavelengths ...
* Thermal contact conductance
*Thermal physics
Thermal physics is the combined study of thermodynamics, statistical mechanics, and kinetic theory of gases. This umbrella-subject is typically designed for physics students and functions to provide a general introduction to each of three core hea ...
* Thermal resistance in electronics
*Heat transfer enhancement Heat transfer enhancement is the process of increasing the effectiveness of heat exchangers. This can be achieved when the heat transfer power of a given device is increased or when the pressure losses generated by the device are reduced. A variety ...
References
External links
A Heat Transfer Textbook
- (free download).
Thermal-FluidsPedia
- An online thermal fluids encyclopedia.
- Overview
- a practical example of how heat transfer is used to heat buildings without burning fossil fuels.
{{DEFAULTSORT:Heat Transfer Chemical engineering Mechanical engineering Unit operations Transport phenomena